
Electronic Clinical Outcome Assessment Solutions Market Report 2026
Global Outlook – By Product (Web Based eCOA Solutions, On Premise Based eCOA Solutions, Cloud Platform Based eCOA Solutions), By Approach (Patient-Reported Outcome (PRO), Clinician-Reported Outcome (ClinRO), Observer-Reported Outcome (ObsRO), Performance Outcome (PerfO) ), By End-User (Hospitals Or Healthcare Providers, Contract Research Organizations (CROs), Pharmaceutical And Biotechnology Firms, Medical Device Companies, Other End-Users) - Market Size, Trends, And Global Forecast 2026-2035
Electronic Clinical Outcome Assessment Solutions Market Overview
• Electronic Clinical Outcome Assessment Solutions market size has reached to $1.94 billion in 2025 • Expected to grow to $3.62 billion in 2030 at a compound annual growth rate (CAGR) of 13.1% • Growth Driver: Rising Research And Development Activities Fueling The Growth Of The Electronic Clinical Outcome Assessment (eCOA) Solutions Market • Market Trend: Advancements In eCOA Toolkit Technology Enhance Patient Usability And Streamline Clinical Data Collection • North America was the largest region in 2025 and Asia-Pacific is the fastest growing region.What Is Covered Under Electronic Clinical Outcome Assessment Solutions Market?
Electronic clinical outcome assessment solutions refer to digital platforms designed to collect, manage, and analyze patient-reported and clinician-reported outcomes during clinical trials. They are used to streamline data capture, improve data accuracy, and enhance real-time monitoring of patient health outcomes. The main types of products in the electronic clinical outcome assessment solutions are web-based eCOA solutions, on-premise-based eCOA solutions, and cloud-platform-based eCOA solutions. Web-based eCOA solutions refer to a programme that is distributed over a network (typically a corporate intranet or the internet) and accessible via a common web browser. The approaches include patient-reported outcome (PRO), clinician-reported outcome (ClinRO), observer-reported outcome (ObsRO), and performance outcome (PerfO) and are used in hospitals or healthcare providers, contract research organizations (CROS), pharmaceutical and biotechnology firms, medical device companies, and other end users.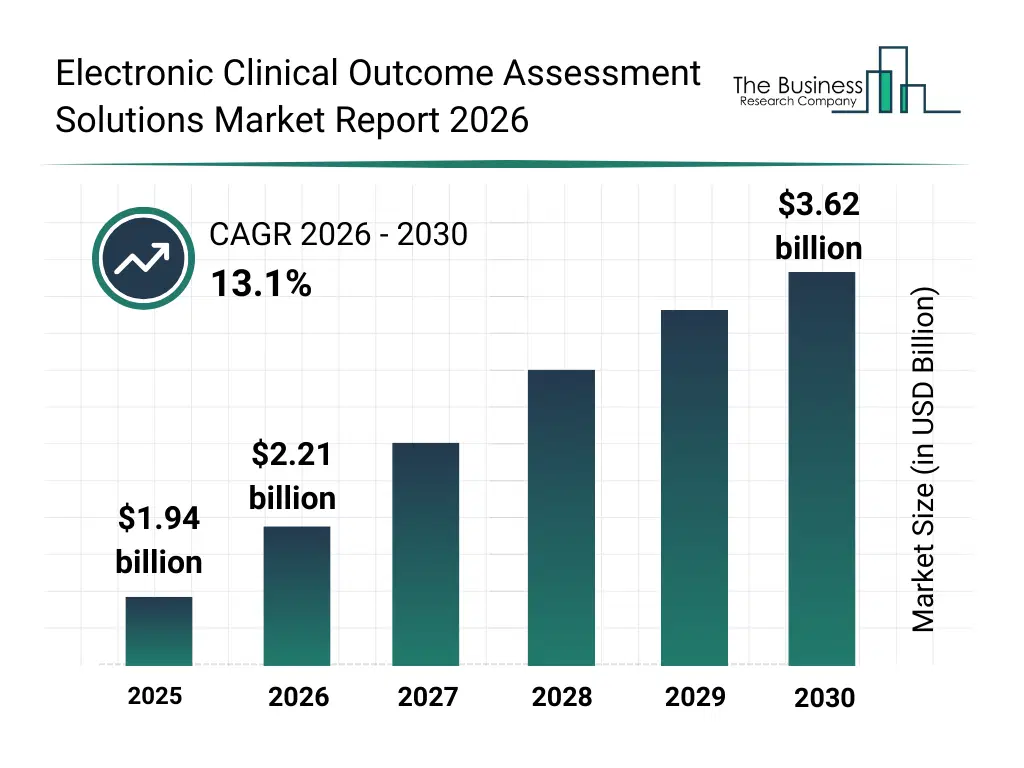
What Is The Electronic Clinical Outcome Assessment Solutions Market Size and Share 2026?
The electronic clinical outcome assessment solutions market size has grown rapidly in recent years. It will grow from $1.94 billion in 2025 to $2.21 billion in 2026 at a compound annual growth rate (CAGR) of 14.1%. The growth in the historic period can be attributed to growth in clinical trials, need for accurate data capture, regulatory compliance requirements, adoption of electronic data systems, inefficiencies in paper-based assessments.What Is The Electronic Clinical Outcome Assessment Solutions Market Growth Forecast?
The electronic clinical outcome assessment solutions market size is expected to see rapid growth in the next few years. It will grow to $3.62 billion in 2030 at a compound annual growth rate (CAGR) of 13.1%. The growth in the forecast period can be attributed to expansion of decentralized trials, AI-driven data analytics, demand for real-time monitoring, growth in SaaS healthcare platforms, patient-centric trial designs. Major trends in the forecast period include digital patient-reported outcome collection, real-time clinical trial data capture, remote patient engagement tools, regulatory-compliant data management, mobile-based outcome assessment.Global Electronic Clinical Outcome Assessment Solutions Market Segmentation
1) By Product: Web Based eCOA Solutions, On Premise Based eCOA Solutions, Cloud Platform Based eCOA Solutions 2) By Approach: Patient-Reported Outcome (PRO), Clinician-Reported Outcome (ClinRO), Observer-Reported Outcome (ObsRO), Performance Outcome (PerfO) 3) By End-User: Hospitals Or Healthcare Providers, Contract Research Organizations (CROs), Pharmaceutical And Biotechnology Firms, Medical Device Companies, Other End-Users Subsegments: 1) By Web-Based eCOA Solutions: Patient-Reported Outcome (PRO) Tools, Clinician-Reported Outcome (ClinRO) Tools, Observer-Reported Outcome (ObsRO) Tools 2) By On-Premise Based eCOA Solutions: Standalone Software Solutions, Integrated On-Premise Solutions 3) By Cloud Platform Based eCOA Solutions: Software As A Service (SaaS) eCOA Solutions, Mobile eCOA Applications, Hybrid Cloud SolutionsWhat Is The Driver Of The Electronic Clinical Outcome Assessment Solutions Market?
The increase in research and development (R&D) activities is expected to propel the growth of the electronic clinical outcome assessment (eCOA) solutions market going forward. Research and development activities involve systematic efforts by organizations to innovate, develop new technologies, and advance therapeutic solutions. The rise in R&D activity is driven by expanding scientific initiatives and the growing need for high-quality clinical evidence, resulting in a greater number of clinical studies requiring precise and efficient outcome measurement. Electronic clinical outcome assessment solutions support these efforts by enabling real-time, digital collection of patient-reported outcomes, improving data accuracy, compliance, and overall trial efficiency. For instance, in November 2023, according to the report published by the European Federation of Pharmaceutical Industries and Associations, a Belgium-based trade association, pharmaceutical industry research and development expenditure rose to 50,000 million in 2023, up from 47,010 million in 2022. Therefore, the rising investment in research and development is driving the growth of the electronic clinical outcome assessment solutions industry.Key Players In The Global Electronic Clinical Outcome Assessment Solutions Market
Major companies operating in the electronic clinical outcome assessment solutions market are IQVIA Holdings Inc., ERT Clinical, OmniComm Systems Inc., Parexel International Corporation, Veeva Systems Inc., Medidata Solutions Inc., Signant Health, Clario Inc., ArisGlobal LLC, YPrime LLC, eClinical Solutions LLC, CRF Bracket Company, Anju Software Inc., Castor EDC B.V., Kayentis, Mednet Solutions LLC, Clinical Ink Inc., Cloudbyz Inc., Climedo Health GmbH, ClinCapture Inc.Global Electronic Clinical Outcome Assessment Solutions Market Trends and Insights
Major companies in the electronic clinical outcome assessment solutions market are developing innovative platforms, such as comprehensive systems to streamline data collection, enhance patient engagement, and improve trial efficiency. A comprehensive system is a complete and all-inclusive setup that integrates multiple functions or components to achieve a goal efficiently. For instance, in May 2024, EDETEK, a US-based technology company that specializes in providing digital solutions for the clinical trial sector, announced the launch of CONFORM eClinical Version 5.1, a comprehensive platform designed to enhance and streamline clinical trial management processes. This new version integrates a wide range of components that facilitate efficient clinical research, addressing the evolving needs of sponsors and stakeholders in the pharmaceutical industry.What Are Latest Mergers And Acquisitions In The Electronic Clinical Outcome Assessment Solutions Market?
In July 2023, Signant Health Private Limited, a US-based healthcare technology company, acquired DSG for an undisclosed amount. With this acquisition, Signant aims to enhance its eClinical solution suite to support both traditional and decentralized clinical trials, expanding its technological capabilities and service offerings. DSG Inc is a US-based healthcare technology company that specializes in providing digital solutions and services designed to optimize clinical trial management and patient engagement.Regional Outlook
North America was the largest region in the electronic clinical outcome assessment solutions market in 2025. Asia-Pacific is expected to be the fastest-growing region in the electronic clinical outcome assessment solutions market during the forecast period. The regions covered in this market report are Asia-Pacific, South East Asia, Western Europe, Eastern Europe, North America, South America, Middle East, Africa. The countries covered in this market report are Australia, Brazil, China, France, Germany, India, Indonesia, Japan, Taiwan, Russia, South Korea, UK, USA, Canada, Italy, Spain.What Defines the Electronic Clinical Outcome Assessment Solutions Market?
The electronic clinical outcome assessment solutions market consists of sales of eCOA live, eCOA multimedia, eCOA rescue studies, and suicidal ideation. Values in this market are ‘factory gate’ values, that is the value of goods sold by the manufacturers or creators of the goods, whether to other entities (including downstream manufacturers, wholesalers, distributors and retailers) or directly to end customers. The value of goods in this market includes related services sold by the creators of the goods.How is Market Value Defined and Measured?
The market value is defined as the revenues that enterprises gain from the sale of goods and/or services within the specified market and geography through sales, grants, or donations in terms of the currency (in USD unless otherwise specified). The revenues for a specified geography are consumption values that are revenues generated by organizations in the specified geography within the market, irrespective of where they are produced. It does not include revenues from resales along the supply chain, either further along the supply chain or as part of other products.What Key Data and Analysis Are Included in the Electronic Clinical Outcome Assessment Solutions Market Report 2026?
The electronic clinical outcome assessment solutions market research report is one of a series of new reports from The Business Research Company that provides market statistics, including industry global market size, regional shares, competitors with the market share, detailed market segments, market trends and opportunities, and any further data you may need to thrive in the electronic clinical outcome assessment solutions industry. The market research report delivers a complete perspective of everything you need, with an in-depth analysis of the current and future state of the industry.Electronic Clinical Outcome Assessment Solutions Market Report Forecast Analysis
| Report Attribute | Details |
|---|---|
| Market Size Value In 2026 | $2.21 billion |
| Revenue Forecast In 2035 | $3.62 billion |
| Growth Rate | CAGR of 14.1% from 2026 to 2035 |
| Base Year For Estimation | 2025 |
| Actual Estimates/Historical Data | 2020-2025 |
| Forecast Period | 2026 - 2030 - 2035 |
| Market Representation | Revenue in USD Billion and CAGR from 2026 to 2035 |
| Segments Covered | Product, Approach, End-User |
| Regional Scope | Asia-Pacific, Western Europe, Eastern Europe, North America, South America, Middle East, Africa |
| Country Scope | The countries covered in the report are Australia, Brazil, China, France, Germany, India, ... |
| Key Companies Profiled | IQVIA Holdings Inc., ERT Clinical, OmniComm Systems Inc., Parexel International Corporation, Veeva Systems Inc., Medidata Solutions Inc., Signant Health, Clario Inc., ArisGlobal LLC, YPrime LLC, eClinical Solutions LLC, CRF Bracket Company, Anju Software Inc., Castor EDC B.V., Kayentis, Mednet Solutions LLC, Clinical Ink Inc., Cloudbyz Inc., Climedo Health GmbH, ClinCapture Inc. |
| Customization Scope | Request for Customization |
| Pricing And Purchase Options | Explore Purchase Options |
