
Infectious Disease In Vitro Diagnostics Market Report 2026
Global Outlook – By Product Type (Instruments, Reagents, Software And Services), By Technology (Molecular Diagnostics, Immunoassay, Microbiology), By Application (Methicillin-Resistant Staphylococcus Aureus (MRSA), Clostridium Difficile, Respiratory Virus, Tuberculosis (TB) And Drug-Resistant Tuberculosis (TB), Gonorrhea, Human Papillomavirus (HPV), Human Immunodeficiency Virus (HIV), Hepatitis C, Hepatitis B, Other Applications), By End-Use (Hospitals And Clinics, Diagnostic Laboratories, Other End-Users) – Market Size, Trends, Strategies, and Forecast to 2035
Infectious Disease In Vitro Diagnostics Market Overview
• Infectious Disease In Vitro Diagnostics market size has reached to $15.1 billion in 2025 • Expected to grow to $21.25 billion in 2030 at a compound annual growth rate (CAGR) of 7.1% • Growth Driver: Surge In Global Prevalence Of Infectious Diseases Fueled The Market Growth Due To Increased Demand For Effective Diagnostic Solutions • Market Trend: Developing Rapid Multi-Pathogen Detection Solutions To Enhance Timely Diagnosis And Expand Point-Of-Care Testing • North America was the largest region in 2025 and Asia-Pacific is the fastest growing region.What Is Covered Under Infectious Disease In Vitro Diagnostics Market?
Infectious disease in vitro diagnostics refers to laboratory-based tests performed on samples such as blood, urine, or swabs to detect, identify, and monitor infectious pathogens, including bacteria, viruses, fungi, and parasites. These diagnostics help to provide accurate and timely diagnosis, guide treatment decisions, and support disease surveillance and infection control measures. The main product types of infectious disease in vitro diagnostics are instruments, reagents, and software and services. Instruments refer to tools or devices used to measure and monitor environmental conditions such as air, water, soil, and noise. It utilizes various technologies such as molecular diagnostics, immunoassay, microbiology, hematology, urinalysis, and others. It finds application in methicillin-resistant Staphylococcus aureus (MRSA), Clostridium difficile, respiratory virus, tuberculosis (TB) and drug-resistant TB, gonorrhea, human papillomavirus (HPV), human immunodeficiency virus (HIV), hepatitis C, hepatitis B, and others, serving end-users such as hospitals and clinics, diagnostic laboratories, academic research institutes, and others.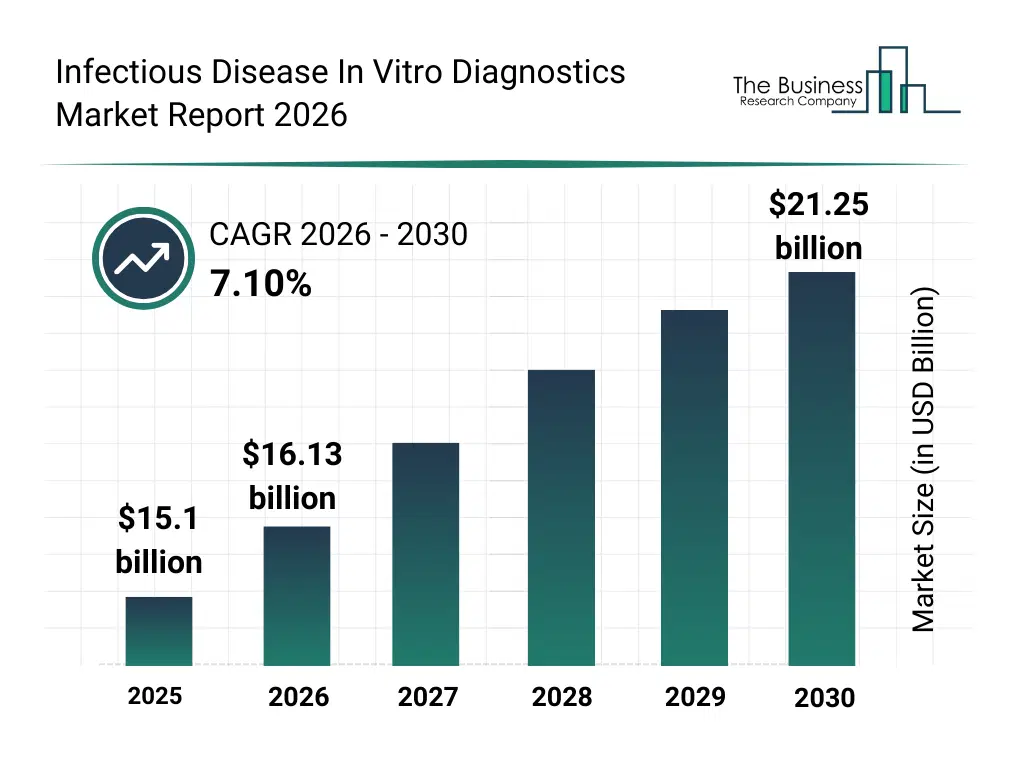
What Is The Infectious Disease In Vitro Diagnostics Market Size and Share 2026?
The infectious disease in vitro diagnostics market size has grown strongly in recent years. It will grow from $15.1 billion in 2025 to $16.13 billion in 2026 at a compound annual growth rate (CAGR) of 6.8%. The growth in the historic period can be attributed to global infectious disease burden, hospital laboratory expansion, demand for early diagnosis, advances in immunoassays, government disease control programs.What Is The Infectious Disease In Vitro Diagnostics Market Growth Forecast?
The infectious disease in vitro diagnostics market size is expected to see strong growth in the next few years. It will grow to $21.25 billion in 2030 at a compound annual growth rate (CAGR) of 7.1%. The growth in the forecast period can be attributed to pandemic preparedness initiatives, growth in molecular diagnostics, rising antimicrobial resistance, expansion of diagnostic labs, increased healthcare screening. Major trends in the forecast period include rapid molecular diagnostic adoption, point of care infectious testing, high sensitivity pathogen detection, multiplex diagnostic panels, increased surveillance testing.Global Infectious Disease In Vitro Diagnostics Market Segmentation
1) By Product Type: Instruments, Reagents, Software And Services 2) By Technology: Molecular Diagnostics, Immunoassay, Microbiology 3) By Application: Methicillin-Resistant Staphylococcus Aureus (MRSA), Clostridium Difficile, Respiratory Virus, Tuberculosis (TB) And Drug-Resistant Tuberculosis (TB), Gonorrhea, Human Papillomavirus (HPV), Human Immunodeficiency Virus (HIV), Hepatitis C, Hepatitis B, Other Applications 4) By End-Use: Hospitals And Clinics, Diagnostic Laboratories, Other End-Users Subsegments: 1) By Instrument: Polymerase Chain Reaction Machines, Immunoassay Analyzers, Microbiology Analyzers, Next Generation Sequencers 2) By Reagent: Molecular Reagents, Immunoassay Reagents, Microbiology Reagents, Enzymes And Buffers, Controls And Calibrators 3) By Software And Services: Data Management Software, Laboratory Information Systems, Cloud Based Solutions, Maintenance And Support Services, Consulting And Training ServicesWhat Is The Driver Of The Infectious Disease In Vitro Diagnostics Market?
The increased global prevalence of infectious diseases fueled the growth of the infectious disease in vitro diagnostics market during the historic period. Infectious diseases refer to the individuals affected by bacterial, viral, fungal, or parasitic infections worldwide at a specific point in time, indicating the overall disease burden within populations. Climate change has expanded the habitats of disease-carrying vectors such as mosquitoes, leading to a rise in the global prevalence of infectious diseases. The infectious disease in vitro diagnostics support this trend by providing precise and timely diagnostic tools that enable healthcare providers to identify pathogens, initiate appropriate treatment, and contain outbreaks more effectively. For instance, in October 2024, according to the World Health Organization, a Switzerland-based health authority, approximately 10.8 million people worldwide contracted tuberculosis, with 55% being men, 33% women, and 12% children and adolescents, and individuals living with human immunodeficiency virus (HIV) representing 6.1% of cases. Therefore, the increasing global prevalence of infectious diseases fueled the growth of the infectious disease in vitro diagnostics industry during the historic period.Key Players In The Global Infectious Disease In Vitro Diagnostics Market
Major companies operating in the infectious disease in vitro diagnostics market are F. Hoffmann-La Roche AG, Abbott Laboratories, Danaher Corporation, Thermo Fisher Scientific Inc., Siemens Healthcare AG, bioMérieux SA, QIAGEN N.V., Becton Dickinson and Company, Hologic Inc., DiaSorin S.p.A., Meridian Bioscience, Quidel Corporation, Revvity Inc., OraSure Technologies Inc., Transasia Bio-Medicals Ltd., Randox Laboratories, Luminex Corporation, Wako Pure Chemical Industries Ltd., Bio-Rad Laboratories Inc., Trivitron HealthcareGlobal Infectious Disease In Vitro Diagnostics Market Trends and Insights
Major companies operating in the infectious disease in vitro diagnostics market are focusing on developing innovative products, such as rapid detection of multiple gastrointestinal pathogens, to enable timely diagnosis, improve patient outcomes, and expand point-of-care testing capabilities. Rapid detection of multiple gastrointestinal pathogens refers to diagnostic tests that can quickly identify the presence of several disease-causing microorganisms in the gastrointestinal tract from a single sample. For instance, in June 2024, MP Biomedicals, a US-based provider of a diverse range of products, chemicals, and diagnostics, launched a range of immunochromatographic-based rapid diagnostic kits for gastrointestinal infectious diseases. These enable quick and accurate detection of Helicobacter pylori, Salmonella typhi, and Vibrio cholerae serogroups O1 and O139. These kits feature rapid, point-of-care qualitative detection, versatile sample use (stool, blood, plasma, serum, water, and food), and suitability for both clinical and research settings. Leveraging cutting-edge immunochromatographic technology, they support early diagnosis, monitoring of treatment effectiveness, and confirmation of pathogen eradication, representing a significant advancement in gastrointestinal health diagnostics by combining speed, precision, and broad pathogen coverage.What Are Latest Mergers And Acquisitions In The Infectious Disease In Vitro Diagnostics Market?
In January 2025, Fremman Capital, a UK-based private equity firm specializing in healthcare and life sciences investments, acquired a majority stake in DIESSE Diagnostica Senese for an undisclosed amount. With this acquisition, Fremman Capital aims to accelerate DIESSE’s international expansion and strengthen its position as a leader in the specialty in vitro diagnostics market by supporting innovation, broadening the product portfolio for autoimmune and infectious disease diagnostics, and enhancing production capacities at DIESSE’s advanced biotech campus, thereby driving long-term sustainable growth. DIESSE Diagnostica Senese is an Italy-based producer of innovative in vitro diagnostics and reagents focused on autoimmunity, infectious diseases, and hematological testing, serving clinical laboratories globally.Regional Outlook
North America was the largest region in the infectious disease in vitro diagnostics market in 2025. Asia-Pacific is expected to be the fastest-growing region in the forecast period. The regions covered in this market report are Asia-Pacific, South East Asia, Western Europe, Eastern Europe, North America, South America, Middle East, Africa. The countries covered in this market report are Australia, Brazil, China, France, Germany, India, Indonesia, Japan, Taiwan, Russia, South Korea, UK, USA, Canada, Italy, SpainWhat Defines the Infectious Disease In Vitro Diagnostics Market?
The infectious disease in vitro diagnostics market consists of revenues earned by entities by providing services such as serology testing, rapid diagnostic testing, disease surveillance and monitoring, sample collection and processing, and consulting and training services. The market value includes the value of related goods sold by the service provider or included within the service offering. The infectious disease in vitro diagnostics market also includes sales of rapid test kits, culture media, controls and calibrators, molecular sequencing kits, flow cytometry reagents, and point-of-care diagnostic devices. Values in this market are ‘factory gate’ values, that is the value of goods sold by the manufacturers or creators of the goods, whether to other entities (including downstream manufacturers, wholesalers, distributors and retailers) or directly to end customers. The value of goods in this market includes related services sold by the creators of the goods.How is Market Value Defined and Measured?
The market value is defined as the revenues that enterprises gain from the sale of goods and/or services within the specified market and geography through sales, grants, or donations in terms of the currency (in USD unless otherwise specified). The revenues for a specified geography are consumption values that are revenues generated by organizations in the specified geography within the market, irrespective of where they are produced. It does not include revenues from resales along the supply chain, either further along the supply chain or as part of other products.Infectious Disease In Vitro Diagnostics Market Report Forecast Analysis
| Report Attribute | Details |
|---|---|
| Market Size Value In 2026 | $16.13 billion |
| Revenue Forecast In 2035 | $21.25 billion |
| Growth Rate | CAGR of 6.8% from 2026 to 2035 |
| Base Year For Estimation | 2025 |
| Actual Estimates/Historical Data | 2020-2025 |
| Forecast Period | 2026 - 2030 - 2035 |
| Market Representation | Revenue in USD Billion and CAGR from 2026 to 2035 |
| Segments Covered | Product Type, Technology, Application, End-Use |
| Regional Scope | Asia-Pacific, Western Europe, Eastern Europe, North America, South America, Middle East, Africa |
| Country Scope | The countries covered in the report are Australia, Brazil, China, France, Germany, India, ... |
| Key Companies Profiled | F. Hoffmann-La Roche AG, Abbott Laboratories, Danaher Corporation, Thermo Fisher Scientific Inc., Siemens Healthcare AG, bioMérieux SA, QIAGEN N.V., Becton Dickinson and Company, Hologic Inc., DiaSorin S.p.A., Meridian Bioscience, Quidel Corporation, Revvity Inc., OraSure Technologies Inc., Transasia Bio-Medicals Ltd., Randox Laboratories, Luminex Corporation, Wako Pure Chemical Industries Ltd., Bio-Rad Laboratories Inc., Trivitron Healthcare |
| Customization Scope | Request for Customization |
| Pricing And Purchase Options | Explore Purchase Options |
