
Lyme Disease Vaccine Market Report 2026
Global Outlook – By Vaccine Type (Recombinant Vaccines, Subunit Vaccines, Live Attenuated Vaccines, Other Vaccine Types), By Technology (Lymphocytic Transformation Test, Urine Antigen Testing, Immunofluorescent Staining Nucleic Acid Test, Serological Test), By Age Group (Pediatric, Adult, Geriatric), By Distribution Channel (Hospitals And Clinics, Pharmacies, Government Supply, Other Distribution Channels), By End User (Hospitals, Clinical Laboratories, Other End Users) – Market Size, Trends, Strategies, and Forecast to 2035
Lyme Disease Vaccine Market Overview
• Lyme Disease Vaccine market size has reached to $1.12 billion in 2025 • Expected to grow to $1.57 billion in 2030 at a compound annual growth rate (CAGR) of 7% • Growth Driver: Increasing Prevalence Of Lyme Disease Fueling The Growth Of The Market Due To Climate-Driven Expansion Of Tick Habitats • Market Trend: Positive Phase 2 Data Highlights Strong Immune Response And Safety Of Booster Dose • North America was the largest region in 2025 and Asia-Pacific is the fastest growing region.What Is Covered Under Lyme Disease Vaccine Market?
Lyme disease vaccine refers to biological preparations designed to stimulate the immune system to recognize and fight Borrelia burgdorferi, the bacteria responsible for Lyme disease, thereby preventing infection following a tick bite. These vaccines work by triggering the production of specific antibodies that neutralize the bacteria, reducing the risk of disease transmission and associated complications, and serve as an important tool in controlling Lyme disease in endemic regions. The main vaccine types of Lyme disease vaccine are recombinant vaccines, subunit vaccines, live attenuated vaccines, and others. Recombinant vaccines refer to vaccines developed by using genetic engineering techniques to produce specific proteins (antigens) from Borrelia burgdorferi, the bacteria that cause Lyme disease. It utilizes various technologies such as lymphocytic transformation test, urine antigen testing, immunofluorescent staining nucleic acid test, and serological test, serving age groups such as pediatric, adult, and geriatric. It is distributed through various channels, including hospitals and clinics, pharmacies, government supply, and others, catering to end users such as hospitals, clinical laboratories, and others.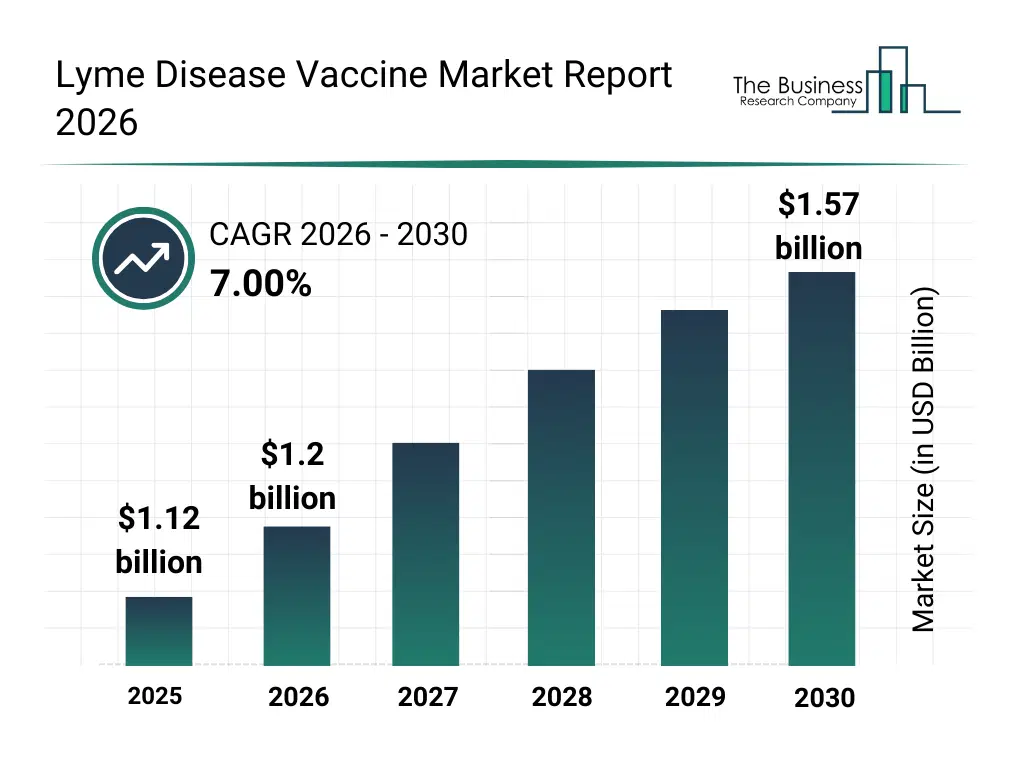
What Is The Lyme Disease Vaccine Market Size and Share 2026?
The lyme disease vaccine market size has grown strongly in recent years. It will grow from $1.12 billion in 2025 to $1.2 billion in 2026 at a compound annual growth rate (CAGR) of 7.3%. The growth in the historic period can be attributed to increasing incidence of lyme disease in endemic regions, historical limitations of antibiotic-only treatment approaches, early-stage vaccine research initiatives, growing awareness of tick-borne infections, public health surveillance improvements.What Is The Lyme Disease Vaccine Market Growth Forecast?
The lyme disease vaccine market size is expected to see strong growth in the next few years. It will grow to $1.57 billion in 2030 at a compound annual growth rate (CAGR) of 7.0%. The growth in the forecast period can be attributed to expansion of vaccination programs in endemic areas, increasing investment in infectious disease prevention, advancement of next-generation vaccine platforms, rising adoption of combination vaccines, growing global focus on zoonotic disease control. Major trends in the forecast period include advancement of recombinant vaccine technologies, increased focus on preventive immunization programs, expansion of clinical research for tick-borne diseases, growing emphasis on long-lasting immunity solutions, rising collaboration between public and private health organizations.Global Lyme Disease Vaccine Market Segmentation
1) By Vaccine Type: Recombinant Vaccines, Subunit Vaccines, Live Attenuated Vaccines, Other Vaccine Types 2) By Technology: Lymphocytic Transformation Test, Urine Antigen Testing, Immunofluorescent Staining Nucleic Acid Test, Serological Test 3) By Age Group: Pediatric, Adult, Geriatric 4) By Distribution Channel: Hospitals And Clinics, Pharmacies, Government Supply, Other Distribution Channels 5) By End User: Hospitals, Clinical Laboratories, Other End Users Subsegments: 1) By Recombinant Vaccines: Protein-Based Recombinant Vaccines, Vector-Based Recombinant Vaccines, Deoxyribonucleic Acid (DNA)-Based Recombinant Vaccines 2) By Subunit Vaccines: Protein Subunit Vaccines, Polysaccharide Subunit Vaccines, Conjugate Subunit Vaccines 3) By Live Attenuated Vaccines: Naturally Attenuated Vaccines, Genetically Attenuated Vaccines, Temperature-Sensitive Attenuated Vaccines 4) By Other Vaccine Types: Toxoid Vaccines, Inactivated Vaccines, Vectored VaccinesWhat Are The Drivers Of The Lyme Disease Vaccine Market?
The increasing prevalence of Lyme disease is expected to propel the growth of the Lyme disease vaccine market going forward. Lyme disease is a tick-borne infection caused by Borrelia bacteria, leading to fever, fatigue, rash, and potentially serious joint, heart, or nerve problems if untreated. The increasing prevalence of Lyme disease is due to climate change as warmer temperatures and milder winters expand tick habitats, increasing human exposure to infected ticks. Lyme disease vaccines aid in Lyme disease prevention by training the immune system to recognize and fight Borrelia bacteria, reducing the risk of infection. They simplify disease management by providing long-term protection, improving public health outcomes. For instance, in March 2025, according to the Centers for Disease Control and Prevention, a US-based federal health agency, in 2023, around 89,000 cases of Lyme disease were reported in the United States, while estimates using other methods suggest that approximately 476,000 people may be diagnosed and treated for Lyme disease each year. Therefore, the increasing Lyme disease prevalence is driving the growth of the Lyme disease vaccine industry. The rising healthcare expenditure is expected to propel the growth of the Lyme disease vaccine market going forward. Healthcare expenditure refers to the total amount of money spent on medical services, treatments, facilities, research, and public health initiatives to maintain or improve health outcomes. The increasing demand for healthcare expenditure is due to the rising prevalence of chronic diseases, which require long-term and costly treatments. Rising healthcare expenditure enhances Lyme disease vaccine development by providing increased funding for research and clinical trials, accelerating innovation and production. It supports advanced vaccine technologies, improving accessibility and effectiveness for at-risk populations. For instance, in May 2024, according to the Office for National Statistics, a UK-based government department, total healthcare expenditure saw a 5.6% increase in nominal terms in 2023, an increase of 0.9% growth compared to 2022. Therefore, the rising healthcare expenditure is driving the growth of the Lyme disease vaccine industry.Key Players In The Global Lyme Disease Vaccine Market
Major companies operating in the lyme disease vaccine market are Pfizer Inc., Merck & Co. Inc., Sanofi S.A., Boehringer Ingelheim GmbH, University of Pennsylvania, Elanco Animal Health Incorporated, Zoetis Inc., Emergent BioSolutions Inc., Bavarian Nordic A/S, Aegis Life Inc., Bioveta a.s., Dynavax Technologies Corporation, Valneva SE, MassBiologics of UMass Chan Medical School, Inovio Pharmaceuticals Inc., US Biologic Inc., Blue Lake Biotechnology Inc.Global Lyme Disease Vaccine Market Trends and Insights
Major companies operating in the Lyme disease vaccine market are focusing on advancing their late-stage product candidates, such as booster vaccines, to address unmet medical needs, enhance immune protection, and simplify vaccination regimens. Booster vaccines are additional doses given after the initial vaccination to restore or enhance long-term immunity. For instance, in September 2025, Valneva SE, a France-based biotechnology company, confirmed the positive immunogenicity and safety data from the ongoing Phase 2 study of Lyme disease vaccine candidate, VLA15. It is a multivalent vaccine that trains the immune system to produce antibodies against OspA proteins on Borrelia bacteria. When a tick bites, these antibodies block the bacteria inside the tick, preventing infection in humans. The strong immune response and favorable safety profile following a third booster dose were consistent with those reported after receiving previous annual booster doses, further demonstrating compatibility with the anticipated benefits of a yearly vaccination prior to each Lyme season. This booster strategy is intended to provide continued protection against Lyme disease, supporting the potential for an annual pre-exposure season booster to simplify protection for at-risk individuals.Regional Insights
North America was the largest region in the Lyme disease vaccine market in 2025. Asia-Pacific is expected to be the fastest-growing region in the forecast period. The regions covered in this market report are Asia-Pacific, South East Asia, Western Europe, Eastern Europe, North America, South America, Middle East, Africa. The countries covered in this market report are Australia, Brazil, China, France, Germany, India, Indonesia, Japan, Taiwan, Russia, South Korea, UK, USA, Canada, Italy, Spain.What Defines the Lyme Disease Vaccine Market?
The Lyme disease vaccine market consists of sales of pre-filled syringes, single or multi-dose vials, vaccine kits, storage and delivery equipment, and cold-chain packaging materials. Values in this market are ‘factory gate’ values, that is, the value of goods sold by the manufacturers or creators of the goods, whether to other entities (including downstream manufacturers, wholesalers, distributors, and retailers) or directly to end customers. The value of goods in this market includes related services sold by the creators of the goods.How is Market Value Defined and Measured?
The market value is defined as the revenues that enterprises gain from the sale of goods and/or services within the specified market and geography through sales, grants, or donations in terms of the currency (in USD unless otherwise specified). The revenues for a specified geography are consumption values that are revenues generated by organizations in the specified geography within the market, irrespective of where they are produced. It does not include revenues from resales along the supply chain, either further along the supply chain or as part of other products.What Key Data and Analysis Are Included in the Lyme Disease Vaccine Market Report 2026?
The lyme disease vaccine market research report is one of a series of new reports from The Business Research Company that provides market statistics, including industry global market size, regional shares, competitors with the market share, detailed market segments, market trends and opportunities, and any further data you may need to thrive in the lyme disease vaccine industry. The market research report delivers a complete perspective of everything you need, with an in-depth analysis of the current and future state of the industry.Lyme Disease Vaccine Market Report Forecast Analysis
| Report Attribute | Details |
|---|---|
| Market Size Value In 2026 | $1.2 billion |
| Revenue Forecast In 2035 | $1.57 billion |
| Growth Rate | CAGR of 7.3% from 2026 to 2035 |
| Base Year For Estimation | 2025 |
| Actual Estimates/Historical Data | 2020-2025 |
| Forecast Period | 2026 - 2030 - 2035 |
| Market Representation | Revenue in USD Billion and CAGR from 2026 to 2035 |
| Segments Covered | Vaccine Type, Technology, Age Group, Distribution Channel, End User |
| Regional Scope | Asia-Pacific, Western Europe, Eastern Europe, North America, South America, Middle East, Africa |
| Country Scope | The countries covered in the report are Australia, Brazil, China, France, Germany, India, ... |
| Key Companies Profiled | Pfizer Inc., Merck & Co. Inc., Sanofi S.A., Boehringer Ingelheim GmbH, University of Pennsylvania, Elanco Animal Health Incorporated, Zoetis Inc., Emergent BioSolutions Inc., Bavarian Nordic A/S, Aegis Life Inc., Bioveta a.s., Dynavax Technologies Corporation, Valneva SE, MassBiologics of UMass Chan Medical School, Inovio Pharmaceuticals Inc., US Biologic Inc., Blue Lake Biotechnology Inc. |
| Customization Scope | Request for Customization |
| Pricing And Purchase Options | Explore Purchase Options |
