
Gastric Cancer Diagnostic Procedure Market Report 2026
Global Outlook – By Procedure Type (Endoscopic Procedure, Biopsy, Tissue Tests, Lab Tests, In-Vitro Diagnostic Tests, Imaging Tests, Molecular Diagnostics, Multiplexing Molecular Diagnostics, Immunoassays, Other Procedure Types ), By Symptom Type (Symptomatic, Asymptomatic), By Offering Type (Instruments, Reagents And Consumables, Services), By Body Fluid Type (Blood, Urine, Saliva, Stomach Juice, Tissue, Other Body Fluid), By Disease Indication (Early Gastric Cancer, Advanced Gastric Cancer) - Market Size, Trends, And Global Forecast 2026-2035
Gastric Cancer Diagnostic Procedure Market Overview
• Gastric Cancer Diagnostic Procedure market size has reached to $1.54 billion in 2025 • Expected to grow to $2.23 billion in 2030 at a compound annual growth rate (CAGR) of 7.7% • Growth Driver: Impact Of Smoking Epidemic On Gastric Cancer Diagnostic Procedures Market • Market Trend: Innovative Advances In Gastric Cancer Diagnostic Technologies Through The Role Of Liquid Biopsy Assays • North America was the largest region in 2025.What Is Covered Under Gastric Cancer Diagnostic Procedure Market?
Gastric cancer diagnostic procedure refers to a technique of identification and medication to treat cancer cells that grow in the stomach. It is used to diagnose stomach cancer. The main procedures of gastric cancer diagnostic procedure are endoscopic procedure, biopsy, tissue tests, lab tests, in-vitro diagnostic tests, imaging tests, molecular diagnostics, multiplexing molecular diagnostics, immunoassays, and others. Endoscopic Procedure refers to a process of observing an internal organ, such as the gastrointestinal tract, by inserting a long, thin tube straight into the body. The endoscopy procedure used in gastric cancer for diagnosing stomach cancer. It is suitable for symptoms such as symptomatic and asymptomatic and offers solutions such as instruments, reagents and consumables, and services. The body fluids that are required for the procedure include blood, urine, saliva, stomach juice, tissue, others to detect and treat early gastric cancer and advanced gastric cancer.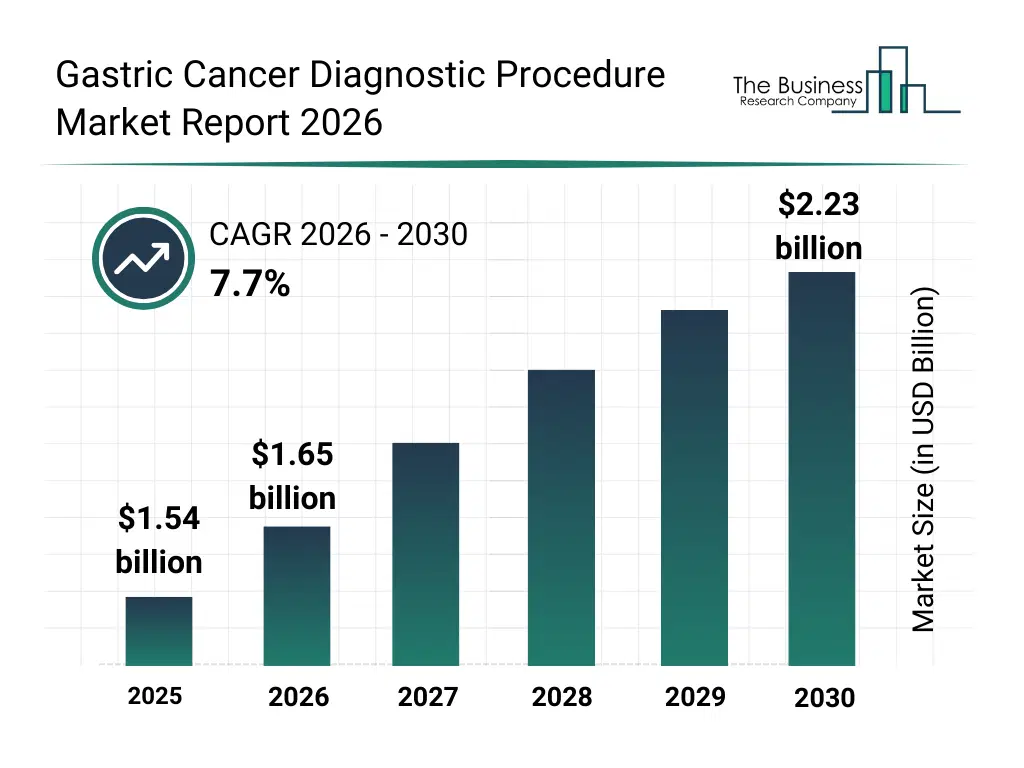
What Is The Gastric Cancer Diagnostic Procedure Market Size and Share 2026?
The gastric cancer diagnostic procedure market size has grown strongly in recent years. It will grow from $1.54 billion in 2025 to $1.65 billion in 2026 at a compound annual growth rate (CAGR) of 7.4%. The growth in the historic period can be attributed to use of endoscopy in gastric diagnosis, adoption of biopsy procedures, availability of imaging tests, hospital-based diagnostic infrastructure, development of pathology labs.What Is The Gastric Cancer Diagnostic Procedure Market Growth Forecast?
The gastric cancer diagnostic procedure market size is expected to see strong growth in the next few years. It will grow to $2.23 billion in 2030 at a compound annual growth rate (CAGR) of 7.7%. The growth in the forecast period can be attributed to growth in liquid biopsy techniques, expansion of molecular diagnostics, demand for early detection, automation in diagnostic workflows, AI-enabled imaging analysis. Major trends in the forecast period include growing use of endoscopic diagnostic techniques, rising adoption of molecular and genetic testing, increased demand for early-stage cancer detection, expansion of non-invasive diagnostic procedures, integration of advanced imaging technologies.Global Gastric Cancer Diagnostic Procedure Market Segmentation
1) By Procedure Type: Endoscopic Procedure, Biopsy, Tissue Tests, Lab Tests, In-Vitro Diagnostic Tests, Imaging Tests, Molecular Diagnostics, Multiplexing Molecular Diagnostics, Immunoassays, Other Procedure Types 2) By Symptom Type: Symptomatic, Asymptomatic 3) By Offering Type: Instruments, Reagents And Consumables, Services 4) By Body Fluid Type: Blood, Urine, Saliva, Stomach Juice, Tissue, Other Body Fluid 5) By Disease Indication: Early Gastric Cancer, Advanced Gastric Cancer Subsegments: 1) By Endoscopic Procedure: Upper Endoscopy (EGD), Endoscopic Ultrasound (EUS) 2) By Biopsy: Endoscopic Biopsy, Fine Needle Aspiration (FNA) Biopsy 3) By Tissue Tests: Histopathology, Immunohistochemistry 4) By Lab Tests: Blood Tests, Tumor Markers 5) By In-Vitro Diagnostic Tests: Genetic Testing, Molecular Profiling 6) By Imaging Tests: Computed Tomography (CT) Scan, Magnetic Resonance Imaging (MRI), Positron Emission Tomography (PET) Scan 7) By Molecular Diagnostics: PCR Testing, Next-Generation Sequencing (NGS) 8) By Multiplexing Molecular Diagnostics: Multiplex PCR, Microarray Analysis 9) By Immunoassays: Enzyme-Linked Immunosorbent Assay (ELISA), Radioimmunoassay (RIA) 10) By Other Procedure Types: Breath Tests, Liquid BiopsiesWhat Is The Driver Of The Gastric Cancer Diagnostic Procedure Market?
The rising surge in smoking is expected to propel the growth of the gastric cancer diagnostic procedure market going forward. Smoking refers to the act of inhaling and exhaling the smoke produced by burning tobacco or other substances. Gastric cancer diagnostic procedures can be crucial for individuals with a history of smoking or those at risk of smoking-related cancers for early detection and identification of precancerous lesions, help in developing personalized and tailored treatment plans, facilitate communication and coordination, and can provide a comprehensive assessment of the gastric mucosa and surrounding tissues. For instance, in July 2023, according to a report shared by the World Health Organization, a Switzerland-based intergovernmental organization, more than 8 million people are killed by tobacco each year, including 1.3 million non-smokers who inhale second-hand smoke. Therefore, the rising surge in smoking is driving the growth of the gastric cancer diagnostic procedure industry.Key Players In The Global Gastric Cancer Diagnostic Procedure Market
Major companies operating in the gastric cancer diagnostic procedure market are Thermo Fisher Scientific Inc., Agilent Technologies Inc., Myriad Genetics Inc., Foundation Medicine Inc., Biocept Inc., Illumina Inc., Roche Diagnostics, QIAGEN N V, Bio‑Rad Laboratories Inc., Siemens Healthineers, Danaher Corporation, Becton Dickinson, bioMérieux SA, PerkinElmer Inc., NeoGenomics Laboratories, Guardant Health Inc., Natera Inc., Caris Molecular Diagnostics, Adaptive Biotechnologies Corp, 10x Genomics IncGlobal Gastric Cancer Diagnostic Procedure Market Trends and Insights
Major companies operating in the gastric cancer diagnostic procedure market are focusing on technological advancements, such as dual cell-free and exosomal miRNA liquid biopsy assays, to gain a competitive advantage. These assays measure small noncoding RNAs released both freely in the bloodstream and packaged within tumor-derived exosomes, enabling highly sensitive and specific detection of gastric cancer from a simple blood sample. For instance, in December 2024, Cizzle Bio, a U.S.-based biotechnology company, introduced its DEX G2 blood test. The test combines cell-free and exosomal miRNAs to achieve approximately 95% sensitivity and 97% specificity and requires only a standard blood draw rather than invasive endoscopy. This innovation promises earlier diagnosis and broader screening reach, though widespread adoption may be constrained by regulatory approval timelines and the necessity for real-world validation.What Are Latest Mergers And Acquisitions In The Gastric Cancer Diagnostic Procedure Market?
In March 2023, Pfizer, a US-based pharmaceutical corporation, acquired Seagen Inc. for $43 billion. This acquisition helps Pfizer enhance its position as a leading player in the oncology market by integrating Seagen's pioneering antibody-drug conjugate (ADC) technology and expanding its portfolio of innovative cancer therapies. Seagen Inc., is a US-based biotechnology company that specializes in discovering, developing, and commercializing transformative cancer medicines.Regional Outlook
North America was the largest region in the gastric cancer diagnostics procedure market in 2025. The regions covered in this market report are Asia-Pacific, South East Asia, Western Europe, Eastern Europe, North America, South America, Middle East, Africa. The countries covered in this market report are Australia, Brazil, China, France, Germany, India, Indonesia, Japan, Taiwan, Russia, South Korea, UK, USA, Canada, Italy, Spain.What Defines the Gastric Cancer Diagnostic Procedure Market?
The gastric cancer diagnostics procedure market includes revenues earned by entities that provide diagnostic procedures such as surgery, chemotherapy, and radiation therapy. The market value includes the value of related goods sold by the service provider or included within the service offering. Only goods and services traded between entities or sold to end consumers are included.How is Market Value Defined and Measured?
The market value is defined as the revenues that enterprises gain from the sale of goods and/or services within the specified market and geography through sales, grants, or donations in terms of the currency (in USD unless otherwise specified). The revenues for a specified geography are consumption values that are revenues generated by organizations in the specified geography within the market, irrespective of where they are produced. It does not include revenues from resales along the supply chain, either further along the supply chain or as part of other products.What Key Data and Analysis Are Included in the Gastric Cancer Diagnostic Procedure Market Report 2026?
The gastric cancer diagnostic procedure market research report is one of a series of new reports from The Business Research Company that provides market statistics, including industry global market size, regional shares, competitors with the market share, detailed market segments, market trends and opportunities, and any further data you may need to thrive in the gastric cancer diagnostic procedure industry. The market research report delivers a complete perspective of everything you need, with an in-depth analysis of the current and future state of the industry.Gastric Cancer Diagnostic Procedure Market Report Forecast Analysis
| Report Attribute | Details |
|---|---|
| Market Size Value In 2026 | $1.65 billion |
| Revenue Forecast In 2035 | $2.23 billion |
| Growth Rate | CAGR of 7.4% from 2026 to 2035 |
| Base Year For Estimation | 2025 |
| Actual Estimates/Historical Data | 2020-2025 |
| Forecast Period | 2026 - 2030 - 2035 |
| Market Representation | Revenue in USD Billion and CAGR from 2026 to 2035 |
| Segments Covered | Procedure Type, Symptom Type, Offering Type, Body Fluid Type, Disease Indication |
| Regional Scope | Asia-Pacific, Western Europe, Eastern Europe, North America, South America, Middle East, Africa |
| Country Scope | The countries covered in the report are Australia, Brazil, China, France, Germany, India, ... |
| Key Companies Profiled | Thermo Fisher Scientific Inc., Agilent Technologies Inc., Myriad Genetics Inc., Foundation Medicine Inc., Biocept Inc., Illumina Inc., Roche Diagnostics, QIAGEN N V, Bio‑Rad Laboratories Inc., Siemens Healthineers, Danaher Corporation, Becton Dickinson, bioMérieux SA, PerkinElmer Inc., NeoGenomics Laboratories, Guardant Health Inc., Natera Inc., Caris Molecular Diagnostics, Adaptive Biotechnologies Corp, 10x Genomics Inc |
| Customization Scope | Request for Customization |
| Pricing And Purchase Options | Explore Purchase Options |
