
eClinical Solutions Market Report 2026
Global Outlook – By Product (Electronic Data Capture (EDC) And Clinical Data Management Systems (CDMS), Clinical Trial Management Systems (CTMS), Clinical Analytics Platforms, Randomization And Trial Supply Management (RTSM), Clinical Data Integration Platforms, Electronic Clinical Outcome Assessment (eCOA), Safety Solutions, Electronic Trial Master File (eTMF)), By Development Phase (Phase I, Phase II, Phase III, Phase IV), By Delivery Mode (Web-Based and Cloud-Based, Enterprise-Based), By End User (Pharmaceutical and Biopharmaceutical Companies, Contract Research Organizations, Consulting Service Companies, Medical Device Manufacturers, Hospitals, Academic Research Institutions) - Market Size, Trends, And Global Forecast 2026-2035
eClinical Solutions Market Overview
• eClinical Solutions market size has reached to $9.13 billion in 2025 • Expected to grow to $17.45 billion in 2030 at a compound annual growth rate (CAGR) of 13.8% • Growth Driver: Rising Number Of Clinical Trials • Market Trend: EDETEK Inc. Unveils CONFORM Eclinical Version 5.1: An Integrated Solution For Enhanced Efficiency In Clinical Trials • North America was the largest region in 2025.What Is Covered Under eClinical Solutions Market?
eClinical solutions refer to the use of computerized solutions and related procedures to aid clinical trial operations by automating previously laborious tasks. eClinical has evolved to include a wide range of technologies that aim to help with one or more phases of clinical trials, from planning through submissions and data mining. The eclinical solutions are used in the clinical development process by combining clinical technology expertise. The main types of eclinical solutions are electronic data capture (EDC) and clinical data management systems (CDMS), clinical trial management systems (CTMS), clinical analytics platforms, randomization and trial supply management (RTSM), clinical data integration platforms, electronic clinical outcome assessment (ECOA), safety solutions, and electronic trial master file (ETMF). The Electronic Data Capture (EDC) and Clinical Data Management Systems (CDMS) markets refer to a program that allows a person to capture and collect clinical documents. The various delivery modes include web-based, cloud-based, and enterprise-based, which have various development phases such as phase I, phase II, phase III, and phase IV. It is employed in several sectors, such as pharmaceutical and biopharmaceutical companies, contract research organizations, consulting service companies, medical device manufacturers, hospitals, and academic research institutions.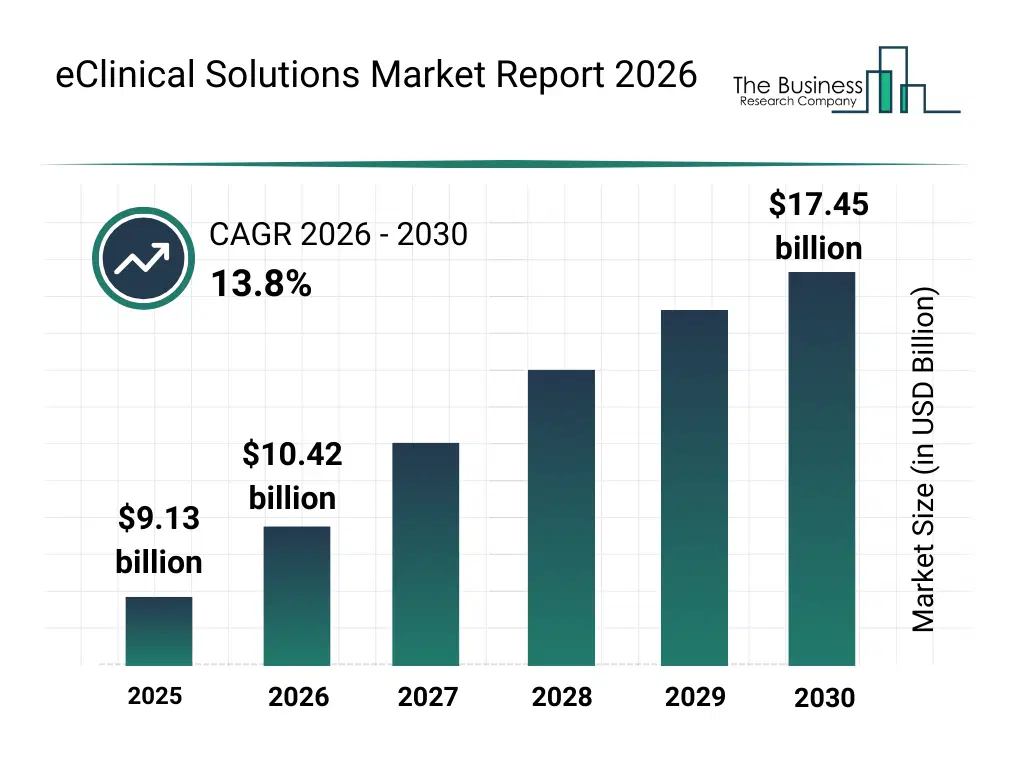
What Is The eClinical Solutions Market Size and Share 2026?
The eclinical solutions market size has grown rapidly in recent years. It will grow from $9.13 billion in 2025 to $10.42 billion in 2026 at a compound annual growth rate (CAGR) of 14.1%. The growth in the historic period can be attributed to clinical trial complexity, data management challenges, regulatory documentation needs, trial cost escalation, globalization of trials.What Is The eClinical Solutions Market Growth Forecast?
The eclinical solutions market size is expected to see rapid growth in the next few years. It will grow to $17.45 billion in 2030 at a compound annual growth rate (CAGR) of 13.8%. The growth in the forecast period can be attributed to virtual trial expansion, ai-based trial optimization, remote patient monitoring, faster regulatory submissions, interoperability standards adoption. Major trends in the forecast period include increasing adoption of cloud-based clinical platforms, growing use of real-time clinical data analytics, expansion of decentralized clinical trials, rising integration of edc and ctms systems, enhanced focus on regulatory compliance automation.Global eClinical Solutions Market Segmentation
1) By Product: Electronic Data Capture (EDC) And Clinical Data Management Systems (CDMS), Clinical Trial Management Systems (CTMS), Clinical Analytics Platforms, Randomization And Trial Supply Management (RTSM), Clinical Data Integration Platforms, Electronic Clinical Outcome Assessment (eCOA), Safety Solutions, Electronic Trial Master File (eTMF) 2) By Development Phase: Phase I, Phase II, Phase III, Phase IV 3) By Delivery Mode: Web-Based and Cloud-Based, Enterprise-Based 4) By End User: Pharmaceutical and Biopharmaceutical Companies, Contract Research Organizations, Consulting Service Companies, Medical Device Manufacturers, Hospitals, Academic Research Institutions Subsegments: 1) By Electronic Data Capture (EDC) And Clinical Data Management Systems (CDMS): Cloud-based EDC Systems, On-premises EDC Systems, Data Management Software 2) By Clinical Trial Management Systems (CTMS): Study Management Systems, Site Management Systems, Budget and Billing Management Systems 3) By Clinical Analytics Platforms: Predictive Analytics Tools, Real-time Data Analytics Solutions, Reporting and Visualization Tools 4) By Randomization And Trial Supply Management (RTSM): Randomization Software, Inventory Management Systems, Supply Chain Management Solutions 5) By Clinical Data Integration Platforms: Data Aggregation Tools, Interoperability Solutions, Data Transformation Software 6) By Electronic Clinical Outcome Assessment (eCOA): Patient-Reported Outcome Measures (PROMs), Clinician-Reported Outcome Measures (CROMs), Observer-Reported Outcome Measures (ObsROs) 7) By Safety Solutions: Pharmacovigilance Software, Adverse Event Reporting Systems, Risk Management Solutions 8) By Electronic Trial Master File (eTMF): Document Management Systems, Compliance Tracking Tools, Workflow Automation SolutionsWhat Is The Driver Of The eClinical Solutions Market?
The increasing number of clinical trials is expected to propel the growth of the eClinical solutions market going forward. Clinical trials are research studies conducted on human participants to assess the safety, efficacy, and therapeutic value of new drugs, medical interventions, or treatments. The number of clinical trials is rising due to advancements in personalized medicine, expanding therapeutic pipelines, increased R&D investments, and growing global participation in human subject research. The eClinical solutions market supports this trend by offering advanced computerized platforms that streamline trial operations, enable digital data capture, enhance biometrics services, and optimize trial management processes. For instance, in December 2024, according to the Association of the British Pharmaceutical Industry (ABPI), a UK-based organization, highlighted that the number of pharmaceutical industry trials launched in the UK increased for the second consecutive year, rising from 411 in 2022 to 426 in 2023. Therefore, the increasing number of clinical trials is driving the growth of the eClinical solutions industry.Key Players In The Global eClinical Solutions Market
Major companies operating in the eclinical solutions market are Anju Software Inc., BioClinicia Inc., Parexel International Corporation, CRF Health, Datatrak International Inc., Eclinical Solutions LLC, ERT Clinical, Medidata Solutions Inc., Veeva Systems Inc., Mednet Solutions, Oracle Corporation, Saama Technologies Inc., Signant Health, 4G Clinical, Research Manager, StudyKIK Inc., Deep 6 AI Inc., Clinical Research Management Inc., Curebase Inc., Bio-Optronics Inc., Complion Inc.Global eClinical Solutions Market Trends and Insights
Major companies operating in the eClinical solutions market are focusing on developing innovative technologies such as comprehensive clinical trial management solutions to streamline research workflows, strengthen data integrity, and improve regulatory compliance across clinical development programs. Comprehensive clinical trial management solutions refer to integrated software platforms that support the end-to-end planning, execution, and oversight of clinical trials through modules such as electronic data capture, randomization management, and electronic trial master file systems. For instance, in May 2024, EDETEK Inc., a US-based clinical solutions company, launched CONFORM eClinical Version 5.1, an advanced unified platform designed to optimize clinical trial processes by enhancing operational efficiency, improving data quality, and supporting regulatory adherence. By providing a self-contained system aligned with industry standards, the solution enables sponsors to maintain greater control over study conduct, achieve substantial cost savings, and accelerate trial timelines.What Are Latest Mergers And Acquisitions In The eClinical Solutions Market?
In September 2024, GI Partners, a US-based private investment firm, acquired a majority stake in eClinical Solutions LLC for an undisclosed amount. With this acquisition GI Partners aims to accelerate eClinical Solutions’ growth, enhance its AI‑powered clinical trial platforms, and expand its global reach. eClinical Solutions LLC is a US-based company that provides eClinical solutions, including cloud-based software and data services for clinical trials.Regional Outlook
North America was the largest region in the eClinical solutions market in 2025. The regions covered in this market report are Asia-Pacific, South East Asia, Western Europe, Eastern Europe, North America, South America, Middle East, Africa. The countries covered in this market report are Australia, Brazil, China, France, Germany, India, Indonesia, Japan, Taiwan, Russia, South Korea, UK, USA, Canada, Italy, Spain.What Defines the eClinical Solutions Market?
The eClinical solutions market includes revenues earned by entities by eConsent, eTMF, eISF, and remote monitoring. The market value includes the value of related goods sold by the service provider or included within the service offering. Only goods and services traded between entities or sold to end consumers are included.How is Market Value Defined and Measured?
The market value is defined as the revenues that enterprises gain from the sale of goods and/or services within the specified market and geography through sales, grants, or donations in terms of the currency (in USD unless otherwise specified). The revenues for a specified geography are consumption values that are revenues generated by organizations in the specified geography within the market, irrespective of where they are produced. It does not include revenues from resales along the supply chain, either further along the supply chain or as part of other products.What Key Data and Analysis Are Included in the eClinical Solutions Market Report 2026?
The eclinical solutions market research report is one of a series of new reports from The Business Research Company that provides market statistics, including industry global market size, regional shares, competitors with the market share, detailed market segments, market trends and opportunities, and any further data you may need to thrive in the eclinical solutions industry. The market research report delivers a complete perspective of everything you need, with an in-depth analysis of the current and future state of the industry.eClinical Solutions Market Report Forecast Analysis
| Report Attribute | Details |
|---|---|
| Market Size Value In 2026 | $10.42 billion |
| Revenue Forecast In 2035 | $17.45 billion |
| Growth Rate | CAGR of 14.1% from 2026 to 2035 |
| Base Year For Estimation | 2025 |
| Actual Estimates/Historical Data | 2020-2025 |
| Forecast Period | 2026 - 2030 - 2035 |
| Market Representation | Revenue in USD Billion and CAGR from 2026 to 2035 |
| Segments Covered | Product, Development Phase, Delivery Mode, End User |
| Regional Scope | Asia-Pacific, Western Europe, Eastern Europe, North America, South America, Middle East, Africa |
| Country Scope | The countries covered in the report are Australia, Brazil, China, France, Germany, India, ... |
| Key Companies Profiled | Anju Software Inc., BioClinicia Inc., Parexel International Corporation, CRF Health, Datatrak International Inc., Eclinical Solutions LLC, ERT Clinical, Medidata Solutions Inc., Veeva Systems Inc., Mednet Solutions, Oracle Corporation, Saama Technologies Inc., Signant Health, 4G Clinical, Research Manager, StudyKIK Inc., Deep 6 AI Inc., Clinical Research Management Inc., Curebase Inc., Bio-Optronics Inc., Complion Inc. |
| Customization Scope | Request for Customization |
| Pricing And Purchase Options | Explore Purchase Options |
