
Hepatitis C Virus (HCV) Viral Load Testing Market Report 2026
Global Outlook – By Technology (Polymerase Chain Reaction, Nucleic Acid Sequence-Based Amplification, Transcription-Mediated Amplification, Other Technologies), By Product Type (Instruments, Kits And Reagents, Software And Services), By Sample Type (Blood, Plasma, Serum, Other Sample Types), By End User (Hospitals And Clinics, Diagnostic Laboratories, Blood Banks, Other End Users) – Market Size, Trends, Strategies, and Forecast to 2035
Hepatitis C Virus (HCV) Viral Load Testing Market Overview
• Hepatitis C Virus (HCV) Viral Load Testing market size has reached to $1.39 billion in 2025 • Expected to grow to $1.98 billion in 2030 at a compound annual growth rate (CAGR) of 7.4% • Growth Driver: Growth In Personalized Medicine Driving Demand For The Market Due To Advancements In Genomic Technologies And Targeted Therapies • Market Trend: Advancements In Point-Of-Care Molecular Viral Load Testing For Decentralized Hepatitis C Management • North America was the largest region in 2025 and Asia-Pacific is the fastest growing region.What Is Covered Under Hepatitis C Virus (HCV) Viral Load Testing Market?
Hepatitis C virus (HCV) viral load testing refers to a laboratory test that measures the quantity of hepatitis C virus (HCV RNA) in a patient’s bloodstream. It is used to confirm active infection and to monitor the effectiveness of antiviral therapy during and after treatment. The main technologies of hepatitis C virus (HCV) viral load testing include polymerase chain reaction, nucleic acid sequence-based amplification, transcription-mediated amplification, and others. Polymerase chain reaction (PCR) is a laboratory technique used to amplify specific DNA sequences, creating millions of copies from a small DNA sample. The various product types include instruments, kits and reagents, and software and services and support sample types such as blood, plasma, serum, and other sample types. The key end users include hospitals and clinics, diagnostic laboratories, blood banks, and other end users.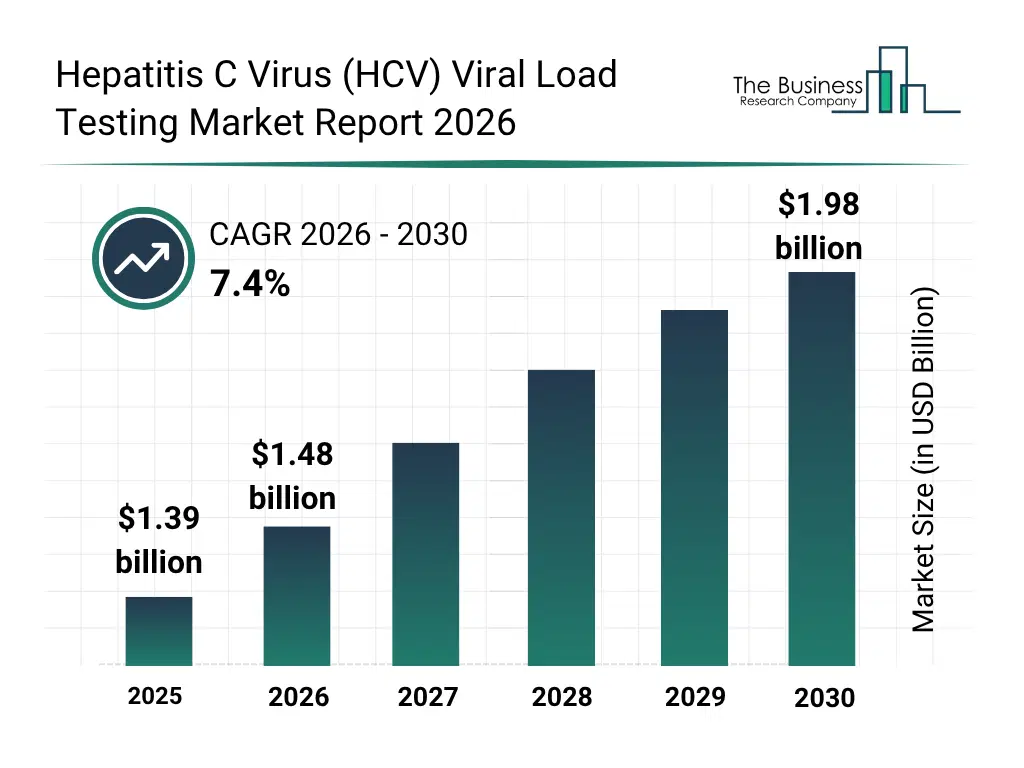
What Is The Hepatitis C Virus (HCV) Viral Load Testing Market Size and Share 2026?
The hepatitis c virus (hcv) viral load testing market size has grown strongly in recent years. It will grow from $1.39 billion in 2025 to $1.48 billion in 2026 at a compound annual growth rate (CAGR) of 7.2%. The growth in the historic period can be attributed to rising global prevalence of hepatitis c infections, increasing awareness of early diagnosis, expansion of diagnostic laboratory infrastructure, adoption of nucleic acid amplification technologies, growing availability of antiviral therapies.What Is The Hepatitis C Virus (HCV) Viral Load Testing Market Growth Forecast?
The hepatitis c virus (hcv) viral load testing market size is expected to see strong growth in the next few years. It will grow to $1.98 billion in 2030 at a compound annual growth rate (CAGR) of 7.4%. The growth in the forecast period can be attributed to increasing government screening programs, growing demand for precision viral load monitoring, expansion of decentralized testing facilities, rising investment in molecular diagnostics, technological advancements in high-sensitivity hcv rna assays. Major trends in the forecast period include increasing adoption of real-time PCR-based viral load testing, rising demand for early detection and monitoring of treatment response, growing use of automated high-throughput diagnostic platforms, expansion of point-of-care HCV RNA testing solutions, increasing standardization of viral load quantification assays.Global Hepatitis C Virus (HCV) Viral Load Testing Market Segmentation
1) By Technology: Polymerase Chain Reaction, Nucleic Acid Sequence-Based Amplification, Transcription-Mediated Amplification, Other Technologies 2) By Product Type: Instruments, Kits And Reagents, Software And Services 3) By Sample Type: Blood, Plasma, Serum, Other Sample Types 4) By End User: Hospitals And Clinics, Diagnostic Laboratories, Blood Banks, Other End Users Subsegments: 1) By Polymerase Chain Reaction: Real Time Polymerase Chain Reaction, Digital Polymerase Chain Reaction, Nested Polymerase Chain Reaction, Reverse Transcription Polymerase Chain Reaction 2) By Nucleic Acid Sequence-Based Amplification: Real Time Nucleic Acid Sequence-Based Amplification, Standard Nucleic Acid Sequence-Based Amplification 3) By Transcription-Mediated Amplification: Real Time Transcription-Mediated Amplification, Isothermal Transcription-Mediated Amplification 4) By Other Technologies: Branched Deoxyribonucleic Acid Assay, Signal Mediated Amplification Of Ribonucleic Acid, Hybrid Capture AssayWhat Is The Driver Of The Hepatitis C Virus (HCV) Viral Load Testing Market?
The growth of personalized medicine is expected to propel the growth of the hepatitis C virus (HCV) viral load testing market going forward. Personalized medicine refers to an approach to healthcare that customizes medical treatment and interventions based on an individual’s genetic makeup, lifestyle, and environmental factors to optimize clinical outcomes. The increasing adoption of personalized medicine is driven by advancements in genomic technologies, which enable precise disease profiling and targeted therapies that improve treatment outcomes while reducing side effects. Hepatitis C viral load testing supports personalized medicine by quantifying the amount of virus in a patient’s blood, enabling clinicians to tailor antiviral therapy selection, dosage, and treatment duration based on individual response and disease severity. For instance, in February 2024, according to the Personalized Medicine Coalition, a US-based non-profit organization, in 2023, the FDA approved 16 new personalized treatments for rare disease patients, up from six in 2022, including seven cancer drugs and three therapies for other diseases and conditions. Therefore, the growth of personalized medicine is driving the growth of the hepatitis C virus (HCV) viral load testing industry.Key Players In The Global Hepatitis C Virus (HCV) Viral Load Testing Market
Major companies operating in the hepatitis c virus (hcv) viral load testing market are F. Hoffmann-La Roche Ltd., Thermo Fisher Scientific Inc., Abbott Laboratories, Siemens Healthineers, Danaher Corporation, Hologic Inc., Revvity, Bio-Rad Laboratories Inc., Qiagen N.V., Cepheid Inc., BioPerfectus Technologies Co. Ltd., Enzo Biochem Inc., Vela Diagnostics International Pte. Ltd., Altona Diagnostics GmbH, Certest Biotec S.L., Molbio Diagnostics Private Limited, 3B BlackBio Dx Limited, Anatolia Geneworks, GeneProof a.s., Sacace Biotechnologies S.r.l.Global Hepatitis C Virus (HCV) Viral Load Testing Market Trends and Insights
Major companies operating in the hepatitis C virus (HCV) viral load testing market are focusing on obtaining regulatory approvals to expand access to rapid and highly accurate molecular diagnostic solutions. Regulatory approvals enable commercial distribution and clinical adoption of advanced nucleic acid amplification tests (NAATs), improving early diagnosis, treatment initiation, and disease monitoring. For instance, in August 2025, Cepheid Inc., a US-based molecular diagnostics company, obtained a medical device license from Health Canada for its Xpert HCV Viral Load test. This assay is designed to detect and quantify hepatitis C virus (HCV) RNA and is conducted on Cepheid’s GeneXpert System platform. The test delivers automated, real-time PCR-based results, enabling faster clinical decision-making, enhanced patient management, and broader access to decentralized HCV testing services.What Are Latest Mergers And Acquisitions In The Hepatitis C Virus (HCV) Viral Load Testing Market?
In May 2024, Bruker Corporation, a US-based manufacturer of scientific instruments and analytical solutions, completed the acquisition of ELITechGroup for an undisclosed amount. Through this acquisition, Bruker aims to strengthen its molecular diagnostics portfolio by integrating ELITechGroup’s innovative PCR-based infectious disease testing solutions and in vitro diagnostic assays, thereby expanding its presence in the clinical diagnostics and infectious disease testing market. ELITechGroup is a France-based molecular diagnostics company that offers HCV viral load testing.Regional Insights
North America was the largest region in the hcv viral load testing market in 2025. Asia-Pacific is expected to be the fastest-growing region in the forecast period. The regions covered in this market report are Asia-Pacific, South East Asia, Western Europe, Eastern Europe, North America, South America, Middle East, Africa. The countries covered in this market report are Australia, Brazil, China, France, Germany, India, Indonesia, Japan, Taiwan, Russia, South Korea, UK, USA, Canada, Italy, Spain.What Defines the Hepatitis C Virus (HCV) Viral Load Testing Market?
The hepatitis C virus (HCV) viral load testing market consists of revenues earned by entities by providing services such as baseline viral load assessment, treatment monitoring, and sustained virologic response (SVR) testing. The market value includes the value of related goods sold by the service provider or included within the service offering. The hepatitis C virus (HCV) viral load testing market also includes sales of genexpert systems, automated RNA extraction systems, thermal cyclers, and fluorescence detection units. Values in this market are ‘factory gate’ values; that is, the value of goods sold by the manufacturers or creators of the goods, whether to other entities (including downstream manufacturers, wholesalers, distributors, and retailers) or directly to end customers. The value of goods in this market includes related services sold by the creators of the goods.How is Market Value Defined and Measured?
The market value is defined as the revenues that enterprises gain from the sale of goods and/or services within the specified market and geography through sales, grants, or donations in terms of the currency (in USD unless otherwise specified). The revenues for a specified geography are consumption values that are revenues generated by organizations in the specified geography within the market, irrespective of where they are produced. It does not include revenues from resales along the supply chain, either further along the supply chain or as part of other products.What Key Data And Analysis Are Included In The Hepatitis C Virus (HCV) Viral Load Testing Market Report 2026?
The hepatitis c virus (hcv) viral load testing market research report is one of a series of new reports from The Business Research Company that provides market statistics, including industry global market size, regional shares, competitors with the market share, detailed market segments, market trends and opportunities, and any further data you may need to thrive in the hepatitis c virus (hcv) viral load testing industry. The market research report delivers a complete perspective of everything you need, with an in-depth analysis of the current and future state of the industry.Hepatitis C Virus (HCV) Viral Load Testing Market Report Forecast Analysis
| Report Attribute | Details |
|---|---|
| Market Size Value In 2026 | $1.48 billion |
| Revenue Forecast In 2035 | $1.98 billion |
| Growth Rate | CAGR of 7.2% from 2026 to 2035 |
| Base Year For Estimation | 2025 |
| Actual Estimates/Historical Data | 2020-2025 |
| Forecast Period | 2026 - 2030 - 2035 |
| Market Representation | Revenue in USD Billion and CAGR from 2026 to 2035 |
| Segments Covered | Technology, Product Type, Sample Type, End User |
| Regional Scope | Asia-Pacific, Western Europe, Eastern Europe, North America, South America, Middle East, Africa |
| Country Scope | The countries covered in the report are Australia, Brazil, China, France, Germany, India, Indonesia, Japan, Taiwan, Russia, South Korea, UK, USA, Canada, Italy, Spain. |
| Key Companies Profiled | F. Hoffmann-La Roche Ltd., Thermo Fisher Scientific Inc., Abbott Laboratories, Siemens Healthineers, Danaher Corporation, Hologic Inc., Revvity, Bio-Rad Laboratories Inc., Qiagen N.V., Cepheid Inc., BioPerfectus Technologies Co. Ltd., Enzo Biochem Inc., Vela Diagnostics International Pte. Ltd., Altona Diagnostics GmbH, Certest Biotec S.L., Molbio Diagnostics Private Limited, 3B BlackBio Dx Limited, Anatolia Geneworks, GeneProof a.s., Sacace Biotechnologies S.r.l. |
| Customization Scope | Request for Customization |
| Pricing And Purchase Options | Explore Purchase Options |
