
Fast Sterility Testing By Adenosine Triphosphate (ATP) Bioluminescence Market Report 2026
Global Outlook – By Technology (Adenosine Triphosphate Bioluminescence, Membrane Filtration, Direct Inoculation), By Product Type (Instruments, Reagents And Kits, Consumables), By Application (Pharmaceutical And Biotechnology, Food And Beverage, Medical Devices, Water And Environmental, Cosmetics And Personal Care, Other Applications), By End-User (Hospitals And Clinics, Laboratories, Research Institutes, Other End-Users) – Market Size, Trends, Strategies, and Forecast to 2035
Fast Sterility Testing By Adenosine Triphosphate (ATP) Bioluminescence Market Overview
• Fast Sterility Testing By Adenosine Triphosphate (ATP) Bioluminescence market size has reached to $0.96 billion in 2025 • Expected to grow to $1.76 billion in 2030 at a compound annual growth rate (CAGR) of 12.9% • Growth Driver: Growth In Pharmaceutical And Biotechnology Sectors Driving The Market Growth Due To Increasing Demand for Rapid and Reliable Contamination Detection • Market Trend: Accelerating Sterility Testing With Rapid ATP Bioluminescence For Faster, Safer Pharmaceutical Batch Release • North America was the largest region in 2025 and Asia-Pacific is the fastest growing region.What Is Covered Under Fast Sterility Testing By Adenosine Triphosphate (ATP) Bioluminescence Market?
Fast sterility testing by adenosine triphosphate (ATP) bioluminescence is a rapid method for detecting microbial contamination by measuring the presence of ATP, a universal energy molecule found in all living cells. The enzyme luciferase catalyzes a reaction with ATP, producing light proportional to the number of viable microorganisms. The intensity of the emitted light provides near-instantaneous assessment of sterility compared to traditional culture-based methods. The main technologies of fast sterility testing by adenosine triphosphate bioluminescence include adenosine triphosphate bioluminescence, membrane filtration, and direct inoculation. Adenosine triphosphate (ATP) bioluminescence refers to a method that uses light emission from ATP-driven reactions to detect and quantify living cells or microbial contamination. The various product types include instruments, reagents and kits, and consumables. They find application in pharmaceutical and biotechnology, Food And Beverage, medical devices, water and environmental, cosmetics and personal care, and other applications, serving end users including hospitals and clinics, laboratories, research institutes, and other end-users.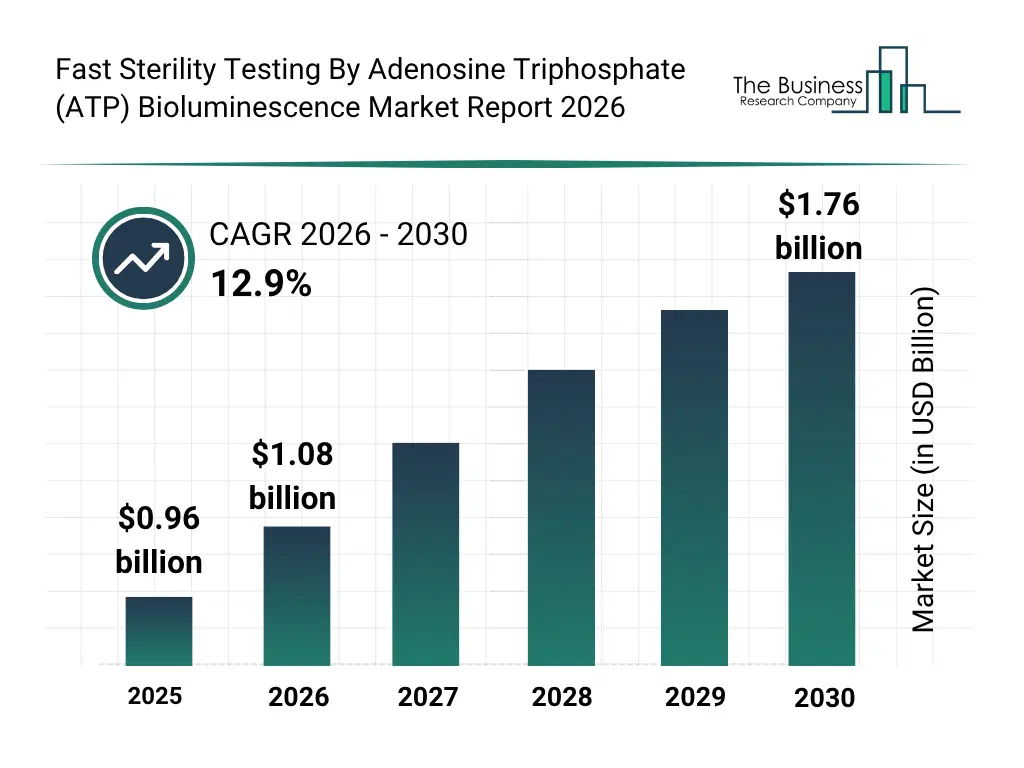
What Is The Fast Sterility Testing By Adenosine Triphosphate (ATP) Bioluminescence Market Size and Share 2026?
The fast sterility testing by adenosine triphosphate (ATP) bioluminescence market size has grown rapidly in recent years. It will grow from $0.96 billion in 2025 to $1.08 billion in 2026 at a compound annual growth rate (CAGR) of 12.7%. The growth in the historic period can be attributed to increasing need for faster sterility testing in pharmaceuticals, limitations of traditional culture-based methods, rising regulatory emphasis on contamination control, growth of biotechnology manufacturing facilities, expanding hospital infection control programs.What Is The Fast Sterility Testing By Adenosine Triphosphate (ATP) Bioluminescence Market Growth Forecast?
The fast sterility testing by adenosine triphosphate (ATP) bioluminescence market size is expected to see rapid growth in the next few years. It will grow to $1.76 billion in 2030 at a compound annual growth rate (CAGR) of 12.9%. The growth in the forecast period can be attributed to growing adoption of automated rapid testing platforms, increasing investment in advanced microbial detection technologies, rising demand from food and beverage safety testing, expansion of cleanroom manufacturing environments, increasing focus on quality assurance and compliance standards. Major trends in the forecast period include increasing adoption of rapid microbial detection methods, rising demand for real-time sterility assessment, growing use of luminometer based systems, expansion of automated bioluminescence analyzers, increasing replacement of culture-based testing methods.Global Fast Sterility Testing By Adenosine Triphosphate (ATP) Bioluminescence Market Segmentation
1) By Technology: Adenosine Triphosphate Bioluminescence, Membrane Filtration, Direct Inoculation 2) By Product Type: Instruments, Reagents And Kits, Consumables 3) By Application: Pharmaceutical And Biotechnology, Food And Beverage, Medical Devices, Water And Environmental, Cosmetics And Personal Care, Other Applications 4) By End-User: Hospitals And Clinics, Laboratories, Research Institutes, Other End-Users Subsegments: 1) By Adenosine Triphosphate Bioluminescence: Luminometer Based Systems, Handheld Rapid Detection Kits, Automated Bioluminescence Analyzers 2) By Membrane Filtration: Vacuum Filtration Units, Gravity Filtration Systems, Automated Membrane Filtration Instruments 3) By Direct Inoculation: Broth Culture Methods, Agar Plate Culture Methods, Automated Inoculation SystemsWhat Is The Driver Of The Fast Sterility Testing By Adenosine Triphosphate (ATP) Bioluminescence Market?
The growth in pharmaceutical and biotechnology sectors is expected to propel the growth of the fast sterility testing by adenosine triphosphate (ATP) bioluminescence market going forward. The pharmaceutical and biotechnology sectors refer to industries engaged in the research, development, production, and commercialization of drugs, vaccines, and biologically-based therapies to prevent or treat diseases. The pharmaceutical and biotechnology sectors are rising due to increasing investment in research and development, which enables companies to discover new drugs, improve therapies, and expand production capabilities to meet growing healthcare demands. Fast sterility testing by adenosine triphosphate (ATP) bioluminescence supports the pharmaceutical and biotechnology sectors by rapidly detecting microbial contamination, ensuring the safety and quality of drugs and biologics, and enabling faster production while maintaining compliance with strict regulatory standards. For instance, in April 2025, according to Eurostat, a Luxembourg-based government agency, in 2024, EU exports of medicinal and pharmaceutical products increased by 13.5% compared with 2023, reaching $316.38 billion (€313.4 billion). Furthermore, in January 2025, according to the BioIndustry Association (BIA), a UK-based trade association, the UK biotech sector experienced substantial growth in 2024, securing £3.5 billion (approximately $ 4.4 billion) in investment, an impressive 94% incrDIGITease from the previous year. Therefore, growth in pharmaceutical and biotechnology sectors is driving the growth of the fast sterility testing by adenosine triphosphate (ATP) bioluminescence industry.Key Players In The Global Fast Sterility Testing By Adenosine Triphosphate (ATP) Bioluminescence Market
Major companies operating in the fast sterility testing by adenosine triphosphate (atp) bioluminescence market are 3M Company, Merck KGaA, Lonza Group AG, SGS SA, Kikkoman Biochemifa, Charles River Laboratories, BioMérieux, Hach Company, Neogen Corporation, Charm Sciences, Promega Corporation, LuminUltra Technologies, Sopex d.o.o., Nelson Laboratories LLC, Hygiena LLC, PCE Instruments, Berthold Technologies, BioThema AB, Pharmetric Laboratory, Biotactical B.V., Promicol BV.Global Fast Sterility Testing By Adenosine Triphosphate (ATP) Bioluminescence Market Trends and Insights
Major companies operating in the fast sterility testing by adenosine triphosphate (ATP) bioluminescence market are focusing on developing innovative methods such as, accelerated sterility tests to shorten batch release times and improve QC efficiency. Accelerated sterility tests, which use rapid detection methods such as ATP bioluminescence to identify microbial contamination, help shorten batch release times, reduce quarantine periods, and improve overall quality control efficiency. For instance, in January 2026, Biotactical B.V., a Netherlands-based biotechnology company, launched its accelerated rapid sterility testing platform, an innovative solution that combines ATP bioluminescence with accelerated growth media to detect microbial contamination in just 48 hours. The system is designed for GMP-compliant environments and supports complex, short-shelf-life products, including advanced therapeutic medicinal products (ATMPs). The platform provides accelerated detection, reduces handling time, minimizes contamination risk, and aligns with USP <71>, <73>, <1223> and Ph. Eur. 2.6 and 5.1.6 standards, enabling faster batch release and more efficient inventory management while addressing a key bottleneck in pharmaceutical quality control workflows.What Are Latest Mergers And Acquisitions In The Fast Sterility Testing By Adenosine Triphosphate (ATP) Bioluminescence Market?
In September 2025, LuminUltra Technologies Ltd., a Canada-based healthcare technology company, acquired GL Biocontrol for an undisclosed amount. With this acquisition, LuminUltra aims to expand its European presence, enhance its microbial water diagnostics capabilities, and accelerate the development and deployment of innovative, rapid testing solutions that address regulatory compliance and operational efficiency for municipal and industrial water systems. GL Biocontrol SAS is a France-based technology company that specializes in providing rapid microbiological testing based on 2nd generation ATP bioluminescence technology.Regional Insights
North America was the largest region in the fast sterility testing by adenosine triphosphate (ATP) bioluminescence market in 2025. Asia-Pacific is expected to be the fastest-growing region in the forecast period. The regions covered in this market report are Asia-Pacific, South East Asia, Western Europe, Eastern Europe, North America, South America, Middle East, Africa. The countries covered in this market report are Australia, Brazil, China, France, Germany, India, Indonesia, Japan, Taiwan, Russia, South Korea, UK, USA, Canada, Italy, Spain.What Defines the Fast Sterility Testing By Adenosine Triphosphate (ATP) Bioluminescence Market?
The fast sterility testing by adenosine triphosphate (ATP) bioluminescence market consists of revenues earned by entities by providing services such as rapid sterility assessment of pharmaceutical and medical products, environmental monitoring in cleanrooms and manufacturing facilities, quality control testing for food and beverage industries, and validation or verification of sterilization processes. The market value includes the value of related goods sold by the service provider or included within the service offering. The fast sterility testing by adenosine triphosphate (ATP) bioluminescence market also includes sales of luminometers and handheld ATP meters, ATP reagents and substrates, swabs and sample collection devices. Values in this market are ‘factory gate’ values, that is the value of goods sold by the manufacturers or creators of the goods, whether to other entities (including downstream manufacturers, wholesalers, distributors and retailers) or directly to end customers. The value of goods in this market includes related services sold by the creators of the goods.How is Market Value Defined and Measured?
The market value is defined as the revenues that enterprises gain from the sale of goods and/or services within the specified market and geography through sales, grants, or donations in terms of the currency (in USD unless otherwise specified). The revenues for a specified geography are consumption values that are revenues generated by organizations in the specified geography within the market, irrespective of where they are produced. It does not include revenues from resales along the supply chain, either further along the supply chain or as part of other products.What Key Data And Analysis Are Included In The Fast Sterility Testing By Adenosine Triphosphate (ATP) Bioluminescence Market Report 2026?
The fast sterility testing by adenosine triphosphate (atp) bioluminescence market research report is one of a series of new reports from The Business Research Company that provides market statistics, including industry global market size, regional shares, competitors with the market share, detailed market segments, market trends and opportunities, and any further data you may need to thrive in the fast sterility testing by adenosine triphosphate (atp) bioluminescence industry. The market research report delivers a complete perspective of everything you need, with an in-depth analysis of the current and future state of the industry.Fast Sterility Testing By Adenosine Triphosphate (ATP) Bioluminescence Market Report Forecast Analysis
| Report Attribute | Details |
|---|---|
| Market Size Value In 2026 | $1.08 billion |
| Revenue Forecast In 2035 | $1.76 billion |
| Growth Rate | CAGR of 12.7% from 2026 to 2035 |
| Base Year For Estimation | 2025 |
| Actual Estimates/Historical Data | 2020-2025 |
| Forecast Period | 2026 - 2030 - 2035 |
| Market Representation | Revenue in USD Billion and CAGR from 2026 to 2035 |
| Segments Covered | Technology, Product Type, Application, End-User |
| Regional Scope | Asia-Pacific, Western Europe, Eastern Europe, North America, South America, Middle East, Africa |
| Country Scope | The countries covered in the report are Australia, Brazil, China, France, Germany, India, Indonesia, Japan, Taiwan, Russia, South Korea, UK, USA, Canada, Italy, Spain. |
| Key Companies Profiled | 3M Company, Merck KGaA, Lonza Group AG, SGS SA, Kikkoman Biochemifa, Charles River Laboratories, BioMérieux, Hach Company, Neogen Corporation, Charm Sciences, Promega Corporation, LuminUltra Technologies, Sopex d.o.o., Nelson Laboratories LLC, Hygiena LLC, PCE Instruments, Berthold Technologies, BioThema AB, Pharmetric Laboratory, Biotactical B.V., Promicol BV. |
| Customization Scope | Request for Customization |
| Pricing And Purchase Options | Explore Purchase Options |
