
Embolization Product Market Report 2026
Global Outlook – By Product Type (Embolization Coils, Embolic Agents, Detachable Balloons, Liquid Embolics, Microcatheters, Guidewires), By Material (Metallic, Polymeric, Biological, Hybrid), By Technology (Bioresorbable Technology, Non-Bioresorbable Technology), By Application (Oncology, Neurology, Peripheral Vascular Disease, Urology, Other Applications), By End User (Hospitals, Ambulatory Surgical Centers, Specialty Clinics) – Market Size, Trends, Strategies, and Forecast to 2035
Embolization Product Market Overview
• Embolization Product market size has reached to $2.08 billion in 2025 • Expected to grow to $3.14 billion in 2030 at a compound annual growth rate (CAGR) of 8.6% • Growth Driver: Increasing Demand For Minimally Invasive Treatment Procedures Fueling The Growth Of The Market Due To Rising Adoption Of Less Invasive Medical Interventions • Market Trend: Increasing Adoption Of Next-Generation Microcatheters In Minimally Invasive Procedures • North America was the largest region in 2025 and Asia-Pacific is the fastest growing region.What Is Covered Under Embolization Product Market?
Embolization products are medical devices used to intentionally block or reduce blood flow in blood vessels. They help treat conditions such as aneurysms, arteriovenous malformations, and tumors by preventing blood supply to targeted areas. These products are designed to be biocompatible, safe, and effective for precise vascular interventions. The main product types of embolization product include Embolization Coils, embolic agents, detachable balloons, liquid embolics, microcatheters, and guidewires. Embolization coils are small, flexible devices used to occlude blood vessels, helping to control bleeding or block blood flow to targeted areas such as aneurysms or tumors. Different materials are metallic, polymeric, biological, and hybrid. Multiple technologies consist of bioresorbable and non-bioresorbable options. Various applications cover oncology, neurology, peripheral vascular disease, urology, and others, and they are used by several end-users include hospitals, ambulatory surgical centers, and specialty clinics.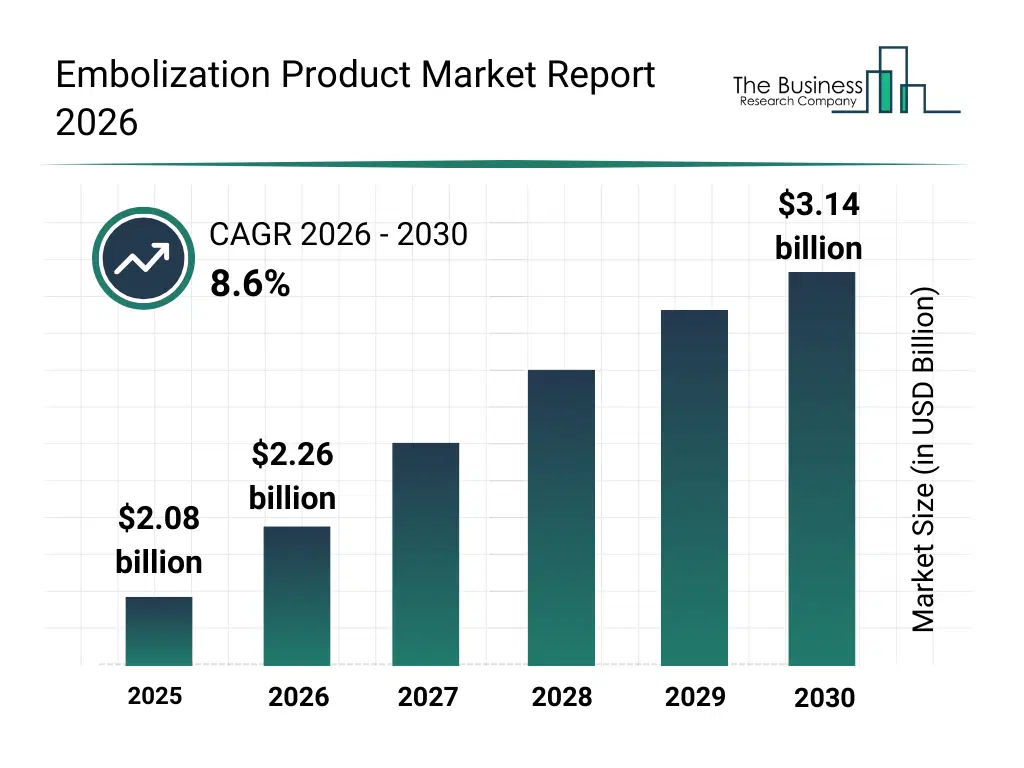
What Is The Embolization Product Market Size and Share 2026?
The embolization product market size has grown strongly in recent years. It will grow from $2.08 billion in 2025 to $2.26 billion in 2026 at a compound annual growth rate (CAGR) of 8.8%. The growth in the historic period can be attributed to increasing prevalence of cardiovascular diseases, rising incidence of aneurysms and vascular malformations, growing adoption of minimally invasive procedures, increasing awareness of interventional radiology, rising demand for targeted cancer treatments, growing geriatric population requiring vascular interventions, increasing preference for outpatient procedures, rising healthcare infrastructure in emerging economies, growing focus on reducing surgical risks and complications, increasing technological advancements in embolization devices.What Is The Embolization Product Market Growth Forecast?
The embolization product market size is expected to see strong growth in the next few years. It will grow to $3.14 billion in 2030 at a compound annual growth rate (CAGR) of 8.6%. The growth in the forecast period can be attributed to advancement in bioresorbable embolic materials, innovation in microcatheter-compatible devices, integration of image-guided navigation systems, advancement in liquid embolic agents, innovation in coil and plug designs, integration with robotic-assisted interventions, advancement in patient-specific device customization, innovation in minimally invasive delivery systems, integration of smart monitoring for real-time feedback, advancement in cost-effective manufacturing processes. Major trends in the forecast period include minimally invasive treatment adoption, personalized vascular therapies, advanced imaging integration, biocompatible material innovation, cost-effective embolization solutions.Global Embolization Product Market Segmentation
1) By Product Type: Embolization Coils, Embolic Agents, Detachable Balloons, Liquid Embolics, Microcatheters, Guidewires 2) By Material: Metallic, Polymeric, Biological, Hybrid 3) By Technology: Bioresorbable Technology, Non-Bioresorbable Technology 4) By Application: Oncology, Neurology, Peripheral Vascular Disease, Urology, Other Applications 5) By End User: Hospitals, Ambulatory Surgical Centers, Specialty Clinics 1) By Embolization Coils: Detachable Coils, Pushable Coils, Helical Coils, Complex-Shaped Coils, Hydrogel-Coated Coils, Bare Platinum Coils, Fibered Coils, Microcoils (For Neurovascular Use) 2) By Embolic Agents: Particulate Embolic Agents, Microspheres (Drug-Eluting And Non-Drug-Eluting), Polyvinyl Alcohol (PVA) Particles, Gelatin Sponge Particles, Radiopaque Embolic Agents, Biodegradable Embolic Agents, Metallic Embolic Particles 3) By Detachable Balloons: Flow-Occlusion Balloons, Aneurysm Treatment Balloons, Temporary Occlusion Balloons, Permanent Occlusion Balloons, Single-Lumen Detachable Balloons, Dual-Lumen Detachable Balloons 4) By Liquid Embolics: Ethylene-Vinyl Alcohol Copolymer (EVOH) Agents, Cyanoacrylate-Based Adhesives (Glue Embolics), Polymerizing Liquid Embolics, Precipitating Liquid Embolics, Radiopaque Liquid Embolics, Non-Adhesive Liquid Embolics 5) By Microcatheters: Single-Lumen Microcatheters, Dual-Lumen Microcatheters, Flow-Directed Microcatheters, Over-The-Wire Microcatheters, High-Flow Microcatheters, Steerable Microcatheters, Neurovascular Microcatheters 6) By Guidewires: Hydrophilic Guidewires, Hydrophobic Guidewires, Stainless Steel Guidewires, Nitinol Guidewires, Shapeable Or Steerable Guidewires, Micro-Guidewires, Specialty Neurovascular GuidewiresWhat Is The Driver Of The Embolization Product Market?
The increasing demand for minimally invasive treatment procedures is expected to propel the growth of the embolization product market going forward. Minimally invasive procedures refer to medical interventions that involve small incisions or catheter-based techniques, minimizing trauma and enabling faster recovery compared with traditional surgeries. The rising preference for such treatments is driving the use of embolization products, which are critical for procedures such as controlling hemorrhages, treating vascular malformations, and tumor management. Embolization products support minimally invasive treatment procedures by allowing physicians to precisely block abnormal blood flow to targeted areas, reducing surgical risk, shortening recovery time, and improving overall patient outcomes. For instance, in August 2025, according to Dr. M. MacDonald, the global number of minimally invasive cosmetic procedures reached nearly 25.4 million in 2023, reflecting a strong upward trend and widespread adoption of less invasive medical interventions. Therefore, increasing demand for minimally invasive treatment procedures is driving the growth of the embolization product industry.Key Players In The Global Embolization Product Market
Major companies operating in the embolization product market are Cardinal Health, Abbott Laboratories, Medtronic plc, Stryker Corporation, Boston Scientific Corporation, MicroVention Inc., Terumo Corporation, Kaneka Corporation, Cook Medical, Merit Medical Systems Inc., Penumbra Inc., Guerbet Group, B. Braun Melsungen AG, Sirtex Medical Limited, Medikit Co. Ltd., Balt USA LLC, Acandis GmbH & Co. KG, ABK Biomedical Inc., CeloNova BioSciences, Shape Memory Medical Inc.Global Embolization Product Market Trends and Insights
Major companies operating in the embolization products market are focusing on developing advanced microcatheter solutions, such as next-generation balloon occlusion devices, to improve procedural efficiency, enhance usability, and address clinical feedback from physicians. Next-generation balloon occlusion microcatheters refer to minimally invasive devices designed to block or reduce blood flow in targeted blood vessels during embolization procedures, offering enhanced control, safety, and ease of use. For instance, in July 2025, Embolx, a US-based medical device company, launched the Sniper G3 Balloon Occlusion Microcatheter. This third-generation device features an extended shelf life, improved ease of use, and streamlined preparation, directly responding to critical feedback from physician users. It enables clinicians to perform targeted embolization procedures more efficiently, reduces procedural complexity, and improves overall patient outcomes.What Are Latest Mergers And Acquisitions In The Embolization Product Market?
In February 2025, Stryker, a US-based global medical technology company, acquired Inari Medical Inc. for an undisclosed amount. Through this acquisition, Stryker aims to expand its interventional endovascular and embolization product portfolio by integrating Inari Medical’s peripheral vascular technologies, enhancing treatment options for VTE and other vascular conditions. Inari Medical Inc. is a US-based medical device company specializing in embolization products for minimally invasive vascular treatments.Regional Outlook
North America was the largest region in the embolization product market in 2025. Asia-Pacific is expected to be the fastest-growing region in the forecast period. The regions covered in this market report are Asia-Pacific, South East Asia, Western Europe, Eastern Europe, North America, South America, Middle East, Africa. The countries covered in this market report are Australia, Brazil, China, France, Germany, India, Indonesia, Japan, Taiwan, Russia, South Korea, UK, USA, Canada, Italy, Spain.What Defines the Embolization Product Market?
The embolization product market consists of sales of coils, particles, liquid embolic agents, vascular plugs, gelatin sponges, and bioresorbable embolic materials. Values in this market are ‘factory gate’ values, that is, the value of goods sold by the manufacturers or creators of the goods, whether to other entities (including downstream manufacturers, wholesalers, distributors, and retailers) or directly to end customers. The value of goods in this market includes related services sold by the creators of the goods.How is Market Value Defined and Measured?
The market value is defined as the revenues that enterprises gain from the sale of goods and/or services within the specified market and geography through sales, grants, or donations in terms of the currency (in USD unless otherwise specified). The revenues for a specified geography are consumption values that are revenues generated by organizations in the specified geography within the market, irrespective of where they are produced. It does not include revenues from resales along the supply chain, either further along the supply chain or as part of other products.What Key Data and Analysis Are Included in the Embolization Product Market Report 2026?
The embolization product market research report is one of a series of new reports from The Business Research Company that provides market statistics, including industry global market size, regional shares, competitors with the market share, detailed market segments, market trends and opportunities, and any further data you may need to thrive in the embolization product industry. The market research report delivers a complete perspective of everything you need, with an in-depth analysis of the current and future state of the industry.Embolization Product Market Report Forecast Analysis
| Report Attribute | Details |
|---|---|
| Market Size Value In 2026 | $2.26 billion |
| Revenue Forecast In 2035 | $3.14 billion |
| Growth Rate | CAGR of 8.8% from 2026 to 2035 |
| Base Year For Estimation | 2025 |
| Actual Estimates/Historical Data | 2020-2025 |
| Forecast Period | 2026 - 2030 - 2035 |
| Market Representation | Revenue in USD Billion and CAGR from 2026 to 2035 |
| Segments Covered | Product Type, Material, Technology, Application, End User, Embolization Coils, Embolic Agents, Detachable Balloons, Liquid Embolics, Microcatheters, Guidewires |
| Regional Scope | Asia-Pacific, Western Europe, Eastern Europe, North America, South America, Middle East, Africa |
| Country Scope | The countries covered in the report are Australia, Brazil, China, France, Germany, India, ... |
| Key Companies Profiled | Cardinal Health, Abbott Laboratories, Medtronic plc, Stryker Corporation, Boston Scientific Corporation, MicroVention Inc., Terumo Corporation, Kaneka Corporation, Cook Medical, Merit Medical Systems Inc., Penumbra Inc., Guerbet Group, B. Braun Melsungen AG, Sirtex Medical Limited, Medikit Co. Ltd., Balt USA LLC, Acandis GmbH & Co. KG, ABK Biomedical Inc., CeloNova BioSciences, Shape Memory Medical Inc. |
| Customization Scope | Request for Customization |
| Pricing And Purchase Options | Explore Purchase Options |
