
Electronic Trial Master File (eTMF) Systems Market Report 2026
Global Outlook – By Component (Services, Software), By Delivery Mode (On-Premise, Cloud-Based), By End-User (Pharmaceutical And Biotechnology Companies, Contract Research Organizations (CROs), Other End-Users) - Market Size, Trends, And Global Forecast 2026-2035
Electronic Trial Master File (eTMF) Systems Market Overview
• Electronic Trial Master File (eTMF) Systems market size has reached to $1.55 billion in 2025 • Expected to grow to $2.67 billion in 2030 at a compound annual growth rate (CAGR) of 11.4% • Growth Driver: The Increase In Clinical Trial Activity Fueling Market Growth Due To Rising Research Demand And Regulatory Requirements • Market Trend: Cloud-Based eTMF Innovations Enhancing Clinical Trial Documentation Accuracy And Operational Efficiency • North America was the largest region in 2025 and Asia-Pacific is the fastest growing region.What Is Covered Under Electronic Trial Master File (eTMF) Systems Market?
An electronic trial master file (eTMF) is a trial master file in electronic or digital format, serving as a content management system for the pharmaceutical industry. The eTMF system manages, stores, tracks, and archives essential clinical study documents electronically. It is designed to ensure compliance with regulatory requirements, such as the FDA's Title 21 CFR Part 11, and to provide a secure, centralized, and easily accessible repository for clinical trial documents. The main components of electronic trial master file (eTMF) systems are services and services. The electronic trial master file (eTMF) system services refer to functionalities or features provided by the eTMF system to support the management of clinical trial documentation in electronic format. It is delivered on-premise and cloud-based and used in pharmaceutical and biotechnology companies, contract research organizations, and other users.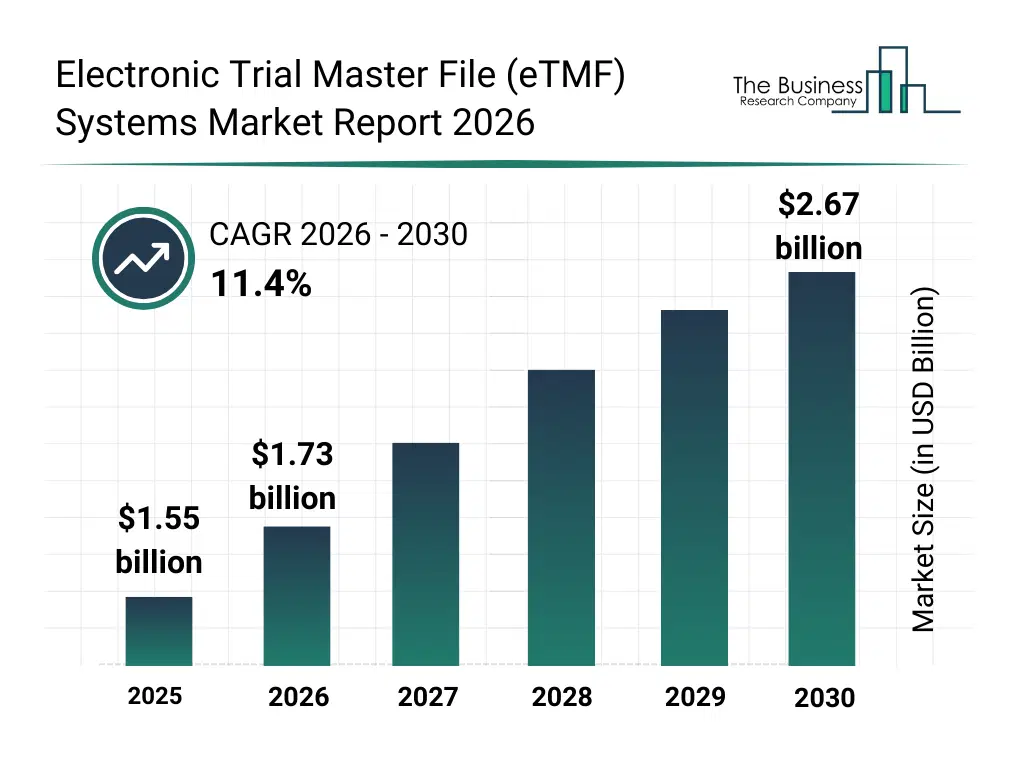
What Is The Electronic Trial Master File (eTMF) Systems Market Size and Share 2026?
The electronic trial master file (etmf) systems market size has grown rapidly in recent years. It will grow from $1.55 billion in 2025 to $1.73 billion in 2026 at a compound annual growth rate (CAGR) of 11.8%. The growth in the historic period can be attributed to regulatory documentation requirements, clinical trial complexity, paper-based tmf limitations, audit readiness needs, global trial expansion.What Is The Electronic Trial Master File (eTMF) Systems Market Growth Forecast?
The electronic trial master file (etmf) systems market size is expected to see rapid growth in the next few years. It will grow to $2.67 billion in 2030 at a compound annual growth rate (CAGR) of 11.4%. The growth in the forecast period can be attributed to decentralized trial growth, ai-driven document classification, cloud adoption in clinical research, regulatory scrutiny increase, data security focus. Major trends in the forecast period include centralized clinical document management, cloud-based etmf platforms, regulatory compliance automation, real-time trial documentation tracking, secure data archiving systems.Global Electronic Trial Master File (eTMF) Systems Market Segmentation
1) By Component: Services, Software 2) By Delivery Mode: On-Premise, Cloud-Based 3) By End-User: Pharmaceutical And Biotechnology Companies, Contract Research Organizations (CROs), Other End-Users Subsegments: 1) By Services: Consulting Services, Implementation Services, Training And Support Services, Data Migration Services 2) By Software: eTMF Management Software, Document Management Software, Cloud-Based eTMF Solutions, Compliance And Regulatory SoftwareWhat Is The Driver Of The Electronic Trial Master File (eTMF) Systems Market?
The increasing number of clinical trials is expected to propel the growth of the electronic trial master file (eTMF) systems market going forward. Clinical trials are structured research studies conducted to evaluate the safety, efficacy, and overall impact of medical interventions such as drugs, therapies, and medical devices in targeted patient populations. The rise in clinical trial activity is driven by growing global healthcare R&D investments, expanding therapeutic innovation, and heightened regulatory requirements that demand precise, compliant, and accessible study documentation. eTMF systems support the clinical research ecosystem by digitizing trial documentation, facilitating real-time collaboration, ensuring audit readiness, and improving operational efficiency across trial sites. For instance, in December 2024, according to the Association of the British Pharmaceutical Industry (ABPI), a UK-based trade association, the number of industry-sponsored clinical trials initiated in the UK increased from 411 in 2022 to 426 in 2023. Therefore, the growing number of clinical trials is driving the expansion of the electronic trial master file (eTMF) systems industry.Key Players In The Global Electronic Trial Master File (eTMF) Systems Market
Major companies operating in the electronic trial master file (etmf) systems market are Veeva Systems, Oracle, TransPerfect, Phlexglobal, SureClinical Inc., MasterControl Inc, Clinevo Technologies, Covance Inc, Ennov, ePharma Solutions, Aris Global LLC, Montrium Inc, IQVIA, Labcorp Drug Development, Freyr, Medidata (Dassault Systèmes), BSI Life Sciences, OmniComm Systems Inc, Signant Health Ltd, Cloudbyz, Florence Healthcare, Egnyte Inc, Trial InteractiveGlobal Electronic Trial Master File (eTMF) Systems Market Trends and Insights
Major companies operating in the electronic trial master file (eTMF) systems market are focusing on developing advanced cloud-based platforms to improve document accuracy, enhance audit readiness, and streamline clinical trial operations. Cloud-based eTMF systems refer to digital, cloud-hosted platforms designed to manage essential clinical trial documentation securely and efficiently while supporting real-time collaboration across global study teams. For instance, in March 2023, Phlexglobal Ltd., a unit of PharmaLex, a Germany-based regulatory and compliance services provider, launched PhlexTMF v21, a next-generation, cloud-based eTMF solution enhanced with advanced artificial intelligence (AI). The platform reduces metadata errors and prevents misfiled documents at the point of upload, improving TMF quality, inspection readiness, and overall trial efficiency. Therefore, innovations in cloud-based eTMF solutions are transforming clinical documentation management and driving adoption across the life sciences sector.What Are Latest Mergers And Acquisitions In The Electronic Trial Master File (eTMF) Systems Market?
In September 2023, Florence Healthcare Inc., a US-based company that provides software solutions for clinical trial management, acquired VersaTrial for an undisclosed amount. The acquisition aimed to enhance site enablement for clinical trials by integrating complementary strengths. This strategic move seeks to maximize the efficiency and capacity of research sites, reduce the tech burden, expedite feasibility responses, and improve communication among study personnel. VersaTrial is a US-based clinical trial solutions provider.Regional Outlook
North America was the largest region in the electronic trial master file (eTMF) systems market in 2025. Asia-Pacific is expected to be the fastest-growing region in the forecast period. The regions covered in this market report are Asia-Pacific, South East Asia, Western Europe, Eastern Europe, North America, South America, Middle East, Africa. The countries covered in this market report are Australia, Brazil, China, France, Germany, India, Indonesia, Japan, Taiwan, Russia, South Korea, UK, USA, Canada, Italy, Spain.What Defines the Electronic Trial Master File (eTMF) Systems Market?
The electronic trial master file (eTMF) Systems market includes revenues earned by entities through implementation and integration services, document migration services, quality control and compliance services and regulatory submission support. The market value includes the value of related goods sold by the service provider or included within the service offering. Only goods and services traded between entities or sold to end consumers are included. The electronic trial master file (eTMF) Systems market consists of sales of cloud-based eTMF solutions, eTMF software solutions, mobile applications, collaboration and communication tools. Values in this market are ‘factory gate’ values, that is the value of goods sold by the manufacturers or creators of the goods, whether to other entities (including downstream manufacturers, wholesalers, distributors and retailers) or directly to end customers. The value of goods in this market includes related services sold by the creators of the goods.How is Market Value Defined and Measured?
The market value is defined as the revenues that enterprises gain from the sale of goods and/or services within the specified market and geography through sales, grants, or donations in terms of the currency (in USD unless otherwise specified). The revenues for a specified geography are consumption values that are revenues generated by organizations in the specified geography within the market, irrespective of where they are produced. It does not include revenues from resales along the supply chain, either further along the supply chain or as part of other products.What Key Data and Analysis Are Included in the Electronic Trial Master File (eTMF) Systems Market Report 2026?
The electronic trial master file (etmf) systems market research report is one of a series of new reports from The Business Research Company that provides market statistics, including industry global market size, regional shares, competitors with the market share, detailed market segments, market trends and opportunities, and any further data you may need to thrive in the electronic trial master file (etmf) systems industry. The market research report delivers a complete perspective of everything you need, with an in-depth analysis of the current and future state of the industry.Electronic Trial Master File (eTMF) Systems Market Report Forecast Analysis
| Report Attribute | Details |
|---|---|
| Market Size Value In 2026 | $1.73 billion |
| Revenue Forecast In 2035 | $2.67 billion |
| Growth Rate | CAGR of 11.8% from 2026 to 2035 |
| Base Year For Estimation | 2025 |
| Actual Estimates/Historical Data | 2020-2025 |
| Forecast Period | 2026 - 2030 - 2035 |
| Market Representation | Revenue in USD Billion and CAGR from 2026 to 2035 |
| Segments Covered | Component, Delivery Mode, End-User |
| Regional Scope | Asia-Pacific, Western Europe, Eastern Europe, North America, South America, Middle East, Africa |
| Country Scope | The countries covered in the report are Australia, Brazil, China, France, Germany, India, ... |
| Key Companies Profiled | Veeva Systems, Oracle, TransPerfect, Phlexglobal, SureClinical Inc., MasterControl Inc, Clinevo Technologies, Covance Inc, Ennov, ePharma Solutions, Aris Global LLC, Montrium Inc, IQVIA, Labcorp Drug Development, Freyr, Medidata (Dassault Systèmes), BSI Life Sciences, OmniComm Systems Inc, Signant Health Ltd, Cloudbyz, Florence Healthcare, Egnyte Inc, Trial Interactive |
| Customization Scope | Request for Customization |
| Pricing And Purchase Options | Explore Purchase Options |
