
Extreme Lateral Interbody Fusion (XLIF) Surgery Market Report 2026
Global Outlook – By Product Type (Extreme Lateral Interbody Fusion (XLIF) Interbody Fusion Systems, Extreme Lateral Interbody Fusion (XLIF) Interbody Cages), By Procedure Type (Single Segment, Multi-Segment, Mini Open), By Application (Spinal Stenosis, Herniated Disc, Degenerative Disc Disease), By End-User (Hospitals, Ambulatory Surgical Centers, Orthopedic Clinic) - Market Size, Trends, And Global Forecast 2026-2035
Extreme Lateral Interbody Fusion (XLIF) Surgery Market Overview
• Extreme Lateral Interbody Fusion (XLIF) Surgery market size has reached to $1.57 billion in 2025 • Expected to grow to $2.05 billion in 2030 at a compound annual growth rate (CAGR) of 5.5% • Growth Driver: Surge In Rising Incidence Of Spinal Illnesses Fueling The Growth Of The Market Due To Aging Populations And Lifestyle Factors • Market Trend: Technological Advancements Drive Innovation In Lateral Plating For XLIF Surgery • North America was the largest region in 2025 and Asia-Pacific is the fastest growing region.What Is Covered Under Extreme Lateral Interbody Fusion (XLIF) Surgery Market?
Extreme lateral interbody fusion (XLIF) surgery is a minimally invasive spinal fusion procedure used to treat various spinal disorders. It involves accessing the spine through a lateral (side) approach, avoiding major back muscles and reducing recovery time, while using specialized surgical instruments and neuromonitoring for safety. The main extreme lateral interbody fusion (xlif) surgery products include extreme lateral interbody fusion (xlif) interbody fusion systems and extreme lateral interbody fusion (xlif) interbody cages. It is used for various procedures such as single segment, multi-segment, and mini open and is applied for spinal stenosis, herniated disc, and degenerative disc disease. It is used by various end-users including hospitals, ambulatory surgical centers, and orthopedic clinic.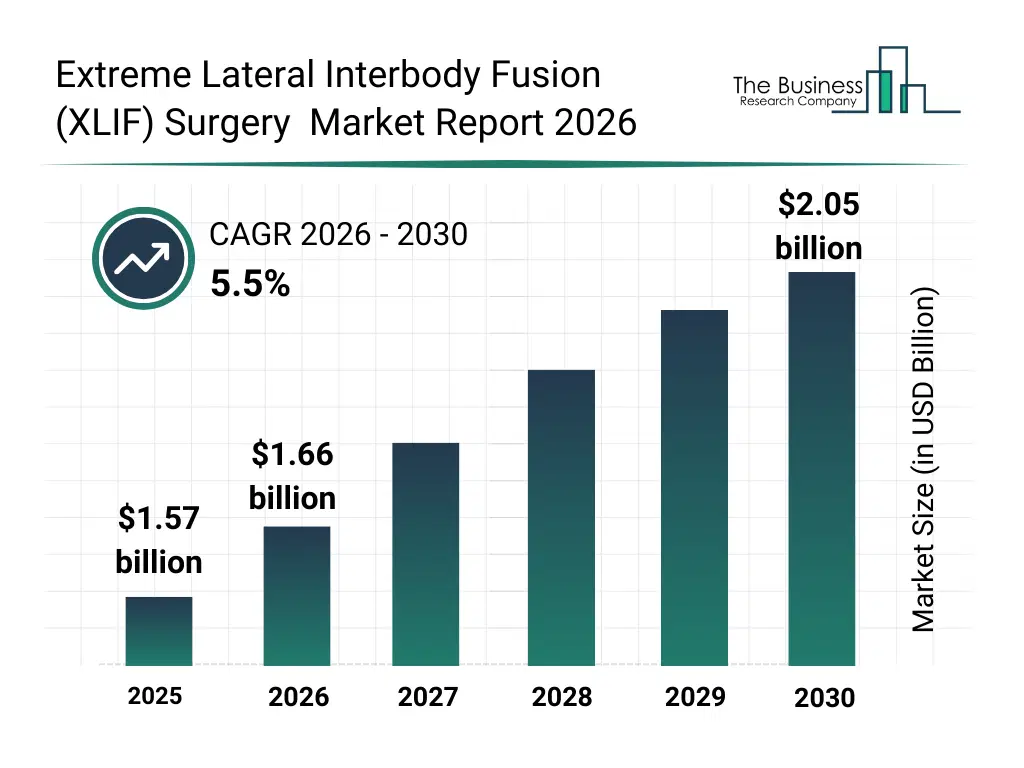
What Is The Extreme Lateral Interbody Fusion (XLIF) Surgery Market Size and Share 2026?
The extreme lateral interbody fusion (xlif) surgery market size has grown strongly in recent years. It will grow from $1.57 billion in 2025 to $1.66 billion in 2026 at a compound annual growth rate (CAGR) of 5.8%. The growth in the historic period can be attributed to increasing prevalence of degenerative disc disease, growth in spinal disorder cases, rising demand for minimally invasive surgery, improved surgical instrumentation, expansion of orthopedic hospitals.What Is The Extreme Lateral Interbody Fusion (XLIF) Surgery Market Growth Forecast?
The extreme lateral interbody fusion (xlif) surgery market size is expected to see strong growth in the next few years. It will grow to $2.05 billion in 2030 at a compound annual growth rate (CAGR) of 5.5%. The growth in the forecast period can be attributed to aging population growth, technological advancements in spinal implants, increasing surgeon preference for lateral approaches, rising healthcare infrastructure investments, growing awareness of minimally invasive spine surgery. Major trends in the forecast period include rising adoption of minimally invasive spine procedures, growing demand for faster post-surgical recovery, increased use of advanced interbody cages, expansion of specialized spine surgery centers, improved surgical outcomes through lateral access techniques.Global Extreme Lateral Interbody Fusion (XLIF) Surgery Market Segmentation
1) By Product Type: Extreme Lateral Interbody Fusion (XLIF) Interbody Fusion Systems, Extreme Lateral Interbody Fusion (XLIF) Interbody Cages 2) By Procedure Type: Single Segment, Multi-Segment, Mini Open 3) By Application: Spinal Stenosis, Herniated Disc, Degenerative Disc Disease 4) By End-User: Hospitals, Ambulatory Surgical Centers, Orthopedic Clinic Subsegments: 1) By XLIF Interbody Fusion Systems: Minimally Invasive XLIF Systems, Stand-Alone XLIF Systems, Expandable XLIF Systems, Biomechanical XLIF System 2) By XLIF Interbody Cages: PEEK (Polyether Ether Ketone) XLIF Cages, Titanium XLIF Cages, 3D-Printed XLIF Cages, Expandable XLIF CagesWhat Is The Driver Of The Extreme Lateral Interbody Fusion (XLIF) Surgery Market?
The rising incidence of spinal illnesses is expected to fuel the growth of the extreme lateral interbody fusion (XLIF) surgery market during the forecast period. Spinal illnesses refer to a range of medical conditions that affect the spine, including its bones (vertebrae), discs, nerves, spinal cord, and surrounding muscles and ligaments. A key factor driving the rise in spinal illnesses is sedentary lifestyles, as extended periods of sitting and limited physical activity contribute to poor posture and weakened core muscles, which heighten the risk of spinal degeneration and chronic back pain. XLIF surgery is required for spinal illnesses because it minimally invasively restores disc height and spinal alignment, relieving nerve compression and stabilizing the spine without cutting through major back muscles. For instance, in June 2024, according to the Australian Institute of Health and Welfare, an Australia-based government agency, by 2023, back problems had become the third leading cause of disease burden in the country, contributing to 4.3% of the total disease burden. Therefore, the rising incidence of spinal illnesses is driving the growth of the extreme lateral interbody fusion (XLIF) surgery industry.Key Players In The Global Extreme Lateral Interbody Fusion (XLIF) Surgery Market
Major companies operating in the extreme lateral interbody fusion (xlif) surgery market are Medtronic plc, Stryker Corporation, Zimmer Biomet Holdings Inc., NuVasive Inc., Globus Medical Inc., Orthofix Medical Inc., Alphatec Holdings Inc. (Alphatec Spine), Spinal Elements Inc., Surgalign Holdings Inc., Spineology Inc., CoreLink LLC, Aurora Spine Corporation, Life Spine Inc., Premia Spine Ltd., Nexxt Spine LLC, Spinal Simplicity LLC, CoreSpine Technologies LLC, NU-Spine LLC, ATEC Spine Inc., Captiva Spine Inc., Xtant Medical Holdings Inc., Centinel Spine LLCGlobal Extreme Lateral Interbody Fusion (XLIF) Surgery Market Trends and Insights
Major companies in the extreme lateral interbody fusion (XLIF) surgery market are focusing on developing technologically advanced products, such as lateral plating systems, to reduce operative time and promote faster patient recovery. Lateral plating systems are orthopedic implants used in spine surgery to provide stabilization and support by attaching plates to the side (lateral aspect) of the vertebrae, often in conjunction with interbody fusion procedures, including XLIF. For instance, in August 2024, Globus Medical Inc., a US-based medical device manufacturer, launched the ADIRA XLIF Plate System. The ADIRA XLIF Plate System offers a streamlined lateral plating solution with a secure coupling mechanism that aligns plates over interbody spacers for greater stability. Compatible with bone screws and lateral MIS anchors, it supports various fixation methods and spacer types. Integrated with the Globus Medical lateral portfolio, it threads directly into multiple interbody spacers to reduce the risk of spacer movement and adapt to different patient needs and surgical preferences, enhancing both efficiency and reliability.What Are Latest Mergers And Acquisitions In The Extreme Lateral Interbody Fusion (XLIF) Surgery Market?
In September 2023, Globus Medical, a US-based medical device company, merged with NuVasive to create a global musculoskeletal company. As a result of the merger between Globus Medical and NuVasive, the combined company will deliver one of the most comprehensive portfolios of musculoskeletal procedural solutions and enabling technologies, enhancing care across the entire treatment continuum. This strategic union brings together complementary global scale, an expanded commercial presence, and robust offerings in both spine and orthopedic segments. It also strengthens the companies' shared commitment to innovation, surgeon education, and operational excellence. NuVasive is a US-based medical device company specializing in XLIF products and solutions.Regional Outlook
North America was the largest region in the extreme lateral interbody fusion (XLIF) surgery market in 2025. Asia-Pacific is expected to be the fastest-growing region in the forecast period. The regions covered in this market report are Asia-Pacific, South East Asia, Western Europe, Eastern Europe, North America, South America, Middle East, Africa. The countries covered in this market report are Australia, Brazil, China, France, Germany, India, Indonesia, Japan, Taiwan, Russia, South Korea, UK, USA, Canada, Italy, Spain.What Defines the Extreme Lateral Interbody Fusion (XLIF) Surgery Market?
The extreme lateral interbody fusion (XLIF) surgery market consists of revenues earned by entities by providing services such as surgical training, intraoperative support, postoperative monitoring, reprocessing and sterilization services. The market value includes the value of related goods sold by the service provider or included within the service offering. The extreme lateral interbody fusion (XLIF) surgery market also includes sales of surgical access systems, fixation devices, neuromonitoring equipment and bone graft materials. Values in this market are ‘factory gate’ values, that is the value of goods sold by the manufacturers or creators of the goods, whether to other entities (including downstream manufacturers, wholesalers, distributors and retailers) or directly to end customers. The value of goods in this market includes related services sold by the creators of the goods.How is Market Value Defined and Measured?
The market value is defined as the revenues that enterprises gain from the sale of goods and/or services within the specified market and geography through sales, grants, or donations in terms of the currency (in USD unless otherwise specified). The revenues for a specified geography are consumption values that are revenues generated by organizations in the specified geography within the market, irrespective of where they are produced. It does not include revenues from resales along the supply chain, either further along the supply chain or as part of other products.What Key Data and Analysis Are Included in the Extreme Lateral Interbody Fusion (XLIF) Surgery Market Report 2026?
The extreme lateral interbody fusion (xlif) surgery market research report is one of a series of new reports from The Business Research Company that provides market statistics, including industry global market size, regional shares, competitors with the market share, detailed market segments, market trends and opportunities, and any further data you may need to thrive in the extreme lateral interbody fusion (xlif) surgery industry. The market research report delivers a complete perspective of everything you need, with an in-depth analysis of the current and future state of the industry.Extreme Lateral Interbody Fusion (XLIF) Surgery Market Report Forecast Analysis
| Report Attribute | Details |
|---|---|
| Market Size Value In 2026 | $1.66 billion |
| Revenue Forecast In 2035 | $2.05 billion |
| Growth Rate | CAGR of 5.8% from 2026 to 2035 |
| Base Year For Estimation | 2025 |
| Actual Estimates/Historical Data | 2020-2025 |
| Forecast Period | 2026 - 2030 - 2035 |
| Market Representation | Revenue in USD Billion and CAGR from 2026 to 2035 |
| Segments Covered | Product Type, Procedure Type, Application, End-User |
| Regional Scope | Asia-Pacific, Western Europe, Eastern Europe, North America, South America, Middle East, Africa |
| Country Scope | The countries covered in the report are Australia, Brazil, China, France, Germany, India, ... |
| Key Companies Profiled | Medtronic plc, Stryker Corporation, Zimmer Biomet Holdings Inc., NuVasive Inc., Globus Medical Inc., Orthofix Medical Inc., Alphatec Holdings Inc. (Alphatec Spine), Spinal Elements Inc., Surgalign Holdings Inc., Spineology Inc., CoreLink LLC, Aurora Spine Corporation, Life Spine Inc., Premia Spine Ltd., Nexxt Spine LLC, Spinal Simplicity LLC, CoreSpine Technologies LLC, NU-Spine LLC, ATEC Spine Inc., Captiva Spine Inc., Xtant Medical Holdings Inc., Centinel Spine LLC |
| Customization Scope | Request for Customization |
| Pricing And Purchase Options | Explore Purchase Options |
