Drug Concentration Analysis System Market Report 2026

Drug Concentration Analysis System Market Report 2026
Global Outlook – By Product Type (Liquid Chromatography Systems, Mass Spectrometry Systems, Spectrophotometry Systems, Immunoassays), By Sample Type (Blood Or Plasma Or Serum, Urine, Tissue Samples, Saliva, Cell Culture And Bioprocess Samples, Cerebrospinal Fluid (CSF), Other Biological Or Chemical Matrices), By Distribution Channel (Online Distribution, Offline Distribution, Direct Sales, Third Party Distributors, Retail Outlets), By Application (Therapeutic Drug Monitoring, Pharmaceutical Research And Development, Clinical Trials, Bioavailability Or Bioequivalence), By End-User (Hospitals And Clinics, Pharmaceutical Companies, Contract Research Organizations (CROs), Academic And Research Institutions, Homecare Settings) – Market Size, Trends, Strategies, and Forecast to 2035
Drug Concentration Analysis System Market Overview
• Drug Concentration Analysis System market size has reached to $2.46 billion in 2025 • Expected to grow to $3.57 billion in 2030 at a compound annual growth rate (CAGR) of 7.7% • Growth Driver: Rising Demand For Personalized Medicine Fueling The Growth Of The Market Due To Advancements In Genomic Technologies And Data-Driven Women’s Healthcare • Market Trend: Advancing Drug Concentration Analysis With Rapid On-Site Quantitative Monitoring, Point-of-Care Accessibility, And Immediate Clinical Insights • North America was the largest region in 2025 and Asia-Pacific is the fastest growing region.What Is Covered Under Drug Concentration Analysis System Market?
A drug concentration analysis system is an integrated analytical solution that combines instruments, detection technologies, and data processing tools to accurately measure the levels of drugs and their metabolites in biological samples. Its purpose is to support therapeutic drug monitoring, optimize dosing decisions, ensure patient safety, and improve treatment effectiveness. The main product types of drug concentration analysis systems include liquid chromatography systems, mass spectrometry systems, spectrophotometry systems, and immunoassays. Liquid Chromatography Systems refer to analytical instruments used to separate, identify, and quantify compounds in liquid samples based on their chemical interactions. The samples type such as blood, plasma, serum, urine, tissue samples, saliva, cell culture, and cerebrospinal fluid and others. The distribution channels online and offline channels, direct sales, third-party distributors, and retail outlets. The applications including therapeutic drug monitoring, pharmaceutical research and development, clinical trials, and bioavailability or bioequivalence and the key end users, including hospitals and clinics, pharmaceutical companies, contract research organizations, academic and research institutions, and homecare settings.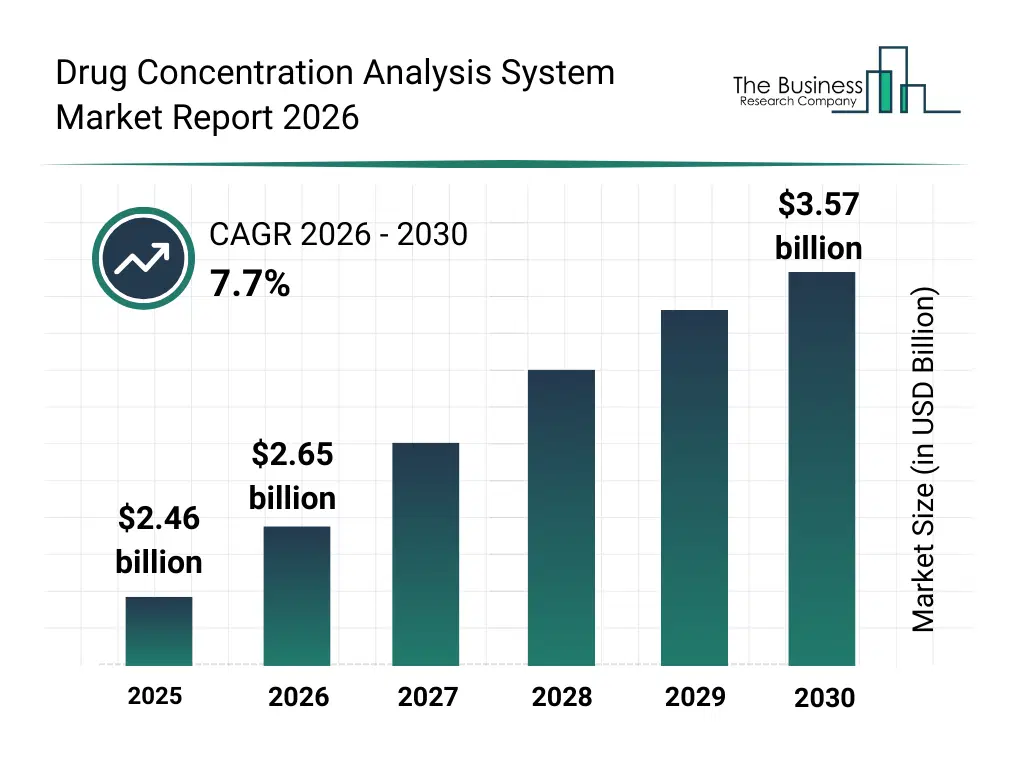
What Is The Drug Concentration Analysis System Market Size and Share 2026?
The drug concentration analysis system market size has grown strongly in recent years. It will grow from $2.46 billion in 2025 to $2.65 billion in 2026 at a compound annual growth rate (CAGR) of 8.0%. The growth in the historic period can be attributed to increasing chronic disease prevalence, rising healthcare expenditure, expanding pharmaceutical research, growing demand for therapeutic drug monitoring, and rising regulatory quality control requirements.What Is The Drug Concentration Analysis System Market Growth Forecast?
The drug concentration analysis system market size is expected to see strong growth in the next few years. It will grow to $3.57 billion in 2030 at a compound annual growth rate (CAGR) of 7.7%. The growth in the forecast period can be attributed to increasing investments in healthcare infrastructure, growing demand for precise drug monitoring, expanding pharmaceutical and biotechnology sectors, rising adoption of personalized medicine, and increasing pharma R&D spending. Major trends in the forecast period include technological advancements in analytical instruments, growth of point-of-care drug concentration systems, integration of automation and high-throughput testing, adoption of advanced bioanalytics methods, and increase in regulatory compliance and quality-assurance testing.Global Drug Concentration Analysis System Market Segmentation
1) By Product Type: Liquid Chromatography Systems, Mass Spectrometry Systems, Spectrophotometry Systems, Immunoassays 2) By Sample Type: Blood Or Plasma Or Serum, Urine, Tissue Samples, Saliva, Cell Culture And Bioprocess Samples, Cerebrospinal Fluid (CSF), Other Biological Or Chemical Matrices 3) By Distribution Channel: Online Distribution, Offline Distribution, Direct Sales, Third Party Distributors, Retail Outlets 4) By Application: Therapeutic Drug Monitoring, Pharmaceutical Research And Development, Clinical Trials, Bioavailability Or Bioequivalence 5) By End-User: Hospitals And Clinics, Pharmaceutical Companies, Contract Research Organizations (CROs), Academic And Research Institutions, Homecare Settings Subsegments: 1) By Liquid Chromatography Systems: High Performance Liquid Chromatography, Ultra High Performance Liquid Chromatography, Fast Protein Liquid Chromatography, Ion Exchange Liquid Chromatography, Size Exclusion Liquid Chromatography 2) By Mass Spectrometry Systems: Quadrupole Mass Spectrometry, Time Of Flight Mass Spectrometry, Ion Trap Mass Spectrometry, Fourier Transform Ion Cyclotron Resonance Mass Spectrometry, Orbitrap Mass Spectrometry 3) By Spectrophotometry Systems: Ultraviolet Visible Spectrophotometry, Infrared Spectrophotometry, Fluorescence Spectrophotometry, Atomic Absorption Spectrophotometry, Raman Spectrophotometry 4) By Immunoassays: Enzyme Linked Immunosorbent Assay, Radioimmunoassay, Chemiluminescent Immunoassay, Fluoroimmunoassay, Lateral Flow ImmunoassayWhat Is The Driver Of The Drug Concentration Analysis System Market?
The rising demand for personalized medicine is expected to propel the growth of the drug concentration analysis system market going forward. Personalized medicine is a medical approach that involves tailoring treatments to individual patients based on their genetic, environmental, and lifestyle factors to optimize outcomes and improve care. The demand for personalized medicine is increasing due to advancements in genomic technologies, which enable the precise identification of genetic variations and tailored treatments for individuals. Drug concentration analysis systems enhance personalized medicine by accurately measuring medication levels in individual patients, enabling tailored dosing and optimized treatment regimens. They improve therapeutic outcomes by minimizing side effects and ensuring each patient receives the most effective and safe medication strategy. For instance, in February 2024, according to the Personalized Medicine Coalition (PMC), a US-based nonprofit organization, the U.S. Food and Drug Administration (FDA) approved 26 new personalized medicines in 2023, a significant increase from the 12 approved in 2022. Therefore, the rising demand for personalized medicine is driving the growth of the drug concentration analysis system industry.Key Players In The Global Drug Concentration Analysis System Market
Major companies operating in the drug concentration analysis system market are F. Hoffmann-La Roche AG, Thermo Fisher Scientific Inc., Abbott Laboratories, Siemens Healthineers AG, Siemens Healthineers AG, Danaher Corporation, Agilent Technologies Inc., BioMérieux Inc., Sartorius AG, Shimadzu Corporation, PerkinElmer Inc., Bruker Corporation, Waters Corporation, Bio-Rad Laboratories Inc., Tecan Group Ltd., Randox Laboratories Ltd., Analytik Jena GmbH & Co. KG, KNAUER Wissenschaftliche Geräte GmbH, Chromsystems Instruments & Chemicals GmbH, American Laboratory Products Company Ltd.Global Drug Concentration Analysis System Market Trends and Insights
Major companies operating in the drug concentration analysis system market are focusing on developing advanced products, such as rapid on-site testing platforms, to boost treatment optimization, enhance personalized care, and reduce time and costs associated with traditional laboratory testing. Rapid on-site testing platforms refer to advanced diagnostic systems that enable immediate, quantitative measurement of drug levels in a clinical setting. For instance, in October 2023, ProciseDx Inc., a US-based in vitro diagnostics company, received de novo FDA clearance for the therapeutic drug monitoring (TDM) tests for adalimumab (Humira and biosimilar Amgevita) and infliximab (Remicade and biosimilars Inflectra and Renflexis). These assays, called Procise ADL and Procise IFX, leverage time-resolved fluorescence resonance energy transfer immunoassay technology to quantify drug concentrations in serum with results delivered in approximately five minutes, enabling near-immediate clinical decisions during patient visits. The launch highlights a significant shift toward point-of-care drug concentration analysis systems that combine quantitative precision with operational simplicity, supporting personalized dosing strategies for patients with conditions such as inflammatory bowel disease.What Are Latest Mergers And Acquisitions In The Drug Concentration Analysis System Market?
In June 2025, SVAR Life Science AB, a Sweden-based provider of clinical diagnostics and laboratory solutions, partnered with Lablytica Life Science Pvt. Ltd. to enhance capabilities in drug concentration analysis systems for research and clinical applications. Through this partnership, SVAR Life Science AB and Lablytica Life Science Pvt. Ltd. aim to combine advanced analytical instrumentation with laboratory expertise to improve pharmacokinetic testing, bioanalysis, and therapeutic monitoring workflows. Lablytica Life Science Pvt. Ltd. is an India-based life sciences and analytical services company.Regional Insights
North America was the largest region in the drug concentration analysis system market in 2025. Asia-Pacific is expected to be the fastest-growing region in the forecast period. The regions covered in this market report are Asia-Pacific, South East Asia, Western Europe, Eastern Europe, North America, South America, Middle East, Africa. The countries covered in this market report are Australia, Brazil, China, France, Germany, India, Indonesia, Japan, Taiwan, Russia, South Korea, UK, USA, Canada, Italy, Spain.What Defines the Drug Concentration Analysis System Market?
The drug concentration analysis system market consists of revenues earned by entities by providing services such as therapeutic drug monitoring, bioanalytical testing, pharmacokinetic analysis, assay development and validation, and clinical laboratory data interpretation services. The market value includes the value of related goods sold by the service provider or included within the service offering. The drug concentration analysis system market also includes sales of analytical instruments, drug concentration analyzers, assay kits and reagents, integrated software platforms, and sample preparation systems. Values in this market are ‘factory gate’ values, that is, the value of goods sold by the manufacturers or creators of the goods, whether to other entities (including downstream manufacturers, wholesalers, distributors, and retailers) or directly to end customers. The value of goods in this market includes related services sold by the creators of the goods.How is Market Value Defined and Measured?
The market value is defined as the revenues that enterprises gain from the sale of goods and/or services within the specified market and geography through sales, grants, or donations in terms of the currency (in USD unless otherwise specified). The revenues for a specified geography are consumption values that are revenues generated by organizations in the specified geography within the market, irrespective of where they are produced. It does not include revenues from resales along the supply chain, either further along the supply chain or as part of other products.What Key Data and Analysis Are Included in the Drug Concentration Analysis System Market Report 2026?
The drug concentration analysis system market research report is one of a series of new reports from The Business Research Company that provides market statistics, including industry global market size, regional shares, competitors with the market share, detailed market segments, market trends and opportunities, and any further data you may need to thrive in the drug concentration analysis system industry. The market research report delivers a complete perspective of everything you need, with an in-depth analysis of the current and future state of the industry.Drug Concentration Analysis System Market Report Forecast Analysis
| Report Attribute | Details |
|---|---|
| Market Size Value In 2026 | $2.65 billion |
| Revenue Forecast In 2035 | $3.57 billion |
| Growth Rate | CAGR of 8.0% from 2026 to 2035 |
| Base Year For Estimation | 2025 |
| Actual Estimates/Historical Data | 2020-2025 |
| Forecast Period | 2026 - 2030 - 2035 |
| Market Representation | Revenue in USD Billion and CAGR from 2026 to 2035 |
| Segments Covered | Product Type, Sample Type, Distribution Channel, Application, End-User |
| Regional Scope | Asia-Pacific, Western Europe, Eastern Europe, North America, South America, Middle East, Africa |
| Country Scope | The countries covered in the report are Australia, Brazil, China, France, Germany, India, ... |
| Key Companies Profiled | F. Hoffmann-La Roche AG, Thermo Fisher Scientific Inc., Abbott Laboratories, Siemens Healthineers AG, Siemens Healthineers AG, Danaher Corporation, Agilent Technologies Inc., BioMérieux Inc., Sartorius AG, Shimadzu Corporation, PerkinElmer Inc., Bruker Corporation, Waters Corporation, Bio-Rad Laboratories Inc., Tecan Group Ltd., Randox Laboratories Ltd., Analytik Jena GmbH & Co. KG, KNAUER Wissenschaftliche Geräte GmbH, Chromsystems Instruments & Chemicals GmbH, American Laboratory Products Company Ltd. |
| Customization Scope | Request for Customization |
| Pricing And Purchase Options | Explore Purchase Options |
Frequently Asked Questions
1) By Product Type: Liquid Chromatography Systems, Mass Spectrometry Systems, Spectrophotometry Systems, Immunoassays
2) By Sample Type: Blood Or Plasma Or Serum, Urine, Tissue Samples, Saliva, Cell Culture And Bioprocess Samples, Cerebrospinal Fluid (CSF), Other Biological Or Chemical Matrices
3) By Distribution Channel: Online Distribution, Offline Distribution, Direct Sales, Third Party Distributors, Retail Outlets
4) By Application: Therapeutic Drug Monitoring, Pharmaceutical Research And Development, Clinical Trials, Bioavailability Or Bioequivalence
5) By End-User: Hospitals And Clinics, Pharmaceutical Companies, Contract Research Organizations (CROs), Academic And Research Institutions, Homecare Settings Subsegments:
1) By Liquid Chromatography Systems: High Performance Liquid Chromatography, Ultra High Performance Liquid Chromatography, Fast Protein Liquid Chromatography, Ion Exchange Liquid Chromatography, Size Exclusion Liquid Chromatography
2) By Mass Spectrometry Systems: Quadrupole Mass Spectrometry, Time Of Flight Mass Spectrometry, Ion Trap Mass Spectrometry, Fourier Transform Ion Cyclotron Resonance Mass Spectrometry, Orbitrap Mass Spectrometry
3) By Spectrophotometry Systems: Ultraviolet Visible Spectrophotometry, Infrared Spectrophotometry, Fluorescence Spectrophotometry, Atomic Absorption Spectrophotometry, Raman Spectrophotometry
4) By Immunoassays: Enzyme Linked Immunosorbent Assay, Radioimmunoassay, Chemiluminescent Immunoassay, Fluoroimmunoassay, Lateral Flow Immunoassay
