
Endovascular Stent Grafts Market Report 2026
Global Outlook – By Product (Hemodialysis Access Grafts, Endovascular Stent Grafts, Coronary Artery By-Pass Grafts, Vascular Grafts For Aorta Disease, Peripheral Vascular Grafts), By Raw Material (Synthetic Vascular Grafts, Biological Vascular Grafts, Hybrid Vascular Grafts), By Application (Cardiac Aneurysm, Kidney Failure, Vascular Occlusion, Coronary Artery Disease) – Market Size, Trends, Strategies, and Forecast to 2035
Endovascular Stent Grafts Market Overview
• Endovascular Stent Grafts market size has reached to $1.45 billion in 2025 • Expected to grow to $7.13 billion in 2030 at a compound annual growth rate (CAGR) of 37.5% • Growth Driver: Impact Of Rising Cardiovascular Diseases On The Growth Of The Endovascular Stent Graft • Market Trend: Innovations In Endovascular Stent Grafts With Multi-Branch Thoracic Aortic Stent-Graft Achieves Milestone With First Clinical Implantation • North America was the largest region in 2025.What Is Covered Under Endovascular Stent Grafts Market?
Endovascular stent grafts are medical devices used to treat conditions involving blood vessels, such as aneurysms or stenosis. They consist of a metal stent (a small mesh tube) covered with a fabric graft. The stent graft is inserted into the affected blood vessel through a catheter, typically into a large artery in the groin or arm. Endovascular stent grafts often repair aneurysms (dilated areas of blood vessels) or other vessel damage, offering a less invasive alternative to traditional surgery. The main product types of endovascular stent grafts are hemodialysis access grafts, endovascular stent grafts, coronary artery bypass grafts, vascular grafts for aorta disease, and peripheral vascular grafts. Hemodialysis access grafts are specially designed vascular grafts that create an access point for hemodialysis in patients with kidney failure. The various raw materials such as synthetic vascular grafts, biological vascular grafts, and hybrid vascular grafts are used in various applications such as cardiac aneurysms, kidney failure, vascular occlusion, and coronary artery diseases.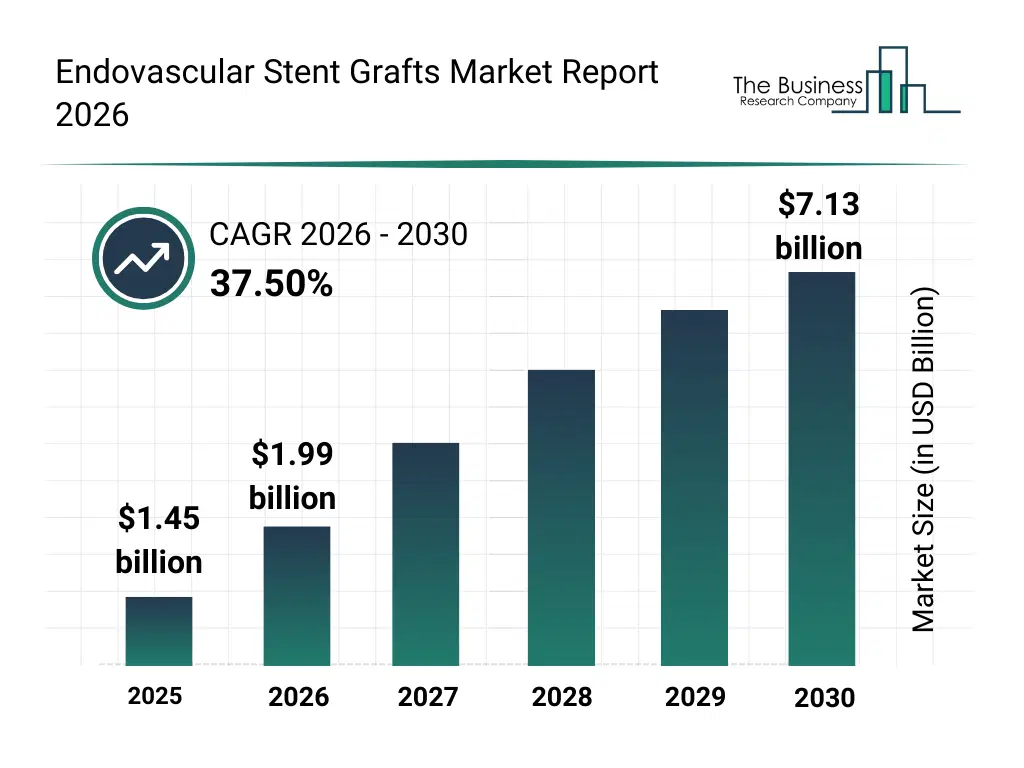
What Is The Endovascular Stent Grafts Market Size and Share 2026?
The endovascular stent grafts market size has grown exponentially in recent years. It will grow from $1.45 billion in 2025 to $1.99 billion in 2026 at a compound annual growth rate (CAGR) of 37.9%. The growth in the historic period can be attributed to reliance on conventional open surgical procedures, limited availability of advanced stent graft materials, growing prevalence of vascular diseases, early adoption of coronary and peripheral grafts, increasing awareness of aortic aneurysm treatments.What Is The Endovascular Stent Grafts Market Growth Forecast?
The endovascular stent grafts market size is expected to see exponential growth in the next few years. It will grow to $7.13 billion in 2030 at a compound annual growth rate (CAGR) of 37.5%. The growth in the forecast period can be attributed to technological innovations in endovascular stent grafts, rising minimally invasive surgeries, growth in aortic aneurysm and dissection cases, increasing adoption of hybrid vascular grafts, expansion of cardiovascular healthcare infrastructure. Major trends in the forecast period include increasing adoption of minimally invasive endovascular procedures, rising demand for customized stent grafts, advancements in hybrid and biologic graft materials, expansion of thoracic and abdominal aortic repair procedures, enhanced focus on patient safety and regulatory compliance.Global Endovascular Stent Grafts Market Segmentation
1) By Product: Hemodialysis Access Grafts, Endovascular Stent Grafts, Coronary Artery By-Pass Grafts, Vascular Grafts For Aorta Disease, Peripheral Vascular Grafts 2) By Raw Material: Synthetic Vascular Grafts, Biological Vascular Grafts, Hybrid Vascular Grafts 3) By Application: Cardiac Aneurysm, Kidney Failure, Vascular Occlusion, Coronary Artery Disease Subsegments: 1) By Hemodialysis Access Grafts: Arteriovenous (AV) Fistula Grafts, Synthetic Hemodialysis Access Grafts, Bioengineered Hemodialysis Access Grafts, Coated Hemodialysis Grafts, Endovascular Hemodialysis Grafts 2) By Endovascular Stent Grafts: Aneurysm Stent Grafts, Stent Grafts For Aortic Dissection, Thoracic Endovascular Stent Grafts, Abdominal Endovascular Stent Grafts, Biliary Endovascular Stent Grafts 3) By Coronary Artery By-Pass Grafts: Saphenous Vein Grafts (SVG), Arterial Coronary Bypass Grafts, Endovascular Coronary Artery Bypass Grafts (CABG), Synthetic Coronary Bypass Grafts 4) By Vascular Grafts For Aorta Disease: Endovascular Aortic Stent Grafts, Aortic Aneurysm Grafts, Aortic Bypass Grafts, Thoracoabdominal Aortic Stent Grafts, Aortic Dissection Stent Grafts 5) By Peripheral Vascular Grafts: Carotid Artery Stent Grafts, Femoral Artery Stent Grafts, Popliteal Artery Stent Grafts, Iliac Artery Stent Grafts, Peripheral Endovascular Stent GraftsWhat Is The Driver Of The Endovascular Stent Grafts Market?
The increasing prevalence of cardiovascular diseases is expected to propel the growth of the endovascular stent graft market going forward. Cardiovascular disease (CVD) refers to a class of diseases that involve the heart or blood vessels. The increasing prevalence of cardiovascular diseases is due to a combination of lifestyle choices, medical conditions, genetic predisposition, and environmental factors. Endovascular stent grafts offer a valuable option for treating cardiovascular diseases with a focus on minimizing invasiveness and improving patient outcomes. The stent graft reinforces the weakened vessel wall, preventing it from rupturing and reducing the risk of life-threatening complications. For instance, in October 2025, according to the Australian Institute Of Health And Welfare, an Australia-based government agency reported that in 2023, an estimated 57,100 adults aged 25 and above experienced acute coronary events, averaging roughly 156 incidents per day. Therefore, an increase in cardiovascular diseases drives the growth of endovascular stent grafts industry.Key Players In The Global Endovascular Stent Grafts Market
Major companies operating in the endovascular stent grafts market are Abbott Laboratories, Medtronic PLC, Boston Scientific Corporation, Terumo Corporation, Cook Group Incorporated, Merit Medical Systems Inc., Biotronik SE & Co KG, MicroPort Scientific Corporation, Endologix LLC, LeMaitre Vascular Inc., Lifetech Scientific Corporation, W. L. Gore & Associates Inc., Braile Biomedica, BrosMed Medical BV, Micro Interventional Devices Inc. (MID), Lombard Medical Inc., Endovastec Medical Technology Co Ltd., Jotec GmbH, Bentley InnoMed GmbH, Bolton Medical Inc., Alvimedica, Endospan Ltd., Vivasure Medical Ltd.Global Endovascular Stent Grafts Market Trends and Insights
Major companies operating in the endovascular stent grafts market are focused on developing innovative products such as thoracic aortic stent-graft to enhance the precision and efficacy of treating thoracic aortic aneurysms and dissections, improve patient outcomes, and address complex anatomical challenges with advanced technology and design improvements. A thoracic aortic stent graft is a specialized medical device used in endovascular surgery to treat diseases and conditions of the thoracic aorta; the section of the aorta located in the chest that offers a combination of safety, effectiveness, and convenience for managing thoracic aortic diseases. For instance, in February 2024, Endovastec, a China-based medical device company, introduced the Multi-Branch Thoracic Aortic Stent-Graft System (Hector) and completed its first clinical application in Switzerland. The Hector system represents a significant advancement in endovascular stent graft technology. Its multi-branch design allows for treating complex thoracic aortic conditions by accommodating and preserving essential branches of the aorta, such as the arteries that supply vital organs.What Are Latest Mergers And Acquisitions In The Endovascular Stent Grafts Market?
In December 2023, TekniPlex Healthcare Inc., a US-based provider of medical device components, acquired Seisa Medical S.A. de C.V. for an undisclosed amount. With this acquisition, it aims to expand its medical device capabilities and enhance its presence in the Latin American healthcare market. Seisa Medical S.A. de C.V. is a Mexico-based company that specializes in manufacturing endovascular stent grafts.Regional Outlook
North America was the largest region in the endovascular stent grafts market in 2025. The regions covered in this market report are Asia-Pacific, South East Asia, Western Europe, Eastern Europe, North America, South America, Middle East, Africa. The countries covered in this market report are Australia, Brazil, China, France, Germany, India, Indonesia, Japan, Taiwan, Russia, South Korea, UK, USA, Canada, Italy, SpainWhat Defines the Endovascular Stent Grafts Market?
The endovascular stent grafts market consists of sales of aortoiliac stent grafts, thoracic endograft, and abdominal endografts. Values in this market are ‘factory gate’ values, that is the value of goods sold by the manufacturers or creators of the goods, whether to other entities (including downstream manufacturers, wholesalers, distributors, and retailers) or directly to end customers. The value of goods in this market includes related services sold by the creators of the goods.How is Market Value Defined and Measured?
The market value is defined as the revenues that enterprises gain from the sale of goods and/or services within the specified market and geography through sales, grants, or donations in terms of the currency (in USD unless otherwise specified). The revenues for a specified geography are consumption values that are revenues generated by organizations in the specified geography within the market, irrespective of where they are produced. It does not include revenues from resales along the supply chain, either further along the supply chain or as part of other products.Endovascular Stent Grafts Market Report Forecast Analysis
| Report Attribute | Details |
|---|---|
| Market Size Value In 2026 | $1.99 billion |
| Revenue Forecast In 2035 | $7.13 billion |
| Growth Rate | CAGR of 37.9% from 2026 to 2035 |
| Base Year For Estimation | 2025 |
| Actual Estimates/Historical Data | 2020-2025 |
| Forecast Period | 2026 - 2030 - 2035 |
| Market Representation | Revenue in USD Billion and CAGR from 2026 to 2035 |
| Segments Covered | Product, Raw Material, Application |
| Regional Scope | Asia-Pacific, Western Europe, Eastern Europe, North America, South America, Middle East, Africa |
| Country Scope | The countries covered in the report are Australia, Brazil, China, France, Germany, India, ... |
| Key Companies Profiled | Abbott Laboratories, Medtronic PLC, Boston Scientific Corporation, Terumo Corporation, Cook Group Incorporated, Merit Medical Systems Inc., Biotronik SE & Co KG, MicroPort Scientific Corporation, Endologix LLC, LeMaitre Vascular Inc., Lifetech Scientific Corporation, W. L. Gore & Associates Inc., Braile Biomedica, BrosMed Medical BV, Micro Interventional Devices Inc. (MID), Lombard Medical Inc., Endovastec Medical Technology Co Ltd., Jotec GmbH, Bentley InnoMed GmbH, Bolton Medical Inc., Alvimedica, Endospan Ltd., Vivasure Medical Ltd. |
| Customization Scope | Request for Customization |
| Pricing And Purchase Options | Explore Purchase Options |
