
Hemodynamic Flow Alteration Devices Market Report 2026
Global Outlook – By Type (Embolic Protection Device, Chronic Total Occlusion Device), By Application (Cardiovascular Diseases, Neurological Disorders, Renal Disorders, Respiratory Disorders), By End User (Hospitals, Clinics, Ambulatory Care Centre, Home Care Setting, Other End Users) – Market Size, Trends, Strategies, and Forecast to 2035
Hemodynamic Flow Alteration Devices Market Overview
• Hemodynamic Flow Alteration Devices market size has reached to $0.88 billion in 2025 • Expected to grow to $1.3 billion in 2030 at a compound annual growth rate (CAGR) of 7.9% • Growth Driver: Rising Cardiovascular Disease Prevalence Drives The Market • Market Trend: Fluid Biomed Achieves First Human Implantation of Polymer-Based Neurovascular Stent for Brain Aneurysms • North America was the largest region in 2025.What Is Covered Under Hemodynamic Flow Alteration Devices Market?
Hemodynamic flow alteration devices are medical tools designed to modify blood flow dynamics within the circulatory system. They are often used in procedures such as vascular surgery or treatment of heart conditions. These devices can influence the speed, direction, or resistance of blood flow to improve circulation or reduce strain on organs. The main types of hemodynamic flow alteration devices are the embolic protection device and the chronic total occlusion device. An embolic protection device is a medical device designed to capture and remove embolic material that may be dislodged during procedures such as angioplasty, preventing it from traveling to vital organs and causing blockages. It is used for various applications such as cardiovascular diseases, neurological disorders, renal disorders, and respiratory disorders by several end users, including hospitals, clinics, ambulatory care centers, home care settings, and other end users.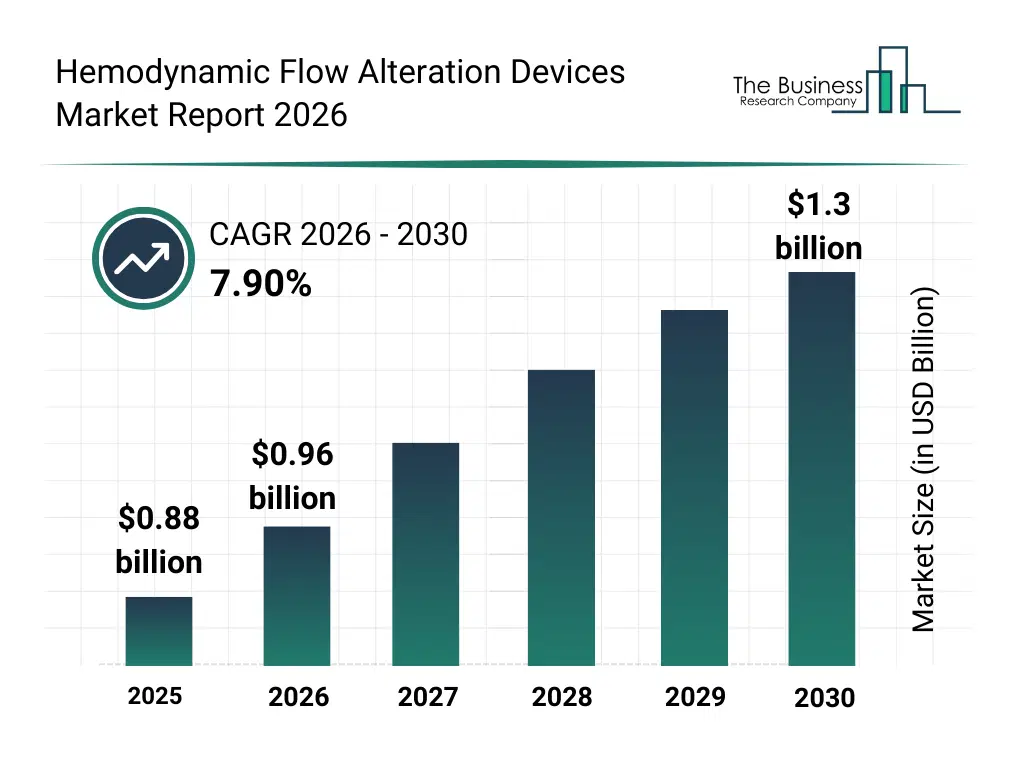
What Is The Hemodynamic Flow Alteration Devices Market Size and Share 2026?
The hemodynamic flow alteration devices market size has grown strongly in recent years. It will grow from $0.88 billion in 2025 to $0.96 billion in 2026 at a compound annual growth rate (CAGR) of 8.2%. The growth in the historic period can be attributed to rising prevalence of cardiovascular disorders, increasing volume of vascular intervention procedures, advancements in catheter-based technologies, wider availability of specialized interventional devices, improved clinical outcomes in flow management.What Is The Hemodynamic Flow Alteration Devices Market Growth Forecast?
The hemodynamic flow alteration devices market size is expected to see strong growth in the next few years. It will grow to $1.3 billion in 2030 at a compound annual growth rate (CAGR) of 7.9%. The growth in the forecast period can be attributed to increasing adoption of precision medicine in vascular care, rising demand for minimally invasive cardiac procedures, expansion of neurovascular intervention capabilities, growing investment in advanced interventional cardiology devices, increasing focus on reducing procedural complications. Major trends in the forecast period include increasing adoption of advanced embolic protection technologies, rising use of minimally invasive flow alteration devices, growing integration of precision-guided cto devices, expansion of procedure-specific device designs, enhanced focus on patient-specific hemodynamic optimization.Global Hemodynamic Flow Alteration Devices Market Segmentation
1) By Type: Embolic Protection Device, Chronic Total Occlusion Device 2) By Application: Cardiovascular Diseases, Neurological Disorders, Renal Disorders, Respiratory Disorders 3) By End User: Hospitals, Clinics, Ambulatory Care Centre, Home Care Setting, Other End Users Subsegments: 1) By Embolic Protection Device: Distal Embolic Protection Devices, Proximal Embolic Protection Devices, Filter-Based Embolic Protection Devices, Balloon-Based Embolic Protection Devices, Mesh Or Cage-Based Embolic Protection Devices 2) By Chronic Total Occlusion Device: Crossing Guidewires For CTO, CTO Balloon Catheters, CTO Microcatheters, Re-Entry Devices For CTO, CTO Support CathetersWhat Is The Driver Of The Hemodynamic Flow Alteration Devices Market?
The increase in the prevalence of cardiovascular diseases is expected to propel the hemodynamic flow alteration devices market going forward. Cardiovascular disease (CVD) is a broad term encompassing various conditions that affect the heart and blood vessels. The increase in the prevalence of cardiovascular diseases is driven by factors such as aging populations, poor dietary habits, sedentary lifestyles, and rising rates of conditions like hypertension and diabetes. Cardiovascular diseases drive the need for hemodynamic flow alteration devices, as they assist in managing and enhancing blood flow in patients with conditions such as heart failure, hypertension, and arterial blockages. For instance, in September 2024, according to the British Heart Foundation, a UK-based cardiovascular research charity, in the UK, approximately 7.6 million individuals are currently affected by heart and circulatory diseases, comprising 4 million men and 3.6 million women. By 2030, this number could increase by 1 million, and by 2040, it may rise to 2 million more than the current figures. Therefore, the increasing prevalence of cardiovascular disease is driving the hemodynamic flow alteration devices industry going forward.Key Players In The Global Hemodynamic Flow Alteration Devices Market
Major companies operating in the hemodynamic flow alteration devices market are Cardinal Health Inc., Abbott Laboratories, Medtronic plc, Siemens Healthcare GmbH, Becton Dickinson and Company, Stryker Corporation, Boston Scientific Corporation, Edwards Lifesciences Corporation, W. L. Gore & Associates Inc., ZOLL Medical Corporation, Getinge AB, Lepu Medical Technology (Beijing) Co. Ltd., Abiomed Inc., LivaNova PLC, Cordis US Corp., Silk Road Medical Inc., Berlin Heart GmbH, CorWave SA, Jarvik Heart Inc., CARMAT SAGlobal Hemodynamic Flow Alteration Devices Market Trends and Insights
Major companies operating in the hemodynamic flow alteration devices market are focusing on technological innovation, such as hybrid polymer-metal flow-diverting stents, to improve the safety, efficacy, and long-term outcomes of neurovascular interventions. Hybrid Polymer-Metal Flow-Diverting Stents combine the mechanical support of metal with the biocompatibility and flexibility of polymer, enabling controlled hemodynamic flow alteration, reduced thrombogenicity, and enhanced vessel healing in patients with brain aneurysms. For instance, in June 2023, Fluid Biomed Inc., a Canada-based medical device company, launched REDIRECT, its first-in-human clinical trial, successfully implanting ReSolv, the world’s first hybrid polymer-metal neurovascular stent. The Phase 1 study demonstrated safe implantation with no adverse events at the 30-day follow-up, marking a milestone in brain aneurysm treatment by combining advanced materials engineering with minimally invasive neurovascular therapy to improve patient outcomes.What Are Latest Mergers And Acquisitions In The Hemodynamic Flow Alteration Devices Market?
In October 2024, Johnson and Johnson, a US-based pharmaceutical company, acquired V-Wave Inc. for an undisclosed amount. This acquisition aims to strengthen Johnson and Johnson's presence in the cardiovascular disease market by incorporating V-Wave’s innovative Ventura interatrial shunt into its MedTech portfolio. V-Wave Inc. is a US-based company specializing in providing hemodynamic flow alteration devices.Regional Outlook
North America was the largest region in the hemodynamic flow alteration devices market in 2025. The regions covered in this market report are Asia-Pacific, South East Asia, Western Europe, Eastern Europe, North America, South America, Middle East, Africa. The countries covered in this market report are Australia, Brazil, China, France, Germany, India, Indonesia, Japan, Taiwan, Russia, South Korea, UK, USA, Canada, Italy, SpainWhat Defines the Hemodynamic Flow Alteration Devices Market?
The hemodynamic flow alteration devices market consists of sales of flow diverters, vascular stents, blood flow regulators, and monitoring tools. Values in and diagnostic tools. Values in this market are ‘factory gate’ values, that is, the value of goods sold by the manufacturers or creators of the goods, whether to other entities (including downstream manufacturers, wholesalers, distributors, and retailers) or directly to end customers. The value of goods in this market includes related services sold by the creators of the goods.How is Market Value Defined and Measured?
The market value is defined as the revenues that enterprises gain from the sale of goods and/or services within the specified market and geography through sales, grants, or donations in terms of the currency (in USD unless otherwise specified). The revenues for a specified geography are consumption values that are revenues generated by organizations in the specified geography within the market, irrespective of where they are produced. It does not include revenues from resales along the supply chain, either further along the supply chain or as part of other products.Hemodynamic Flow Alteration Devices Market Report Forecast Analysis
| Report Attribute | Details |
|---|---|
| Market Size Value In 2026 | $0.96 billion |
| Revenue Forecast In 2035 | $1.3 billion |
| Growth Rate | CAGR of 8.2% from 2026 to 2035 |
| Base Year For Estimation | 2025 |
| Actual Estimates/Historical Data | 2020-2025 |
| Forecast Period | 2026 - 2030 - 2035 |
| Market Representation | Revenue in USD Billion and CAGR from 2026 to 2035 |
| Segments Covered | Type, Application, End User |
| Regional Scope | Asia-Pacific, Western Europe, Eastern Europe, North America, South America, Middle East, Africa |
| Country Scope | The countries covered in the report are Australia, Brazil, China, France, Germany, India, ... |
| Key Companies Profiled | Cardinal Health Inc., Abbott Laboratories, Medtronic plc, Siemens Healthcare GmbH, Becton Dickinson and Company, Stryker Corporation, Boston Scientific Corporation, Edwards Lifesciences Corporation, W. L. Gore & Associates Inc., ZOLL Medical Corporation, Getinge AB, Lepu Medical Technology (Beijing) Co. Ltd., Abiomed Inc., LivaNova PLC, Cordis US Corp., Silk Road Medical Inc., Berlin Heart GmbH, CorWave SA, Jarvik Heart Inc., CARMAT SA |
| Customization Scope | Request for Customization |
| Pricing And Purchase Options | Explore Purchase Options |
