
Fabry Disease Treatment Market Report 2026
Global Outlook – By Treatment (Substrate Reduction Therapy, Enzyme Replacement Therapy, Chaperone Treatment, Other Treatments), By Route Of Administration (Oral Route, Intravenous Route), By Disease Severity (Classic Fabry Disease, Late-Onset Fabry Disease), By Distribution Channel (Hospital Pharmacies, Retail Pharmacies, Online Pharmacies) - Market Size, Trends, And Global Forecast 2026-2035
Fabry Disease Treatment Market Overview
• Fabry Disease Treatment market size has reached to $2.27 billion in 2025 • Expected to grow to $3.34 billion in 2030 at a compound annual growth rate (CAGR) of 7.8% • Growth Driver: Impact of Increasing Renal Disease Prevalence on the Fabry Disease Treatment Market • Market Trend: Advancements in Plant Cell-Based Treatment for Fabry Disease • North America was the largest region in 2025 and Asia-Pacific is the fastest growing region.What Is Covered Under Fabry Disease Treatment Market?
Fabry disease is a rare genetic disorder caused by a deficiency of the enzyme alpha-galactosidase A. This enzyme deficiency leads to the buildup of a type of fat called globotriaosylceramide (Gb3 or GL-3) in the body's cells, which can cause a wide range of symptoms and complications, including pain, kidney failure, heart disease, and stroke. The primary purpose of treating fabry disease is to reduce the accumulation of Gb3 in the body, alleviate symptoms, prevent or delay the progression of organ damage, and improve the overall quality of life for patients. The main types of treatments in fabry disease treatment market are substrate reduction therapy, enzyme replacement therapy, chaperone treatment, and others. Substrate reduction therapy (SRT) is a type of treatment for fabry disease that works by reducing the buildup of a specific substance in the body. These treatment medications can be administered through oral and intravenous routes. The different disease severities include classic fabry disease, and late-onset fabry disease. They can be distributed through various channels including hospital pharmacies, retail pharmacies, and online pharmacies.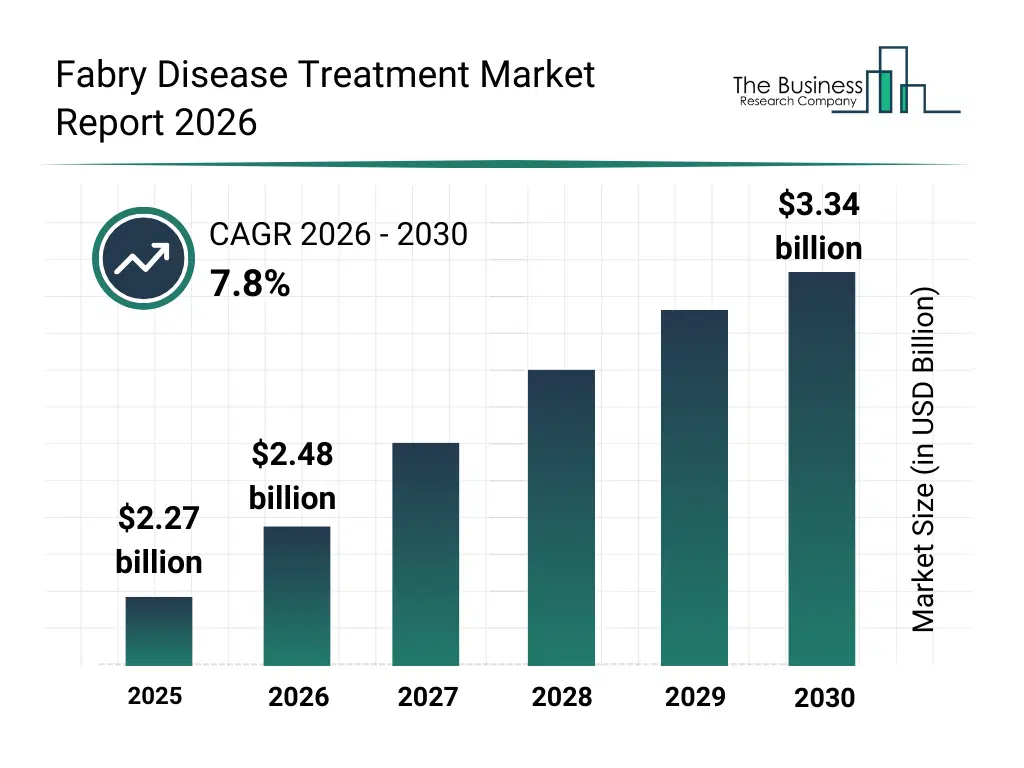
What Is The Fabry Disease Treatment Market Size and Share 2026?
The fabry disease treatment market size has grown strongly in recent years. It will grow from $2.27 billion in 2025 to $2.48 billion in 2026 at a compound annual growth rate (CAGR) of 8.8%. The growth in the historic period can be attributed to improved awareness of rare genetic disorders, approval of enzyme replacement therapies, expansion of genetic testing, growth in specialty hospitals, increased patient advocacy.What Is The Fabry Disease Treatment Market Growth Forecast?
The fabry disease treatment market size is expected to see strong growth in the next few years. It will grow to $3.34 billion in 2030 at a compound annual growth rate (CAGR) of 7.8%. The growth in the forecast period can be attributed to advancements in gene therapy, growing use of personalized medicine, improved newborn screening programs, expansion of oral therapies, increased rare disease funding. Major trends in the forecast period include increasing adoption of enzyme replacement therapy, growing focus on early genetic diagnosis, expansion of oral chaperone therapies, rising research in gene therapy approaches, improved patient access to rare disease treatments.Global Fabry Disease Treatment Market Segmentation
1) By Treatment: Substrate Reduction Therapy, Enzyme Replacement Therapy, Chaperone Treatment, Other Treatments 2) By Route Of Administration: Oral Route, Intravenous Route 3) By Disease Severity: Classic Fabry Disease, Late-Onset Fabry Disease 4) By Distribution Channel: Hospital Pharmacies, Retail Pharmacies, Online Pharmacies Subsegments: 1) By Substrate Reduction Therapy: Migalastat Hydrochloride (Galafold), Other Substrate Reduction Therapies 2) By Enzyme Replacement Therapy: Agalsidase Beta (Fabrazyme), Agalsidase Alfa (Replagal), Other Enzyme Replacement Therapies 3) By Chaperone Treatment: Migalastat Hydrochloride (Galafold), Other Chaperone Therapies 4) By Other Treatments: Gene Therapy, Symptomatic TreatmentWhat Is The Driver Of The Fabry Disease Treatment Market?
The increasing prevalence of renal diseases is expected to propel the growth of the fabry disease treatment market going forward. Renal diseases, also known as kidney diseases, refer to any conditions impairing the kidneys' function. The cases of renal diseases are growing due to genetic predispositions, lifestyle choices, environmental factors, and underlying health conditions. Fabry disease is caused by the accumulation of globotriaosylceramide (Gb3) in kidney cells, leading to damage and dysfunction, highlighting the critical need for early detection and intervention to prevent kidney complications. For instance, in March 2023, according to the International Society of Nephrology (ISN), a Belgium-based professional society, approximately 850 million people worldwide lived with chronic kidney disease (CKD), marking an increase compared to prior years. Therefore, the increasing prevalence of renal diseases is driving the growth of the fabry disease treatment industry.Key Players In The Global Fabry Disease Treatment Market
Major companies operating in the fabry disease treatment market are Sanofi S.A., Takeda Pharmaceutical Company Limited, Ultragenyx Pharmaceutical Inc., Sangamo Therapeutics Inc., Protalix BioTherapeutics, Avrobio Inc., Freeline Therapeutics Holdings PLC, M6P Therapeutics Inc., Yuhan Corporation, Orchard Therapeutics Ltd., Regenxbio Inc., Synlogic Inc., AceLink Therapeutics Inc., Greenovation Biotech GmbH, CANbridge Life Sciences Ltd., Sigilon Therapeutics Inc., BioMarin Pharmaceutical Inc., Sarepta Therapeutics Inc., Daiichi Sankyo Company Ltd., Audentes Therapeutics Inc., Genesis Therapeutics Inc., Idorsia Pharmaceuticals Ltd., JCR Pharmaceuticals Co Ltd., ISU Abxis Co Ltd.Global Fabry Disease Treatment Market Trends and Insights
Major companies operating in the Fabry disease treatment market are developing plant cell-based treatments to enhance therapeutic efficacy, reduce treatment costs, and improve patient outcomes through innovative and sustainable approaches. Plant cell-based treatment for fabry disease involves using plant cells to produce deficient enzymes in individuals with fabry disease. These plant-derived enzymes can be used as a therapeutic option to replace the missing or defective enzymes in the body, aiming to alleviate symptoms and improve the health of patients affected by Fabry disease. For instance, in May 2023, Chiesi Global Rare Diseases, an Italy-based pharmaceutical company, and Protalix BioTherapeutics Inc., an Israel-based pharmaceutical company, received approval for ELFABRIO. ELFABRIO presents an alternative treatment choice for individuals with Fabry disease. It is an enzyme replacement therapy (ERT) where the recombinant human α-Galactosidase-A enzyme is produced in plant-cell culture and modified with PEGylation to extend its half-life significantly. This treatment reduces frequency, enhances convenience, and improves the overall management of Fabry disease symptoms, enhancing patient quality of life.What Are Latest Mergers And Acquisitions In The Fabry Disease Treatment Market?
In March 2024, CENTOGENE, a Germany-based biotechnology research company, extended its partnership with Takeda Pharmaceutical Company Limited. This partnership ensures Takeda's ongoing access to diagnostic testing for patients worldwide. The agreement improves patient access to quick and reliable diagnostics for lysosomal storage disorders (LSDs), such as fabry disease, Gaucher disease, and Hunter syndrome. Takeda Pharmaceutical Company Limited is a Japan-based pharmaceutical company that concentrates on developing therapies for lysosomal storage disorders such as fabry disease.Regional Outlook
North America was the largest region in the fabry disease treatment market in 2025. Asia-Pacific is expected to be the fastest-growing region in the forecast period. The regions covered in this market report are Asia-Pacific, South East Asia, Western Europe, Eastern Europe, North America, South America, Middle East, Africa. The countries covered in this market report are Australia, Brazil, China, France, Germany, India, Indonesia, Japan, Taiwan, Russia, South Korea, UK, USA, Canada, Italy, Spain.What Defines the Fabry Disease Treatment Market?
The fabry disease treatment market consists of revenues earned by entities by providing services such as diagnosis and screening, enzyme replacement therapy, gene therapy, disease management programs, and patient education and support. The market value includes the value of related goods sold by the service provider or included within the service offering. The Fabry disease treatment market also includes sales of Agalsidase alfa, Agalsidase beta, and Migalastat. Values in this market are ‘factory gate’ values, that is the value of goods sold by the manufacturers or creators of the goods, whether to other entities (including downstream manufacturers, wholesalers, distributors and retailers) or directly to end customers. The value of goods in this market includes related services sold by the creators of the goods.How is Market Value Defined and Measured?
The market value is defined as the revenues that enterprises gain from the sale of goods and/or services within the specified market and geography through sales, grants, or donations in terms of the currency (in USD unless otherwise specified). The revenues for a specified geography are consumption values that are revenues generated by organizations in the specified geography within the market, irrespective of where they are produced. It does not include revenues from resales along the supply chain, either further along the supply chain or as part of other products.What Key Data and Analysis Are Included in the Fabry Disease Treatment Market Report 2026?
The fabry disease treatment market research report is one of a series of new reports from The Business Research Company that provides market statistics, including industry global market size, regional shares, competitors with the market share, detailed market segments, market trends and opportunities, and any further data you may need to thrive in the fabry disease treatment industry. The market research report delivers a complete perspective of everything you need, with an in-depth analysis of the current and future state of the industry.Fabry Disease Treatment Market Report Forecast Analysis
| Report Attribute | Details |
|---|---|
| Market Size Value In 2026 | $2.48 billion |
| Revenue Forecast In 2035 | $3.34 billion |
| Growth Rate | CAGR of 8.8% from 2026 to 2035 |
| Base Year For Estimation | 2025 |
| Actual Estimates/Historical Data | 2020-2025 |
| Forecast Period | 2026 - 2030 - 2035 |
| Market Representation | Revenue in USD Billion and CAGR from 2026 to 2035 |
| Segments Covered | Treatment, Route Of Administration, Disease Severity, Distribution Channel |
| Regional Scope | Asia-Pacific, Western Europe, Eastern Europe, North America, South America, Middle East, Africa |
| Country Scope | The countries covered in the report are Australia, Brazil, China, France, Germany, India, ... |
| Key Companies Profiled | Sanofi S.A., Takeda Pharmaceutical Company Limited, Ultragenyx Pharmaceutical Inc., Sangamo Therapeutics Inc., Protalix BioTherapeutics, Avrobio Inc., Freeline Therapeutics Holdings PLC, M6P Therapeutics Inc., Yuhan Corporation, Orchard Therapeutics Ltd., Regenxbio Inc., Synlogic Inc., AceLink Therapeutics Inc., Greenovation Biotech GmbH, CANbridge Life Sciences Ltd., Sigilon Therapeutics Inc., BioMarin Pharmaceutical Inc., Sarepta Therapeutics Inc., Daiichi Sankyo Company Ltd., Audentes Therapeutics Inc., Genesis Therapeutics Inc., Idorsia Pharmaceuticals Ltd., JCR Pharmaceuticals Co Ltd., ISU Abxis Co Ltd. |
| Customization Scope | Request for Customization |
| Pricing And Purchase Options | Explore Purchase Options |
