
Foot Drop Treatment Devices Market Report 2026
Global Outlook – By Product Type (Electrical Stimulator, Braces Or Splints), By Application (Neuropathy, Muscle Disorders, Brain And Spinal Disorders, Other Applications), By End User (Hospitals, Ambulatory Surgical Centers, Clinics, Homecare Settings) – Market Size, Trends, Strategies, and Forecast to 2035
Foot Drop Treatment Devices Market Overview
• Foot Drop Treatment Devices market size has reached to $2.45 billion in 2025 • Expected to grow to $3.59 billion in 2030 at a compound annual growth rate (CAGR) of 7.9% • Growth Driver: Rising Neurological Disorders Driving Demand For Advanced Treatment Solutions • Market Trend: CIONIC Expands Neural Sleeve Capabilities to Enhance Mobility and Daily Function for Patients with Disabilities • North America was the largest region in 2025 and Asia-Pacific is the fastest growing region.What Is Covered Under Foot Drop Treatment Devices Market?
Foot drop treatment devices are specialized medical aids designed to assist individuals with foot drop, a condition characterized by difficulty in lifting the front part of the foot, often due to neurological or muscular disorders. These devices work by supporting the foot in a raised position, preventing it from dragging or slapping during walking. Foot drop treatment devices aim to improve mobility, reduce the risk of falls, and enhance the quality of life for individuals with this condition. The main product types of foot drop treatment devices include electrical stimulators and braces or splints. An electrical stimulator is a device that uses electrical impulses to stimulate nerves or muscles. It is applied for neuropathy, muscle disorders, brain and spinal disorders, and others and is used by hospitals, ambulatory surgical centers, clinics, and home care settings.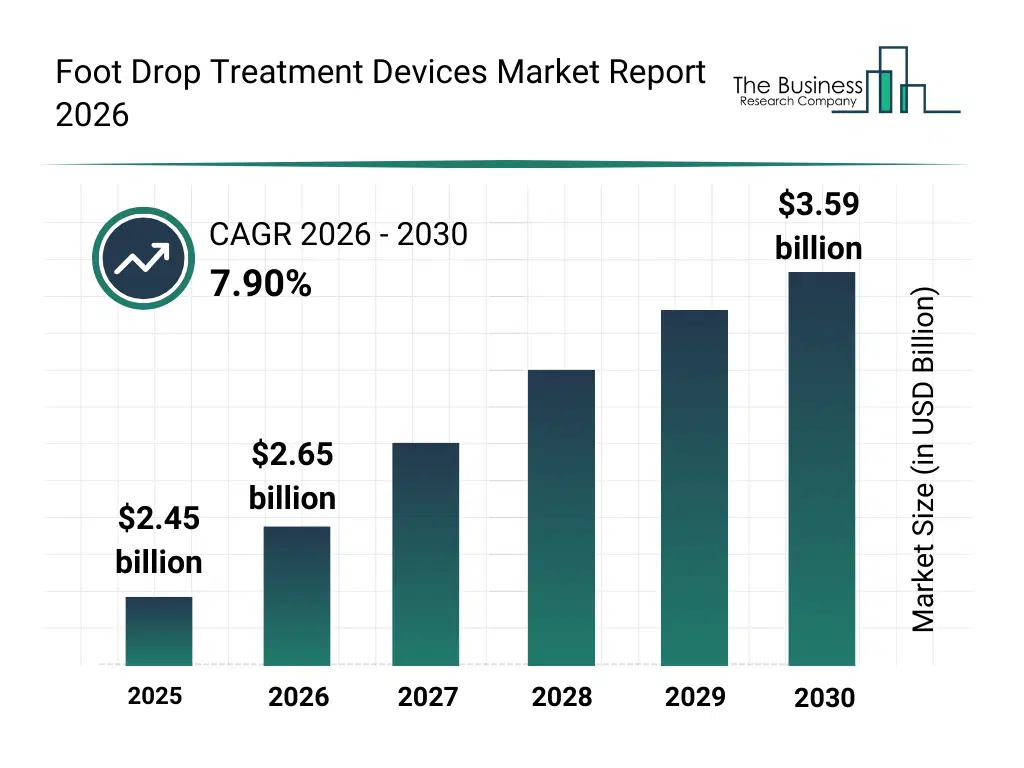
What Is The Foot Drop Treatment Devices Market Size and Share 2026?
The foot drop treatment devices market size has grown strongly in recent years. It will grow from $2.45 billion in 2025 to $2.65 billion in 2026 at a compound annual growth rate (CAGR) of 8.1%. The growth in the historic period can be attributed to increasing incidence of neurological disorders, rising cases of stroke-related mobility impairment, expansion of rehabilitation centers, growing availability of orthotic devices, increased clinical awareness of foot drop management.What Is The Foot Drop Treatment Devices Market Growth Forecast?
The foot drop treatment devices market size is expected to see strong growth in the next few years. It will grow to $3.59 billion in 2030 at a compound annual growth rate (CAGR) of 7.9%. The growth in the forecast period can be attributed to increasing adoption of advanced neurorehabilitation technologies, rising demand for homecare mobility aids, expansion of personalized orthotic solutions, growing focus on improving quality of life, technological advancements in wearable medical devices. Major trends in the forecast period include increasing adoption of functional electrical stimulation devices, rising demand for custom-fitted orthotic braces, growing use of lightweight and flexible materials, expansion of home-based rehabilitation solutions, enhanced focus on patient mobility improvement.Global Foot Drop Treatment Devices Market Segmentation
1) By Product Type: Electrical Stimulator, Braces Or Splints 2) By Application: Neuropathy, Muscle Disorders, Brain And Spinal Disorders, Other Applications 3) By End User: Hospitals, Ambulatory Surgical Centers, Clinics, Homecare Settings Subsegments: 1) By Electrical Stimulator: Functional Electrical Stimulation (FES) Devices, Neuromuscular Electrical Stimulation (NMES) Devices, Transcutaneous Electrical Nerve Stimulation (TENS) Devices, Others 2) By Braces Or Splints: Ankle-Foot Orthoses (AFO), Dynamic Ankle-Foot Orthoses (DAFO), Hinged Ankle Braces, Custom-Made Braces, OthersWhat Is The Driver Of The Foot Drop Treatment Devices Market?
The increasing incidence of neurological disorders and injuries is expected to propel the growth of the foot drop treatment devices market going forward. Neurological disorders and injuries refer to conditions that affect the brain, spinal cord, and nerves, leading to dysfunction in motor skills, sensation, cognition, or behavior due to damage, disease, or degeneration of the nervous system. The rising prevalence of neurological disorders and injuries is driven by factors such as an aging global population, increased awareness and diagnosis, lifestyle factors, environmental influences, and advancements in medical technology that allow for better detection and treatment. Foot drop treatment devices are essential for individuals with neurological disorders and injuries, as these conditions often affect the ability to lift the foot, causing difficulty walking and a higher risk of falls. These devices offer vital support and stability by assisting in lifting the foot, thereby improving mobility and enhancing the quality of life for those impacted by these conditions. For instance, in October 2023, the World Federation of Neurology, a UK-based association of national neurological societies, reported that more than 40% of the global population is currently affected by some form of neurological condition. This burden is expected to nearly double by 2050. Therefore, the increasing incidence of neurological disorders and injuries is driving the growth of the foot drop treatment devices industry.Key Players In The Global Foot Drop Treatment Devices Market
Major companies operating in the foot drop treatment devices market are Cardinal Health Inc., Stryker Corporation, Henry Schein Inc., Smith & Nephew LLC, Ottobock, Hanger Inc., Ossur, Bauerfeind USA Inc., Breg Inc., B. Braun Melsungen AG, Trulife Ltd., Orthomerica Products Inc., Bioness Inc., Evolution Health Care Pvt. Ltd., Aspen Medical Products, Allard Inc., Footmaxx, ProCare, DJO Global Inc., ALIMCO, Saebo Inc., Axio Bionics LLC, Thrive Orthopedics, Turbomed Orthotics, Accord Medical Products Private LimitedGlobal Foot Drop Treatment Devices Market Trends and Insights
Major companies operating in the foot drop treatment devices market are focusing on technological innovation, such as software-powered, wearable bionic clothing that integrates functional electrical stimulation with real-time mobility tracking, to improve walking, strength, and independence for individuals with neuromuscular impairments. Software-Powered Wearable Bionic Devices enable personalized functional training, comprehensive progress monitoring, and integration into activities of daily living, helping patients regain mobility, climb stairs, walk, and perform essential daily tasks more independently. For instance, in May 2024, CIONIC, a U.S.-based neurotechnology company, unveiled updates to its FDA-cleared Cionic Neural Sleeve, introducing new functional exercises and detailed activity tracking reports for patients with multiple sclerosis, strokes, spinal cord injuries, and other upper motor neuron diagnoses. The lightweight, durable leg sleeve combines gait lab diagnostics with functional electrical stimulation and is smartphone-operated, allowing software-driven updates to be deployed to all users simultaneously, empowering patients to enhance their functional mobility and daily independence.What Are Latest Mergers And Acquisitions In The Foot Drop Treatment Devices Market?
In January 2024, OrthoPediatrics, a US-based medical device company, acquired Boston Orthotics & Prosthetics for a deal of $22 million. This acquisition is expected to significantly bolster OrthoPediatrics' Specialty Bracing division, which focuses on non-surgical treatment options for children with orthopedic conditions. Boston Orthotics & Prosthetics is a US-based company that manufactures devices for the treatment of foot drop, specifically through their production of ankle foot orthotics.Regional Outlook
North America was the largest region in the foot drop treatment devices market in 2025. Asia-Pacific is expected to be the fastest-growing region in the forecast period. The regions covered in this market report are Asia-Pacific, South East Asia, Western Europe, Eastern Europe, North America, South America, Middle East, Africa. The countries covered in this market report are Australia, Brazil, China, France, Germany, India, Indonesia, Japan, Taiwan, Russia, South Korea, UK, USA, Canada, Italy, SpainWhat Defines the Foot Drop Treatment Devices Market?
The foot drop treatment devices market consists of sales of ankle-foot orthoses, toe lift assist devices, exoskeletons for lower limb support, and foot drop shoe inserts. Values in this market are ‘factory gate’ values, that is the value of goods sold by the manufacturers or creators of the goods, whether to other entities (including downstream manufacturers, wholesalers, distributors and retailers) or directly to end customers. The value of goods in this market includes related services sold by the creators of the goods.How is Market Value Defined and Measured?
The market value is defined as the revenues that enterprises gain from the sale of goods and/or services within the specified market and geography through sales, grants, or donations in terms of the currency (in USD unless otherwise specified). The revenues for a specified geography are consumption values that are revenues generated by organizations in the specified geography within the market, irrespective of where they are produced. It does not include revenues from resales along the supply chain, either further along the supply chain or as part of other products.Foot Drop Treatment Devices Market Report Forecast Analysis
| Report Attribute | Details |
|---|---|
| Market Size Value In 2026 | $2.65 billion |
| Revenue Forecast In 2035 | $3.59 billion |
| Growth Rate | CAGR of 8.1% from 2026 to 2035 |
| Base Year For Estimation | 2025 |
| Actual Estimates/Historical Data | 2020-2025 |
| Forecast Period | 2026 - 2030 - 2035 |
| Market Representation | Revenue in USD Billion and CAGR from 2026 to 2035 |
| Segments Covered | Product Type, Application, End User |
| Regional Scope | Asia-Pacific, Western Europe, Eastern Europe, North America, South America, Middle East, Africa |
| Country Scope | The countries covered in the report are Australia, Brazil, China, France, Germany, India, ... |
| Key Companies Profiled | Cardinal Health Inc., Stryker Corporation, Henry Schein Inc., Smith & Nephew LLC, Ottobock, Hanger Inc., Ossur, Bauerfeind USA Inc., Breg Inc., B. Braun Melsungen AG, Trulife Ltd., Orthomerica Products Inc., Bioness Inc., Evolution Health Care Pvt. Ltd., Aspen Medical Products, Allard Inc., Footmaxx, ProCare, DJO Global Inc., ALIMCO, Saebo Inc., Axio Bionics LLC, Thrive Orthopedics, Turbomed Orthotics, Accord Medical Products Private Limited |
| Customization Scope | Request for Customization |
| Pricing And Purchase Options | Explore Purchase Options |
