
Heart Defect Closure Device Market Report 2026
Global Outlook – By Type (Atrial Septal Defect (ASD) Closure Device, Left Atrial Appendage (LAA) Closure Devices, Patent Foramen Ovale (PFO) Closure Devices, Patent Ductus Arteriosus (PDA) Closure Devices, Ventricular Septal Defect (VSD) Closure Devices), By Material (Nitinol-Based Devices, Stainless Steel Devices, Other Materials), By Mode Of Delivery (Transcatheter Delivery, Surgical Delivery, Other Modes), By End-User (Hospitals, Clinics, Ambulatory Surgical Centers, Other End-Users) – Market Size, Trends, Strategies, and Forecast to 2035
Heart Defect Closure Device Market Overview
• Heart Defect Closure Device market size has reached to $1.27 billion in 2025 • Expected to grow to $2.36 billion in 2030 at a compound annual growth rate (CAGR) of 12.9% • Growth Driver: Congenital Heart Problems Boost Heart Defect Closure Device Market Growth • Market Trend: AI-Driven Device Tracking Enhances Precision In Heart Valve Procedures • North America was the largest region in 2025 and Asia-Pacific is the fastest growing region.What Is Covered Under Heart Defect Closure Device Market?
A heart defect closure device is a medical device used to close abnormal openings or defects in the heart's structure, typically in the septum, which is the wall separating the heart's chambers. It is used to treat certain heart defects, such as atrial septal defects or patent foramen ovale, by closing heart's wall. They are delivered via minimally invasive procedures, providing an effective and less invasive alternative to open-heart surgery for patients with these conditions. The main types of heart defect closure devices are atrial septal defect (ASD) closure device, left atrial appendage (LAA) closure devices, patent foramen ovale (PFO) closure devices, patent ductus arteriosus (PDA) closure devices, and ventricular septal defect (VSD) closure devices. An atrial septal defect (ASD) closure device is used to seal a hole in the wall between the two upper chambers of the heart. These devices are made from various materials, including nitinol and stainless steel, and are delivered through transcatheter, surgical, and other delivery modes by hospitals, clinics, ambulatory surgical centers, and others.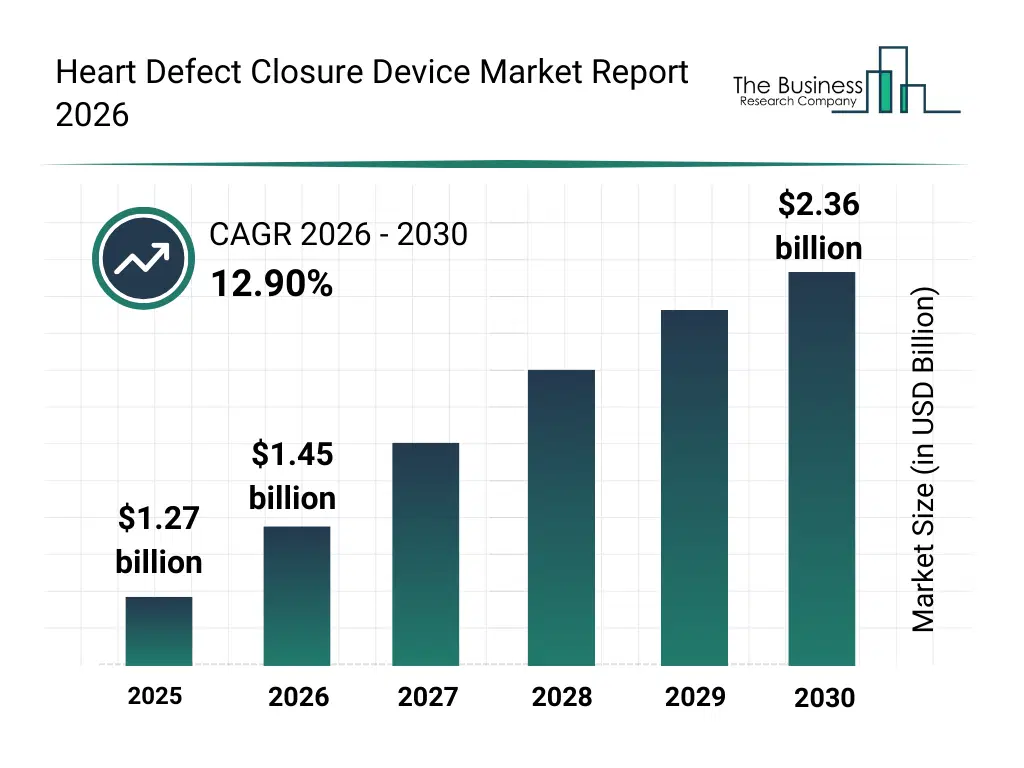
What Is The Heart Defect Closure Device Market Size and Share 2026?
The heart defect closure device market size has grown rapidly in recent years. It will grow from $1.27 billion in 2025 to $1.45 billion in 2026 at a compound annual growth rate (CAGR) of 14.5%. The growth in the historic period can be attributed to increasing prevalence of congenital heart defects, advancements in interventional cardiology, growing awareness of septal defect treatments, expansion of cardiac catheterization labs, rising demand for minimally invasive cardiac procedure.What Is The Heart Defect Closure Device Market Growth Forecast?
The heart defect closure device market size is expected to see rapid growth in the next few years. It will grow to $2.36 billion in 2030 at a compound annual growth rate (CAGR) of 12.9%. The growth in the forecast period can be attributed to growing elderly and pediatric patient population with structural heart defects, rising adoption of transcatheter cardiac interventions, increasing healthcare expenditure on cardiovascular care, expansion of specialized cardiac centers, growing focus on early diagnosis and treatment of heart defects. Major trends in the forecast period include rising adoption of minimally invasive transcatheter closure procedures, growing use of nitinol-based and self-expanding closure devices, increasing preference for device-based alternatives to open-heart surgery, expansion of structural heart disease treatment programs, rising focus on improved safety, efficacy, and long-term outcomes.Global Heart Defect Closure Device Market Segmentation
1) By Type: Atrial Septal Defect (ASD) Closure Device, Left Atrial Appendage (LAA) Closure Devices, Patent Foramen Ovale (PFO) Closure Devices, Patent Ductus Arteriosus (PDA) Closure Devices, Ventricular Septal Defect (VSD) Closure Devices 2) By Material: Nitinol-Based Devices, Stainless Steel Devices, Other Materials 3) By Mode Of Delivery: Transcatheter Delivery, Surgical Delivery, Other Modes 4) By End-User: Hospitals, Clinics, Ambulatory Surgical Centers, Other End-Users Subsegments: 1) By Atrial Septal Defect (ASD) Closure Device: Occluder Devices (Septal Occluders), Implantable Patch Devices, Self-Expanding Devices, Balloon-Expandable Devices 2) By Left Atrial Appendage (LAA) Closure Devices: Occluder Devices (WATCHMAN Device), Clip-Based Devices, Self-Expanding LAA Closure Devices 3) By Patent Foramen Ovale (PFO) Closure Devices: Septal Occluders, Self-Expanding Occluders, Balloon-Expandable Occluders 4) By Patent Ductus Arteriosus (PDA) Closure Devices: Coil-Based Closure Devices, Stent-Based Closure Devices, Amplatzer Occluders 5) By Ventricular Septal Defect (VSD) Closure Devices: Septal Occluder Devices, Balloon-Expandable Occluders, Self-Expanding OccludersWhat Is The Driver Of The Heart Defect Closure Device Market?
The growing number of people with congenital heart problems is expected to propel the growth of the heart defect closure device market going forward. Congenital heart problems refer to structural defects in the heart that are present at birth. The increase in congenital heart problems is due to genetic predispositions, environmental influences, and maternal health issues such as diabetes, obesity, and exposure to certain medications or infections during pregnancy. Heart defect closure devices help to treat congenital heart problems by offering minimally invasive solutions to seal abnormal heart openings, thus restoring normal blood flow, reducing complications, and improving overall cardiac function. For instance, in August 2025, according to the National Center for Biotechnology Information, a US-based governmental agency, between 2025 and 2050, cardiovascular disease is projected to rise significantly, with prevalence increasing by 90.0%, crude mortality by 73.4%, and crude DALYs by 54.7%, leading to an estimated 35.6 million cardiovascular deaths in 2050, up from 20.5 million in 2025. Therefore, the growing number of people with congenital heart problems is driving the heart defect closure device industry.Key Players In The Global Heart Defect Closure Device Market
Major companies operating in the heart defect closure device market are Abbott Laboratories, Medtronic, W L Gore and Associates, Occlutech, LifeTech Scientific Corporation, Lepu Medical Technology, Meril Life Sciences Pvt Ltd, MicroPort Scientific Corporation, Boston Scientific Corporation, Edwards Lifesciences, Terumo Corporation, Cardia Inc, Coherex Medical Inc, AtriCure Inc, Sahajanand Medical Technologies Limited, Vivasure Medical, Transmural Systems Inc, Cormatrix Cardiovascular Inc, Baylis Medical Company Inc, Biosense Webster Inc, BIOTRONIK SE and Co KG, B BraunGlobal Heart Defect Closure Device Market Trends and Insights
Major companies in the heart defect closure device market are advancing technologies such as device tracking tools to enhance procedural precision and clinician confidence. A device tracking tool is a system that provides real-time visualization of the position and movement of medical devices during treatment. For instance, in November 2025, Philips, a Netherlands-based health technology company, introduced DeviceGuide—an AI-driven tracking solution designed to support clinicians during complex minimally invasive heart valve repair procedures, especially for mitral valve regurgitation. Integrated with the EchoNavigator platform, DeviceGuide merges live X-ray and ultrasound imaging to generate a real-time 3D model of the repair device within the beating heart, improving visualization, orientation, and accuracy. Developed in collaboration with Edwards Lifesciences, the system streamlines workflows, enhances team coordination, and supports broader access to advanced cardiac interventions worldwide.What Are Latest Mergers And Acquisitions In The Heart Defect Closure Device Market?
In February 2023, Boston Scientific Corporation, a US-based medical device manufacturing company, acquired a 65% stake in Acotec Scientific Holdings Limited for $0.523 billion. Following this acquisition, the position of Boston Scientific Corporation will be strengthened in China and will commercialize Acotec Scientific Holdings Limited's products globally. Acotec Scientific Holdings Limited is a China-based medical device company focused on interventional treatment of vascular diseases.Regional Outlook
North America was the largest region in the heart defect closure device market in 2025. Asia-Pacific is expected to be the fastest-growing region in the forecast period. The regions covered in this market report are Asia-Pacific, South East Asia, Western Europe, Eastern Europe, North America, South America, Middle East, Africa. The countries covered in this market report are Australia, Brazil, China, France, Germany, India, Indonesia, Japan, Taiwan, Russia, South Korea, UK, USA, Canada, Italy, SpainWhat Defines the Heart Defect Closure Device Market?
The heart defect closure device market consists of sales of amplatzer septal occluder, gore cardioform septal occluder, and HELEX septal occluder. Values in this market are ‘factory gate’ values, that is, the value of goods sold by the manufacturers or creators of the goods, whether to other entities (including downstream manufacturers, wholesalers, distributors, and retailers) or directly to end customers. The value of goods in this market includes related services sold by the creators of the goods.How is Market Value Defined and Measured?
The market value is defined as the revenues that enterprises gain from the sale of goods and/or services within the specified market and geography through sales, grants, or donations in terms of the currency (in USD unless otherwise specified). The revenues for a specified geography are consumption values that are revenues generated by organizations in the specified geography within the market, irrespective of where they are produced. It does not include revenues from resales along the supply chain, either further along the supply chain or as part of other products.Heart Defect Closure Device Market Report Forecast Analysis
| Report Attribute | Details |
|---|---|
| Market Size Value In 2026 | $1.45 billion |
| Revenue Forecast In 2035 | $2.36 billion |
| Growth Rate | CAGR of 14.5% from 2026 to 2035 |
| Base Year For Estimation | 2025 |
| Actual Estimates/Historical Data | 2020-2025 |
| Forecast Period | 2026 - 2030 - 2035 |
| Market Representation | Revenue in USD Billion and CAGR from 2026 to 2035 |
| Segments Covered | Type, Material, Mode Of Delivery, End-User |
| Regional Scope | Asia-Pacific, Western Europe, Eastern Europe, North America, South America, Middle East, Africa |
| Country Scope | The countries covered in the report are Australia, Brazil, China, France, Germany, India, ... |
| Key Companies Profiled | Abbott Laboratories, Medtronic, W L Gore and Associates, Occlutech, LifeTech Scientific Corporation, Lepu Medical Technology, Meril Life Sciences Pvt Ltd, MicroPort Scientific Corporation, Boston Scientific Corporation, Edwards Lifesciences, Terumo Corporation, Cardia Inc, Coherex Medical Inc, AtriCure Inc, Sahajanand Medical Technologies Limited, Vivasure Medical, Transmural Systems Inc, Cormatrix Cardiovascular Inc, Baylis Medical Company Inc, Biosense Webster Inc, BIOTRONIK SE and Co KG, B Braun |
| Customization Scope | Request for Customization |
| Pricing And Purchase Options | Explore Purchase Options |
