
Drug Eluting Balloons Catheters Market Report 2026
Global Outlook – By Product (Coronary Artery Disease, Peripheral Vascular Disease, Drug-Eluting Balloon Catheters), By Drug (Paclitaxel, Sirolimus, Other Drugs), By Raw Material (Nylon, Polyurethane, Other Raw Materials), By End-User (Hospitals And Clinics, Ambulatory Surgical Centers, Catheterization Laboratories) – Market Size, Trends, Strategies, and Forecast to 2035
Drug Eluting Balloons Catheters Market Overview
• Drug Eluting Balloons Catheters market size has reached to $1.65 billion in 2025 • Expected to grow to $2.92 billion in 2030 at a compound annual growth rate (CAGR) of 11.9% • Growth Driver: Expanding Prevalence Of Cardiovascular And Peripheral Diseases Fuels Drug-Eluting Balloon Catheter Market Growth • Market Trend: Introducing Newest-Generation Products For Advancing Coronary Artery Disease Treatment • North America was the largest region in 2025 and Asia-Pacific is the fastest growing region.What Is Covered Under Drug Eluting Balloons Catheters Market?
A drug-eluting balloon catheter is a medical device used to treat coronary artery disease by percutaneous coronary intervention (PCI). It is a semi-compliant angioplasty balloon coated with an anti-proliferative drug that is released into the vessel wall during inflation, which helps to prevent restenosis and reduce the risk of late thrombosis without implanting a permanent foreign object. The main types of products included in drug-eluting balloon catheters are coronary artery disease drug-eluting balloon catheters and peripheral vascular disease drug-eluting balloon catheters. Coronary artery disease drug-eluting balloon catheters refer to balloon catheters designed to deliver antiproliferative drugs directly to the vessel wall via a lipophilic matrix, removing the requirement for a lasting structure such as a metal prosthesis or a persistent polymer. Various types of drugs are used, including paclitaxel, sirolimus, and others. These are made of several raw materials, including nylon, polyurethane, and others, and are used by numerous end-users, such as hospitals and clinics, ambulatory surgical centers, and catheterization laboratories.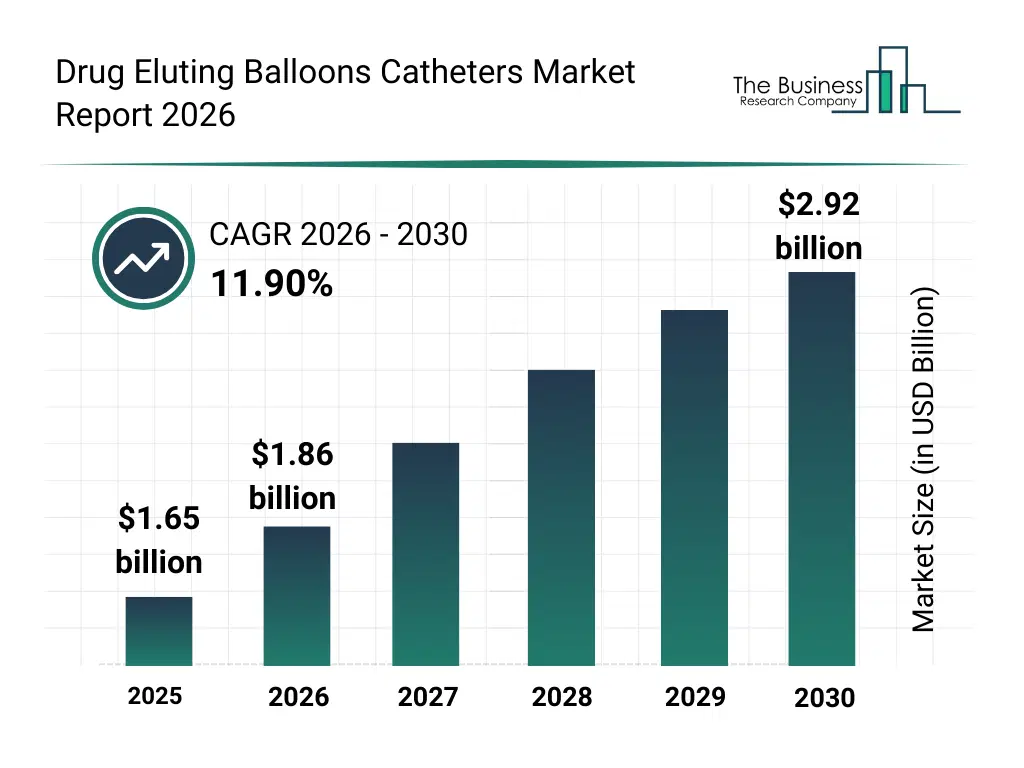
What Is The Drug Eluting Balloons Catheters Market Size and Share 2026?
The drug eluting balloons catheters market size has grown rapidly in recent years. It will grow from $1.65 billion in 2025 to $1.86 billion in 2026 at a compound annual growth rate (CAGR) of 12.5%. The growth in the historic period can be attributed to increasing prevalence of coronary artery disease, growing adoption of percutaneous coronary interventions, advancements in balloon catheter materials, clinical acceptance of paclitaxel-coated devices, expansion of interventional cardiology centers.What Is The Drug Eluting Balloons Catheters Market Growth Forecast?
The drug eluting balloons catheters market size is expected to see rapid growth in the next few years. It will grow to $2.92 billion in 2030 at a compound annual growth rate (CAGR) of 11.9%. The growth in the forecast period can be attributed to increasing demand for minimally invasive vascular therapies, rising adoption of sirolimus-based balloons, expansion of peripheral vascular disease treatments, growing investments in next-generation catheter technologies, increasing focus on long-term patient outcomes. Major trends in the forecast period include increasing adoption of drug-coated balloon technologies, rising use in coronary and peripheral interventions, growing preference for stent-free treatment options, expansion of advanced drug coating techniques, enhanced focus on restenosis prevention.Global Drug Eluting Balloons Catheters Market Segmentation
1) By Product: Coronary Artery Disease, Peripheral Vascular Disease, Drug-Eluting Balloon Catheters 2) By Drug: Paclitaxel, Sirolimus, Other Drugs 3) By Raw Material: Nylon, Polyurethane, Other Raw Materials 4) By End-User: Hospitals And Clinics, Ambulatory Surgical Centers, Catheterization Laboratories Subsegments: 1) By Coronary Artery Disease: Coronary Drug-Eluting Balloon Catheters, Coronary In-Stent Restenosis Drug-Eluting Balloon Catheters, Coronary Small-Vessel Drug-Eluting Balloon Catheters 2) By Peripheral Vascular Disease: Drug-Eluting Balloon Catheters, Femoro-Popliteal Drug-Eluting Balloon Catheters, Below-the-Knee (BTK) Drug-Eluting Balloon Catheters 3) By Drug-Eluting Balloon Catheters: Drug-Eluting Angioplasty Balloon CathetersWhat Is The Driver Of The Drug Eluting Balloons Catheters Market?
The growing prevalence of peripheral and cardiovascular diseases is expected to propel the growth of the drug-eluting balloon catheter market going forward. Cardiovascular diseases (CVDs) refer to a group of conditions that impact the heart and the circulatory system, whereas peripheral artery disease (PAD) specifically refers to a condition where narrowed arteries reduce blood flow primarily to the arms or legs. Drug-eluting balloon catheters are a type of treatment for coronary artery disease (CAD) that uses a semi-compliant angioplasty balloon coated with an anti-proliferative substance that, when it enters the vessel, can have an anti-restenotic effect. For instance, in January 2024, according to the American Heart Association, a US-based non-profit organization, age-adjusted cardiovascular disease mortality rate rose to 233.3 deaths per 100,000 in 2024, reflecting a 4.0% year-on-year increase from 224.4 deaths per 100,000 in 2023. Therefore, the growing prevalence of peripheral and cardiovascular diseases is driving the drug-eluting balloon catheter market.Key Players In The Global Drug Eluting Balloons Catheters Market
Major companies operating in the drug eluting balloons catheters market are Abbott Laboratories, Medtronic PLC, Boston Scientific Corporation, B Braun Melsungen Aktiengesellschaft, Terumo Corporation, Cook Medical LLC, Lepu Medical Technology Co. Ltd., Meril Life Sciences Pvt. Ltd., MicroPort Scientific Corporation, BIOTRONIK SE & Co. KG, Acotec Scientific Co. Ltd., Cordis Corporation, Biosensors International Group Ltd., Zylox-Tonbridge Medical Technology Co. Ltd., Hexacath Limited, Zhejiang Barty Medical Technology Co. Ltd., Cardionovum GmbH, Nano Therapeutics Pvt. Ltd., Tokai Medical Products Inc., Opto Circuits Limited, Acrostak (Schweiz) AG, Concept Medical Inc., Aachen Resonance GmbH, AR Baltic Medical UABGlobal Drug Eluting Balloons Catheters Market Trends and Insights
Major companies operating in the drug-eluting balloon catheter market are increasing their focus on developing the newest-generation products with advanced technologies to gain a competitive edge in the market. For instance, in March 2024, Boston Scientific Corporation, a US-based biomedical/biotechnology engineering firm and multinational manufacturer of medical devices, announced it has received U.S. Food and Drug Administration (FDA) approval for the AGENT Drug-Coated Balloon (DCB). AGENT DCB is the first drug-coated coronary balloon available in the U.S. for treating in-stent restenosis (ISR), where plaque or scar tissue blocks a stented vessel. It features a paclitaxel coating that delivers a therapeutic dose to the vessel wall, reducing ISR recurrence without additional stents or radiation. Clinical trials showed a 49% decrease in heart attack risk at the target site and reported low rates of adverse events.What Are Latest Mergers And Acquisitions In The Drug Eluting Balloons Catheters Market?
In June 2023, Translumina Therapeutics Company, a Germany-based interventional cardiovascular devices company, acquired Blue Medical Devices BV for an undisclosed amount. With this acquisition, Translumina aimed to integrate Blue Medical's unique drug application technology into its existing product lines, broadening its offerings in neurovascular and peripheral interventions and enhancing its drug coating capabilities. Blue Medical Devices BV is a Netherlands-based medical device company specializing in producing drug-eluting balloon catheters.Regional Outlook
North America was the largest region in the drug eluting balloons catheters market in 2025. Asia-Pacific is expected to be the fastest-growing region in the forecast period. The regions covered in this market report are Asia-Pacific, South East Asia, Western Europe, Eastern Europe, North America, South America, Middle East, Africa. The countries covered in this market report are Australia, Brazil, China, France, Germany, India, Indonesia, Japan, Taiwan, Russia, South Korea, UK, USA, Canada, Italy, SpainWhat Defines the Drug Eluting Balloons Catheters Market?
The drug-eluting balloon catheter market consists of sales of everolimus and stellarex. Values in this market are ‘factory gate’ values, that is the value of goods sold by the manufacturers or creators of the goods, whether to other entities (including downstream manufacturers, wholesalers, distributors, and retailers) or directly to end customers. The value of goods in this market includes related services sold by the creators of the goods.How is Market Value Defined and Measured?
The market value is defined as the revenues that enterprises gain from the sale of goods and/or services within the specified market and geography through sales, grants, or donations in terms of the currency (in USD unless otherwise specified). The revenues for a specified geography are consumption values that are revenues generated by organizations in the specified geography within the market, irrespective of where they are produced. It does not include revenues from resales along the supply chain, either further along the supply chain or as part of other products.Drug Eluting Balloons Catheters Market Report Forecast Analysis
| Report Attribute | Details |
|---|---|
| Market Size Value In 2026 | $1.86 billion |
| Revenue Forecast In 2035 | $2.92 billion |
| Growth Rate | CAGR of 12.5% from 2026 to 2035 |
| Base Year For Estimation | 2025 |
| Actual Estimates/Historical Data | 2020-2025 |
| Forecast Period | 2026 - 2030 - 2035 |
| Market Representation | Revenue in USD Billion and CAGR from 2026 to 2035 |
| Segments Covered | Product, Drug, Raw Material, End-User |
| Regional Scope | Asia-Pacific, Western Europe, Eastern Europe, North America, South America, Middle East, Africa |
| Country Scope | The countries covered in the report are Australia, Brazil, China, France, Germany, India, ... |
| Key Companies Profiled | Abbott Laboratories, Medtronic PLC, Boston Scientific Corporation, B Braun Melsungen Aktiengesellschaft, Terumo Corporation, Cook Medical LLC, Lepu Medical Technology Co. Ltd., Meril Life Sciences Pvt. Ltd., MicroPort Scientific Corporation, BIOTRONIK SE & Co. KG, Acotec Scientific Co. Ltd., Cordis Corporation, Biosensors International Group Ltd., Zylox-Tonbridge Medical Technology Co. Ltd., Hexacath Limited, Zhejiang Barty Medical Technology Co. Ltd., Cardionovum GmbH, Nano Therapeutics Pvt. Ltd., Tokai Medical Products Inc., Opto Circuits Limited, Acrostak (Schweiz) AG, Concept Medical Inc., Aachen Resonance GmbH, AR Baltic Medical UAB |
| Customization Scope | Request for Customization |
| Pricing And Purchase Options | Explore Purchase Options |
