
Handheld Multi-Parameter Monitoring Device Market Report 2026
Global Outlook – By Device Type (Basic Vital Sign Monitors, Advanced Multi-Parameter Monitors, Fetal Monitors, Remote Patient Monitoring Devices (RPM) Devices), By Connectivity (Wired Devices, Wireless Devices), By Application (Intensive Care, Clinical Care, Ambulatory Care, Pediatric Care, Other Applications), By End User (Hospitals, Ambulatory Surgical Centers, Homecare Settings, Other End Users) – Market Size, Trends, Strategies, and Forecast to 2035
Handheld Multi-Parameter Monitoring Device Market Overview
• Handheld Multi-Parameter Monitoring Device market size has reached to $3.69 billion in 2025 • Expected to grow to $5.38 billion in 2030 at a compound annual growth rate (CAGR) of 7.8% • Growth Driver: The Rising Demand For Remote Patient Monitoring Is Fueling The Market Growth Due To Increasing Need For Continuous Health Tracking • Market Trend: Advancements In Sensor-Based Wearable Monitoring Technology • North America was the largest region in 2025.What Is Covered Under Handheld Multi-Parameter Monitoring Device Market?
A handheld multi-parameter monitoring device refers to a portable medical device that measures and displays multiple vital signs such as heart rate, blood pressure, temperature, and oxygen saturation in real-time for patient monitoring. This device is utilized by healthcare professionals to quickly assess and monitor a patient’s vital signs at the point of care, enabling timely diagnosis and intervention. The main device types in handheld multi-parameter monitoring devices include basic vital sign monitors, advanced multi-parameter monitors, fetal monitors, and remote patient monitoring (RPM) devices. Basic vital sign monitors are portable devices that track key health indicators such as heart rate, blood pressure, and oxygen levels in real time. Based on connectivity, these devices are categorized as wired or wireless. They are used in various applications, including intensive care, clinical care, ambulatory care, pediatric care, and other settings. The primary end users of these devices are hospitals, ambulatory surgical centers, homecare settings, and other healthcare providers.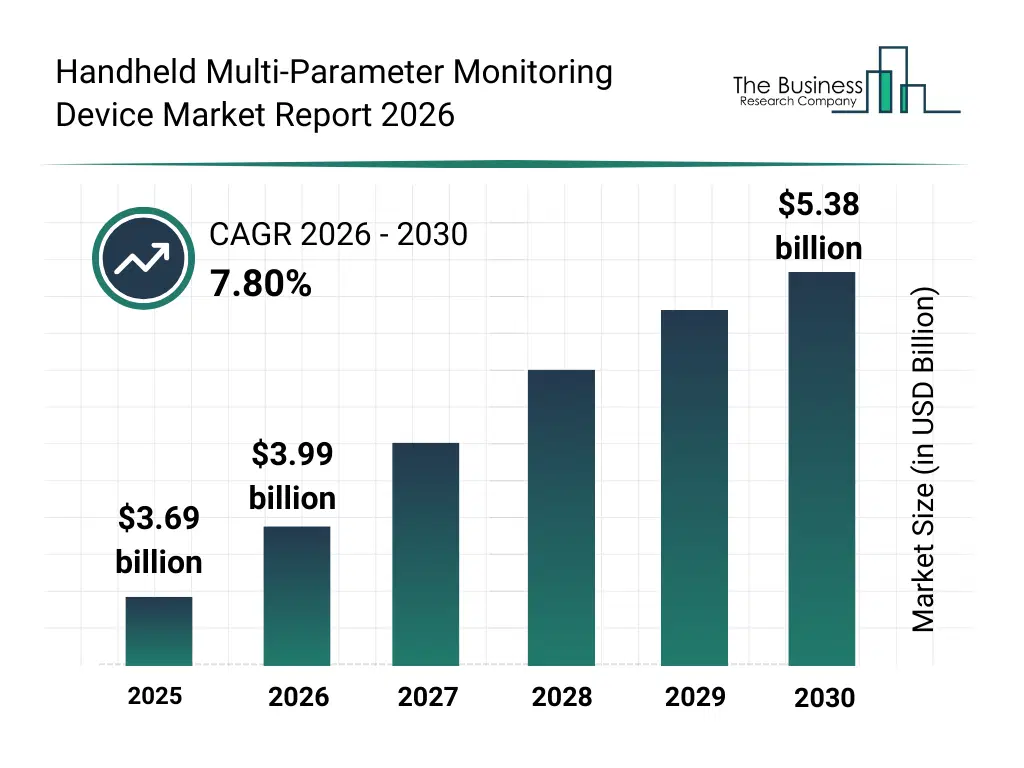
What Is The Handheld Multi-Parameter Monitoring Device Market Size and Share 2026?
The handheld multi-parameter monitoring device market size has grown strongly in recent years. It will grow from $3.69 billion in 2025 to $3.99 billion in 2026 at a compound annual growth rate (CAGR) of 8.1%. The growth in the historic period can be attributed to limited availability of portable monitoring devices, reliance on wired connectivity, growing hospital and clinical care infrastructure, increasing demand for vital sign monitoring, conventional patient assessment methods.What Is The Handheld Multi-Parameter Monitoring Device Market Growth Forecast?
The handheld multi-parameter monitoring device market size is expected to see strong growth in the next few years. It will grow to $5.38 billion in 2030 at a compound annual growth rate (CAGR) of 7.8%. The growth in the forecast period can be attributed to advancements in wireless and wearable technologies, increasing telehealth and homecare adoption, rising demand for advanced multi-parameter monitoring, integration with ai and cloud-based platforms, expansion of pediatric and ambulatory care services. Major trends in the forecast period include rising adoption of wireless and wearable monitoring devices, increasing use of remote patient monitoring (rpm) solutions, integration of multi-parameter monitors in ambulatory and homecare settings, growing demand for portable and user-friendly devices, enhanced focus on real-time data accuracy and patient safety.Global Handheld Multi-Parameter Monitoring Device Market Segmentation
1) By Device Type: Basic Vital Sign Monitors, Advanced Multi-Parameter Monitors, Fetal Monitors, Remote Patient Monitoring Devices (RPM) Devices 2) By Connectivity: Wired Devices, Wireless Devices 3) By Application: Intensive Care, Clinical Care, Ambulatory Care, Pediatric Care, Other Applications 4) By End User: Hospitals, Ambulatory Surgical Centers, Homecare Settings, Other End Users Subsegments: 1) By Basic Vital Sign Monitors: Pulse Oximeters, Blood Pressure Monitors, Thermometers, Glucose Monitors, Heart Rate Monitors 2) By Advanced Multi-Parameter Monitors: Electrocardiogram Monitors (ECG) Monitors, Capnography Monitors, Respiratory Rate Monitors, Hemoglobin Monitors 3) By Fetal Monitors: Doppler Fetal Monitors, Fetal Heart Rate Monitors 4) By Remote Patient Monitoring Devices (RPM) Devices: Wearable Health Trackers, Telehealth Monitoring Devices, Mobile Health (mHealth) Devices, Wireless Vital Sign MonitorsWhat Are The Drivers Of The Handheld Multi-Parameter Monitoring Device Market?
The rising demand for remote patient monitoring is expected to propel the growth of the handheld multi-parameter monitoring device market going forward. Remote patient monitoring refers to using digital technologies to collect health data from patients in one location and electronically transmit it to healthcare providers in a different location for assessment and recommendations. The rising demand for remote patient monitoring is primarily due to the increasing prevalence of chronic diseases, as these conditions require continuous health tracking to manage symptoms, prevent complications, and reduce hospital visits. A handheld multi-parameter monitoring device supports remote patient monitoring by tracking key vital signs like heart rate, blood pressure, and oxygen levels. It enables early detection of health issues, continuous care for chronic conditions, and real-time data sharing with healthcare providers, improving convenience and reducing hospital visits. For instance, in August 2023, according to the Alliance for Connected Care, a US-based nonprofit advocacy organization, 81% of clinicians were using remote patient monitoring (RPM), and 84% of those planned to expand usage in 2024, with 45% of providers utilizing RPM for acute care, including hospital-at-home programs. Therefore, the rising demand for remote patient monitoring is driving the growth of the handheld multi-parameter monitoring device industry. The rising prevalence of chronic disease is expected to propel the growth of the handheld multi-parameter monitoring device market going forward. Chronic diseases are long-lasting health conditions, typically persisting for three months or more, requiring ongoing management. They include diabetes, heart disease, and arthritis, impacting quality of life significantly. The growing prevalence of chronic diseases is attributed to factors such as aging populations, unhealthy lifestyle choices, poor diet, physical inactivity, and increased stress levels. Handheld multi-parameter monitoring devices support chronic disease management by enabling continuous, real-time tracking of key vitals outside clinical settings, allowing earlier detection of deterioration. For instance, in April 2025, according to the NIHCM Foundation, a US-based nonprofit organization, chronic diseases affect approximately 60% of Americans, with many individuals suffering from more than one chronic illness. Therefore, the rising prevalence of chronic disease is driving the growth of the handheld multi-parameter monitoring device industry.Key Players In The Global Handheld Multi-Parameter Monitoring Device Market
Major companies operating in the handheld multi-parameter monitoring device market are Abbott Laboratories, Medtronic Public Limited Company, Koninklijke Philips N.V., GE HealthCare Technologies Inc., Omron Healthcare Co. Ltd., Shenzhen Mindray Bio-Medical Electronics Co. Ltd., Masimo Corporation, Nihon Kohden Corporation, Lepu Medical Technology Beijing Co. Ltd., Qingdao Meditech Equipment Co. Ltd., Wuhan Zoncare Bio-Medical Electronics Co. Ltd., BPL Medical Technologies Pvt. Ltd., Schiller Healthcare India Pvt. Ltd., Contec Medical Systems Co. Ltd., aXcent medical GmbH, Jiangsu Yuyue Medical Instruments Co. Ltd., Edan Instruments Inc., iHealth Labs Inc., Nonin Medical Inc., Welch Allyn Inc., Beurer GmbH, SunTech Medical Inc.Global Handheld Multi-Parameter Monitoring Device Market Trends and Insights
Major companies operating in the handheld multi-parameter monitoring device market are focusing on advancements in medical technology, such as sensor technology, to enhance the accuracy, reliability, and real-time monitoring of vital signs, enabling early detection of health issues. Sensor technology refers to the use of devices that detect and measure physical or environmental changes, such as temperature, motion, or pressure. For instance, in May 2024, Sempulse Corporation, a U.S.-based medical device manufacturer, launched the Halo Vital Signs Monitoring System, which aims to enhance real-time physiological monitoring accuracy and support rapid clinical decision-making in emergency and remote care settings. The Halo is a miniature, rugged wearable sensor that attaches behind the ear or on the side of the neck, offering truly non-invasive, continuous monitoring of core body temperature, skin temperature, oxygen saturation (SpO₂), pulse rate, and respiratory rate even while the subject is in motion or in austere environments.Regional Outlook
North America was the largest region in the handheld multi-parameter monitoring device market in 2025. The regions covered in this market report are Asia-Pacific, South East Asia, Western Europe, Eastern Europe, North America, South America, Middle East, Africa. The countries covered in this market report are Australia, Brazil, China, France, Germany, India, Indonesia, Japan, Taiwan, Russia, South Korea, UK, USA, Canada, Italy, SpainWhat Defines the Handheld Multi-Parameter Monitoring Device Market?
The handheld multi-parameter monitoring device market consists of sales of handheld pulse oximeters, portable ECG monitors, handheld blood pressure monitors, handheld capnography devices, handheld glucose monitoring devices, infrared thermometers with vital sign monitoring, and compact multi-parameter patient monitors. Values in this market are ‘factory gate’ values, that is the value of goods sold by the manufacturers or creators of the goods, whether to other entities (including downstream manufacturers, wholesalers, distributors and retailers) or directly to end customers. The value of goods in this market includes related services sold by the creators of the goods.How is Market Value Defined and Measured?
The market value is defined as the revenues that enterprises gain from the sale of goods and/or services within the specified market and geography through sales, grants, or donations in terms of the currency (in USD unless otherwise specified). The revenues for a specified geography are consumption values that are revenues generated by organizations in the specified geography within the market, irrespective of where they are produced. It does not include revenues from resales along the supply chain, either further along the supply chain or as part of other products.Handheld Multi-Parameter Monitoring Device Market Report Forecast Analysis
| Report Attribute | Details |
|---|---|
| Market Size Value In 2026 | $3.99 billion |
| Revenue Forecast In 2035 | $5.38 billion |
| Growth Rate | CAGR of 8.1% from 2026 to 2035 |
| Base Year For Estimation | 2025 |
| Actual Estimates/Historical Data | 2020-2025 |
| Forecast Period | 2026 - 2030 - 2035 |
| Market Representation | Revenue in USD Billion and CAGR from 2026 to 2035 |
| Segments Covered | Device Type, Connectivity, Application, End User |
| Regional Scope | Asia-Pacific, Western Europe, Eastern Europe, North America, South America, Middle East, Africa |
| Country Scope | The countries covered in the report are Australia, Brazil, China, France, Germany, India, ... |
| Key Companies Profiled | Abbott Laboratories, Medtronic Public Limited Company, Koninklijke Philips N.V., GE HealthCare Technologies Inc., Omron Healthcare Co. Ltd., Shenzhen Mindray Bio-Medical Electronics Co. Ltd., Masimo Corporation, Nihon Kohden Corporation, Lepu Medical Technology Beijing Co. Ltd., Qingdao Meditech Equipment Co. Ltd., Wuhan Zoncare Bio-Medical Electronics Co. Ltd., BPL Medical Technologies Pvt. Ltd., Schiller Healthcare India Pvt. Ltd., Contec Medical Systems Co. Ltd., aXcent medical GmbH, Jiangsu Yuyue Medical Instruments Co. Ltd., Edan Instruments Inc., iHealth Labs Inc., Nonin Medical Inc., Welch Allyn Inc., Beurer GmbH, SunTech Medical Inc. |
| Customization Scope | Request for Customization |
| Pricing And Purchase Options | Explore Purchase Options |
