
Generic Pharmaceuticals Contract Manufacturing Market Report 2026
Global Outlook – By Drug Type (Branded Generics, Unbranded Generics), By Product Type (Pharmaceutical Active Pharmaceutical Ingredients, Formulations, Capsules, Tablets, Injectables), By Route Of Administration (Oral, Parenteral, Topical, Other Routes Of Administration), By Application (Oncology, Immunology, Antidiabetic, Neurology, Anticoagulants, Cardiovascular, Respiratory, Pain, Human Immunodeficiency Virus Antivirals), By End-User Industry (Pharmaceutical Companies, Contract Research Organizations, Biotechnology Companies, Generic Drug Manufacturers) – Market Size, Trends, Strategies, and Forecast to 2035
Generic Pharmaceuticals Contract Manufacturing Market Overview
• Generic Pharmaceuticals Contract Manufacturing market size has reached to $66.82 billion in 2025 • Expected to grow to $93.56 billion in 2030 at a compound annual growth rate (CAGR) of 6.9% • Growth Driver: Rising Demand For Biosimilars Driving The Market Growth Due To Cost-Effective Treatment Alternatives • Market Trend: Lupin Enters CDMO Market With Strategic Expansion Initiative • North America was the largest region in 2025 and Asia-Pacific is the fastest growing region.What Is Covered Under Generic Pharmaceuticals Contract Manufacturing Market?
Generic pharmaceuticals contract manufacturing refers to the outsourcing of production processes for generic drugs to third-party manufacturers. These manufacturers provide services such as formulation development, active pharmaceutical ingredient production, tablet or injectable manufacturing, packaging, and regulatory support, helping pharmaceutical companies reduce costs and focus on core competencies. The main drug types of generic pharmaceuticals contract manufacturing are branded generics and unbranded generics. Branded generics refer to off-patent drugs that are marketed under a proprietary brand name by a pharmaceutical company, even though they contain the same active ingredients as the original branded product. The different product types include pharmaceutical active pharmaceutical ingredients, formulations, capsules, tablets, and injectables. These are administered through various routes of administration, such as oral, parenteral, topical, and others. These are used for various applications, including oncology, immunology, antidiabetics, neurology, anticoagulants, cardiovascular, respiratory, pain, and human immunodeficiency virus antivirals, and cater to various end-user industries, including pharmaceutical companies, contract research organizations, biotechnology companies, and generic drug manufacturers.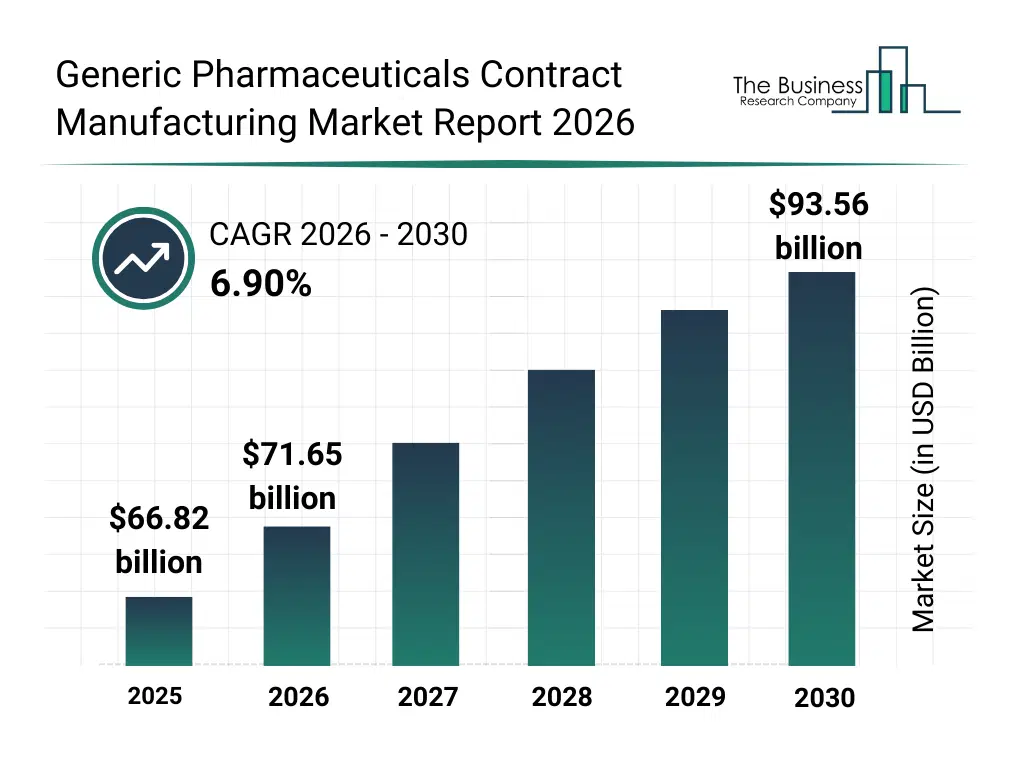
What Is The Generic Pharmaceuticals Contract Manufacturing Market Size and Share 2026?
The generic pharmaceuticals contract manufacturing market size has grown strongly in recent years. It will grow from $66.82 billion in 2025 to $71.65 billion in 2026 at a compound annual growth rate (CAGR) of 7.2%. The growth in the historic period can be attributed to limited contract manufacturing options, reliance on in-house generic drug production, rising demand for off-patent apis, increasing generic drug approvals, cost pressures on pharmaceutical companies.What Is The Generic Pharmaceuticals Contract Manufacturing Market Growth Forecast?
The generic pharmaceuticals contract manufacturing market size is expected to see strong growth in the next few years. It will grow to $93.56 billion in 2030 at a compound annual growth rate (CAGR) of 6.9%. The growth in the forecast period can be attributed to growth in global generic drug demand, expansion of contract manufacturing organizations (cmos), adoption of advanced formulation technologies, rising focus on biologics and complex generics, increased cross-border regulatory collaborations. Major trends in the forecast period include rising outsourcing of generic drug manufacturing, focus on cost-efficient production and packaging, expansion of injectable and tablet manufacturing services, increased regulatory support and compliance services, growth in licensing and technology transfer for generic products.Global Generic Pharmaceuticals Contract Manufacturing Market Segmentation
1) By Drug Type: Branded Generics, Unbranded Generics 2) By Product Type: Pharmaceutical Active Pharmaceutical Ingredients, Formulations, Capsules, Tablets, Injectables 3) By Route Of Administration: Oral, Parenteral, Topical, Other Routes Of Administration 4) By Application: Oncology, Immunology, Antidiabetic, Neurology, Anticoagulants, Cardiovascular, Respiratory, Pain, Human Immunodeficiency Virus Antivirals 5) By End-User Industry: Pharmaceutical Companies, Contract Research Organizations, Biotechnology Companies, Generic Drug Manufacturers Subsegments: 1) By Branded Generics: Contract Development of Branded Formulations, Branded Generic Tablet Manufacturing, Branded Injectable Manufacturing, Packaging and Labeling for Branded Generics, Regulatory Support For Branded Generics 2) By Unbranded Generics: Bulk Manufacturing of Off-Patent Active Pharmaceutical Ingredients, Generic Tablet Or Capsule Production, Parenteral Or Injectable Generic Manufacturing, Cost-Efficient Packaging for Unbranded Products, Licensing And Tech Transfer for Generic ProductsWhat Is The Driver Of The Generic Pharmaceuticals Contract Manufacturing Market?
The rising demand for biosimilars is expected to propel the growth of the generic pharmaceuticals contract manufacturing market going forward. Biosimilars are biologic medical products that are highly similar to already approved reference biologics, with no clinically meaningful differences in safety, purity, or potency. The rising demand for biosimilars is primarily due to their potential to provide cost-effective alternatives to expensive biologic drugs, making advanced treatments more accessible to patients and healthcare systems. Generic pharmaceuticals contract manufacturing supports biosimilars by providing cost-effective, large-scale production capabilities and regulatory expertise needed to accelerate market entry and ensure consistent quality. For instance, in January 2023, according to the Cardinal Health Inc., a US-based healthcare company, as of January 2022, the U.S. had 33 biosimilar products approved by the FDA, with 21 of them available on the market and by 2023, the number of FDA-approved biosimilars had risen to 40, with 25 commercially available. Therefore, the rising demand for biosimilars is driving the growth of the generic pharmaceuticals contract manufacturing industry.Key Players In The Global Generic Pharmaceuticals Contract Manufacturing Market
Major companies operating in the generic pharmaceuticals contract manufacturing market are WuXi AppTec Co. Ltd., Recipharm AB, Siegfried Holding AG, Curia Global Inc., Piramal Pharma Solutions, Laurus Labs Limited., Cambrex Corp., Alcami Corp. Inc, Neuland Laboratories Limited.Global Generic Pharmaceuticals Contract Manufacturing Market Trends and Insights
Major companies operating in the generic pharmaceutical contract manufacturing market are expanding their service portfolios by establishing dedicated subsidiaries to meet the growing demand for outsourced development and manufacturing. This strategic move of establishing dedicated subsidiaries enables organizations to streamline the production of active pharmaceutical ingredients (APIs), ensuring compliance, scalability, and operational efficiency across the drug development lifecycle. For instance, in July 2025, Lupin, an India-based generic pharmaceutical company, launched its new subsidiary, Lupin Manufacturing Solutions (LMS), to enter the contract development and manufacturing organization (CDMO) market. It is a specialized division aimed at supporting the development, manufacturing, and sale of APIs for global pharmaceutical partners. With a leadership team experienced in biotech and pharma, the subsidiary is positioned to deliver high-quality contract services, helping pharmaceutical companies accelerate time-to-market while maintaining regulatory standards and cost efficiency.What Are Latest Mergers And Acquisitions In The Generic Pharmaceuticals Contract Manufacturing Market?
In April 2024, Bora Pharmaceuticals, a Taiwan-based contract development and manufacturing organization (CDMO), acquired Upsher-Smith Laboratories for $210 million. The acquisition brings additional capacity and expertise, strengthening Bora’s offering across the entire pharmaceutical value chain, from drug development to manufacturing, quality, packaging, and distribution. Upsher-Smith Laboratories is a US-based company that provides generic pharmaceutical contract manufacturing services.Regional Insights
North America was the largest region in the generic pharmaceuticals contract manufacturing market in 2025. Asia-Pacific is expected to be the fastest-growing region in the forecast period. The regions covered in this market report are Asia-Pacific, South East Asia, Western Europe, Eastern Europe, North America, South America, Middle East, Africa. The countries covered in this market report are Australia, Brazil, China, France, Germany, India, Indonesia, Japan, Taiwan, Russia, South Korea, UK, USA, Canada, Italy, Spain.What Defines the Generic Pharmaceuticals Contract Manufacturing Market?
The generic pharmaceuticals contract manufacturing market consists of sales of creams, syrups, sterile formulations, topical medications, and high-potency generic drugs. Values in this market are ‘factory gate’ values, that is, the value of goods sold by the manufacturers or creators of the goods, whether to other entities (including downstream manufacturers, wholesalers, distributors, and retailers) or directly to end customers. The value of goods in this market includes related services sold by the creators of the goods.How is Market Value Defined and Measured?
The market value is defined as the revenues that enterprises gain from the sale of goods and/or services within the specified market and geography through sales, grants, or donations in terms of the currency (in USD unless otherwise specified). The revenues for a specified geography are consumption values that are revenues generated by organizations in the specified geography within the market, irrespective of where they are produced. It does not include revenues from resales along the supply chain, either further along the supply chain or as part of other products.What Key Data and Analysis Are Included in the Generic Pharmaceuticals Contract Manufacturing Market Report 2026?
The generic pharmaceuticals contract manufacturing market research report is one of a series of new reports from The Business Research Company that provides market statistics, including industry global market size, regional shares, competitors with the market share, detailed market segments, market trends and opportunities, and any further data you may need to thrive in the generic pharmaceuticals contract manufacturing industry. The market research report delivers a complete perspective of everything you need, with an in-depth analysis of the current and future state of the industry.Generic Pharmaceuticals Contract Manufacturing Market Report Forecast Analysis
| Report Attribute | Details |
|---|---|
| Market Size Value In 2026 | $71.65 billion |
| Revenue Forecast In 2035 | $93.56 billion |
| Growth Rate | CAGR of 7.2% from 2026 to 2035 |
| Base Year For Estimation | 2025 |
| Actual Estimates/Historical Data | 2020-2025 |
| Forecast Period | 2026 - 2030 - 2035 |
| Market Representation | Revenue in USD Billion and CAGR from 2026 to 2035 |
| Segments Covered | Drug Type, Product Type, Route Of Administration, Application, End-User Industry |
| Regional Scope | Asia-Pacific, Western Europe, Eastern Europe, North America, South America, Middle East, Africa |
| Country Scope | The countries covered in the report are Australia, Brazil, China, France, Germany, India, ... |
| Key Companies Profiled | WuXi AppTec Co. Ltd., Recipharm AB, Siegfried Holding AG, Curia Global Inc., Piramal Pharma Solutions, Laurus Labs Limited., Cambrex Corp., Alcami Corp. Inc, Neuland Laboratories Limited. |
| Customization Scope | Request for Customization |
| Pricing And Purchase Options | Explore Purchase Options |
