
GMP Cell Therapy Consumables Market Report 2026
Global Outlook – By Product (Kits, Reagents Or Molecular Biology Reagents, Growth Factors Or Cytokines And Interleukins, Other Products), By Cell Therapy (NK Cell Therapy, Stem Cell Therapy, T-Cell Therapy), By Process (Cell Collection And Characterization Or Sorting And Separation, Cell Culture And Expansion Or Preparation, Cryopreservation, Cell Processing And Formulation, Cell Isolation And Activation), By End-Use (Clinical, Commercial) – Market Size, Trends, Strategies, and Forecast to 2035
GMP Cell Therapy Consumables Market Overview
• GMP Cell Therapy Consumables market size has reached to $22.73 billion in 2025 • Expected to grow to $73.96 billion in 2030 at a compound annual growth rate (CAGR) of 26.6% • Growth Driver: Rising Prevalence Of Chronic Diseases To Drive Market Growth • Market Trend: Advancements in GMP-Compliant CD34+ Hematopoietic Stem Cells: OrganaBio’s Launch and Industry Developments • North America was the largest region in 2025 and Asia-Pacific is the fastest growing region.What Is Covered Under GMP Cell Therapy Consumables Market?
GMP cell therapy consumables refer to consumable products and goods produced under good manufacturing practices (GMP) guidelines for cell-based therapies. Consumables and tools for cell culture are made to help researchers grow new cells in culture media for gene therapy, vaccine manufacture, tissue culture, toxicity testing, and drug development. The main products of GMP cell therapy consumables are kits, reagents or molecular biology reagents, growth factors or cytokines and interleukins, and others. Kit refers to a collection of items, such as tools or equipment, utilized for a specific activity or purpose. Kits in GMP cell therapy consumables are used for the isolation, expansion, harvesting, and formulation of cell therapies. The various therapies include NK cell therapy, stem cell therapy, T-cell therapy, and others that follows processes such as cell collection and characterization or sorting and separation, cell culture and expansion or preparation, cryopreservation, cell processing and formulation, cell isolation and activation, cell distribution or handling, process monitoring and control or re-administration or quality assurance, and others. The GMP cell therapy consumables are used in clinicals, commercial, and research.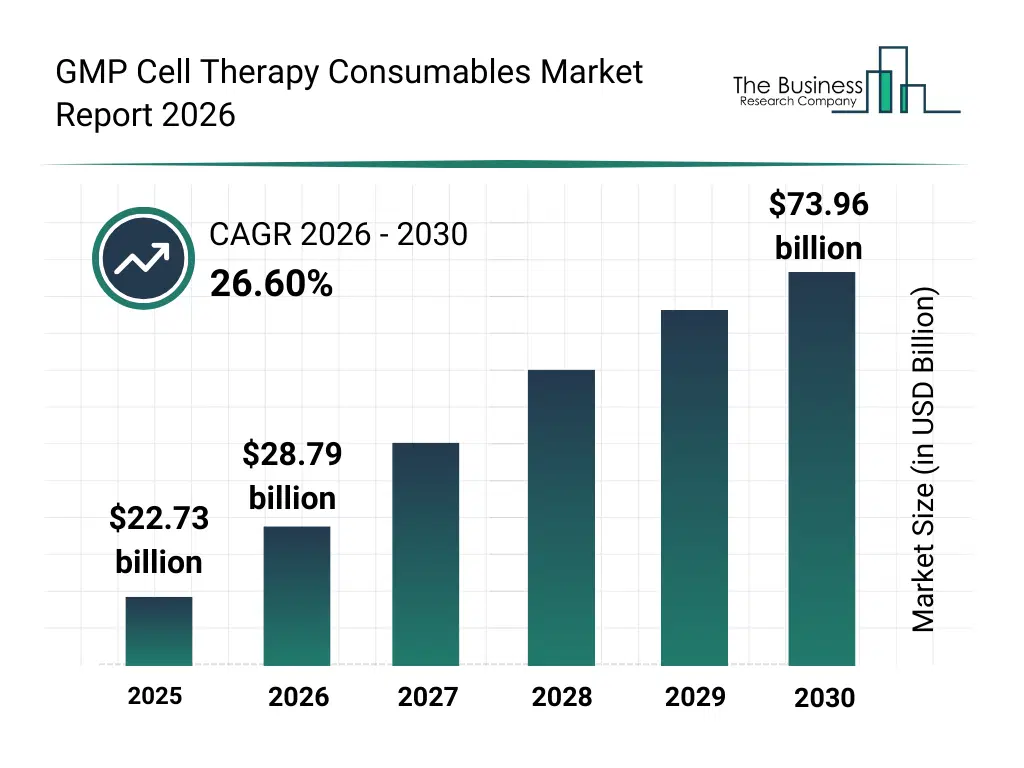
What Is The GMP Cell Therapy Consumables Market Size and Share 2026?
The gmp cell therapy consumables market size has grown exponentially in recent years. It will grow from $22.73 billion in 2025 to $28.79 billion in 2026 at a compound annual growth rate (CAGR) of 26.7%. The growth in the historic period can be attributed to expansion of clinical cell therapy trials, growth of regenerative medicine research, increasing adoption of gmp manufacturing standards, rising investments in cell-based therapies, availability of advanced cell culture tools.What Is The GMP Cell Therapy Consumables Market Growth Forecast?
The gmp cell therapy consumables market size is expected to see exponential growth in the next few years. It will grow to $73.96 billion in 2030 at a compound annual growth rate (CAGR) of 26.6%. The growth in the forecast period can be attributed to increasing commercialization of cell therapies, rising demand for scalable gmp production, expansion of gene and immunotherapy pipelines, growing focus on process automation, increasing regulatory scrutiny of cell therapy manufacturing. Major trends in the forecast period include increasing demand for gmp-compliant cell therapy consumables, rising adoption of single-use bioprocessing products, growing use of automated cell processing systems, expansion of standardized cell culture solutions, enhanced focus on quality and regulatory compliance.Global GMP Cell Therapy Consumables Market Segmentation
1) By Product: Kits, Reagents Or Molecular Biology Reagents, Growth Factors Or Cytokines And Interleukins, Other Products 2) By Cell Therapy: NK Cell Therapy, Stem Cell Therapy, T-Cell Therapy 3) By Process: Cell Collection And Characterization Or Sorting And Separation, Cell Culture And Expansion Or Preparation, Cryopreservation, Cell Processing And Formulation, Cell Isolation And Activation 4) By End-Use: Clinical, Commercial Subsegments: 1) By Kits: Cell Culture Kits, Cell Isolation Kits, Transfection Kits 2) By Reagents Or Molecular Biology Reagents: Buffers And Solutions, Enzymes, Staining Reagents 3) By Growth Factors Or Cytokines And Interleukins: Hematopoietic Growth Factors, Immunomodulatory Cytokines 4) By Other Products: Cell Culture Vessels, Media And SupplementsWhat Is The Driver Of The GMP Cell Therapy Consumables Market?
The increasing prevalence of chronic diseases is expected to propel the growth of GMP cell therapy consumables market going forward. Chronic disease refers to a form of illness that lasts for three months or more and can deteriorate with time. Surge in chronic diseases leads to increased research and development in innovative cell therapies to address chronic conditions. The demand for GMP consumables is rising as cell therapy applications expand across various chronic diseases, necessitating quality maintenance in clinical trials. Stringent regulatory compliance and patient demand for innovative therapies further contribute to the market's growth. For instance, in June 2024, according to the National Health Service, a UK-based government department, 3,615,330 individuals registered with a general practitioner (GP) were diagnosed with non-diabetic hyperglycemia or pre-diabetes (a condition with elevated blood sugar levels, not high enough to be classified as diabetes) in 2023, marking an 18% increase from 3,065,825 cases in 2022. Therefore, the increasing prevalence of chronic diseases is driving the growth of the GMP cell therapy consumables industry.Key Players In The Global GMP Cell Therapy Consumables Market
Major companies operating in the gmp cell therapy consumables market are Thermo Fisher Scientific Inc., Merck KGaA, Lonza Group AG, Sartorius AG, Corning Incorporated, Avantor Inc., Fresenius Kabi AG, Miltenyi Biotec, Bio-Techne Corp, STEMCELL Technologies Inc., FUJIFILM Irvine Scientific Inc., BioLife Solutions Inc., PromoCell GmbH, PeproTech Inc., Sino Biological Inc., Wilson Wolf Corporation, Cellexus Ltd., MaxCyte Inc., BioLegend Inc., Repligen Corporation, Terumo CorporationGlobal GMP Cell Therapy Consumables Market Trends and Insights
Major companies operating in the GMP cell therapy consumables market are developing GMP-compliant CD34+ hematopoietic stem cells (HSCs) to support advanced therapies. GMP-compliant CD34+ hematopoietic stem cells (HSCs) refer to a specific type of stem cell that expresses the CD34 marker on its surface and is produced under Good Manufacturing Practice (GMP) conditions. For instance, in April 2024, OrganaBio, a US-based pharmaceutical company, launched HematoPAC-HSC-CB-GMP, an on-demand source of GMP-compliant CD34+ hematopoietic stem cells (HSCs) derived from fresh human cord blood. OrganaBio utilizes its extensive experience in cell isolation and GMP manufacturing to produce high yields of highly viable CD34+ HSCs within 24 hours of collection. This launch aims to facilitate the development of next-generation therapies, including those for blood cancers and genetic disorders.What Are Latest Mergers And Acquisitions In The GMP Cell Therapy Consumables Market?
In March 2023, Sartorius AG, a Germany-based provider of bioprocess solutions, single-use technologies, and laboratory instruments for the biopharmaceutical industry, acquired Polyplus for approximately €2.4 billion (about US$2.6 billion). Sartorius+1 With this acquisition, Sartorius aims to reinforce its position as a leading supplier of critical upstream technologies for cell and gene therapies by integrating Polyplus’s transfection reagents and GMP-grade plasmid DNA into its existing portfolio of cell culture media and other critical bioprocess components, thereby enhancing its ability to support viral vector manufacturing workflows for advanced therapies. Polyplus is a France-based provider of innovative upstream technologies for cell and gene therapies that develops and produces transfection reagents, other DNA/RNA delivery reagents, and plasmid DNA in high quality and GMP grade, which serve as key components in the production of viral vectors used in cell and gene therapies and other advanced medicinal products.Regional Outlook
North America was the largest region in the GMP cell therapy consumables market in 2025. Asia-Pacific is expected to be the fastest-growing region in the forecast period. The regions covered in this market report are Asia-Pacific, South East Asia, Western Europe, Eastern Europe, North America, South America, Middle East, Africa. The countries covered in this market report are Australia, Brazil, China, France, Germany, India, Indonesia, Japan, Taiwan, Russia, South Korea, UK, USA, Canada, Italy, SpainWhat Defines the GMP Cell Therapy Consumables Market?
The GMP cell therapy consumables market consists of sales of cell culture media, media supplements, extracellular matrices, flasks, tubes, dishes or cryovials, serological pipettes, or centrifugation tubes. Values in this market are ‘factory gate’ values, that is the value of goods sold by the manufacturers or creators of the goods, whether to other entities (including downstream manufacturers, wholesalers, distributors, and retailers) or directly to end customers. The value of goods in this market includes related services sold by the creators of the goods.How is Market Value Defined and Measured?
The market value is defined as the revenues that enterprises gain from the sale of goods and/or services within the specified market and geography through sales, grants, or donations in terms of the currency (in USD unless otherwise specified). The revenues for a specified geography are consumption values that are revenues generated by organizations in the specified geography within the market, irrespective of where they are produced. It does not include revenues from resales along the supply chain, either further along the supply chain or as part of other products.GMP Cell Therapy Consumables Market Report Forecast Analysis
| Report Attribute | Details |
|---|---|
| Market Size Value In 2026 | $28.79 billion |
| Revenue Forecast In 2035 | $73.96 billion |
| Growth Rate | CAGR of 26.7% from 2026 to 2035 |
| Base Year For Estimation | 2025 |
| Actual Estimates/Historical Data | 2020-2025 |
| Forecast Period | 2026 - 2030 - 2035 |
| Market Representation | Revenue in USD Billion and CAGR from 2026 to 2035 |
| Segments Covered | Product, Cell Therapy, Process, End-Use |
| Regional Scope | Asia-Pacific, Western Europe, Eastern Europe, North America, South America, Middle East, Africa |
| Country Scope | The countries covered in the report are Australia, Brazil, China, France, Germany, India, ... |
| Key Companies Profiled | Thermo Fisher Scientific Inc., Merck KGaA, Lonza Group AG, Sartorius AG, Corning Incorporated, Avantor Inc., Fresenius Kabi AG, Miltenyi Biotec, Bio-Techne Corp, STEMCELL Technologies Inc., FUJIFILM Irvine Scientific Inc., BioLife Solutions Inc., PromoCell GmbH, PeproTech Inc., Sino Biological Inc., Wilson Wolf Corporation, Cellexus Ltd., MaxCyte Inc., BioLegend Inc., Repligen Corporation, Terumo Corporation |
| Customization Scope | Request for Customization |
| Pricing And Purchase Options | Explore Purchase Options |
