
Hernia Mesh Devices Market Report 2026
Global Outlook – By Mesh Type (Biologic Mesh, Synthetic Mesh), By Procedure (Open Surgeries, Laparoscopic Surgeries, Robotic Surgeries), By Hernia Type (Incisional Hernia, Umbilical Hernia, Inguinal Hernia, Femoral Hernia, Other Hernia Types), By End User (Hospitals, Clinics, Ambulatory Surgical Centers, Other End Users) – Market Size, Trends, Strategies, and Forecast to 2035
Hernia Mesh Devices Market Overview
• Hernia Mesh Devices market size has reached to $4.51 billion in 2025 • Expected to grow to $5.73 billion in 2030 at a compound annual growth rate (CAGR) of 4.9% • Growth Driver: Obesity Fuels Expansion Of Hernia Mesh Devices Market • Market Trend: Technological Advancements Driving Innovation In Hernia Mesh Devices • North America was the largest region in 2025 and Asia-Pacific is the fastest growing region.What Is Covered Under Hernia Mesh Devices Market?
A hernia mesh device is a medical implant or surgical mesh used to treat hernias. A hernia occurs when an organ or fatty tissue protrudes through a weak spot or opening in the surrounding muscle or connective tissue, typically in the abdominal wall. Hernia mesh devices are used for hernia repair surgeries to support damaged tissue and strengthen the abdominal wall, reducing the risk of recurrence. The main types of hernia mesh devices are biologic mesh and synthetic mesh. Biologic mesh, also known as bioprosthetic mesh or biological mesh, is a type of surgical mesh used in hernia repair and other soft tissue surgeries. They are used in various procedures such as open surgeries, laparoscopic surgeries, and robotic surgeries of incisional hernia, umbilical hernia, inguinal hernia, femoral hernia, and others by hospitals, clinics, ambulatory surgical centers, and other end users.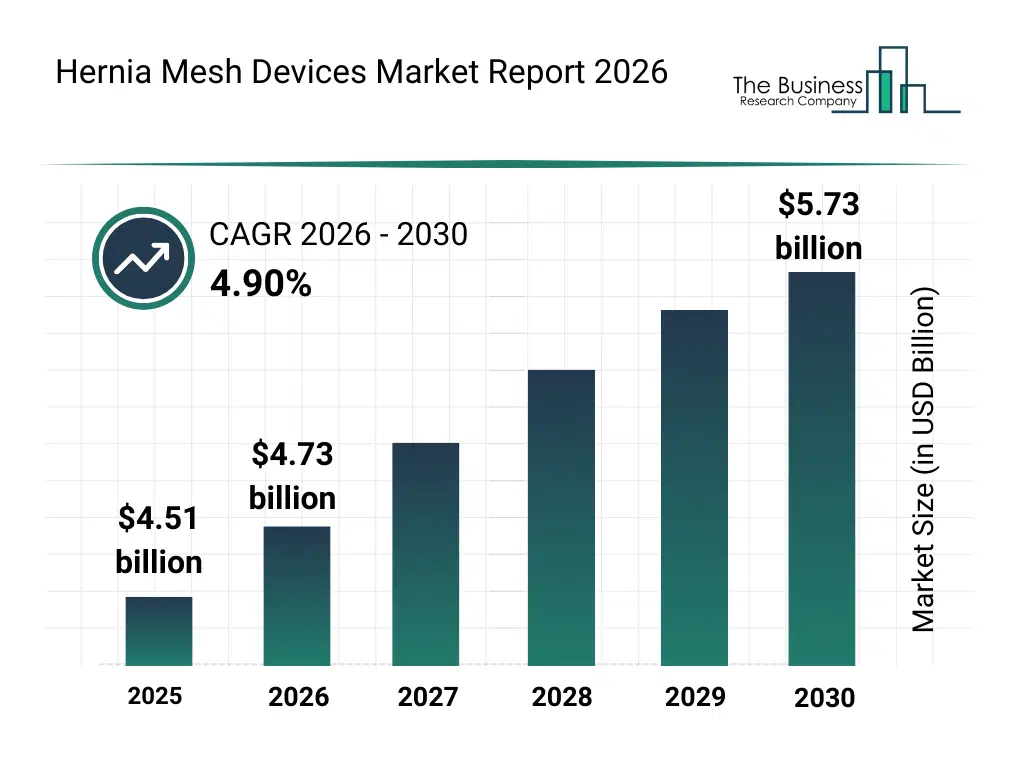
What Is The Hernia Mesh Devices Market Size and Share 2026?
The hernia mesh devices market size has grown steadily in recent years. It will grow from $4.51 billion in 2025 to $4.73 billion in 2026 at a compound annual growth rate (CAGR) of 4.9%. The growth in the historic period can be attributed to increasing prevalence of abdominal and inguinal hernias, rising number of surgical hernia repair procedures, wider adoption of synthetic mesh products, improvements in surgical techniques, expansion of hospital surgical infrastructure.What Is The Hernia Mesh Devices Market Growth Forecast?
The hernia mesh devices market size is expected to see steady growth in the next few years. It will grow to $5.73 billion in 2030 at a compound annual growth rate (CAGR) of 4.9%. The growth in the forecast period can be attributed to increasing demand for advanced biocompatible mesh solutions, rising adoption of robotic-assisted hernia surgeries, growing focus on patient-specific mesh designs, expansion of outpatient surgical procedures, increasing investments in surgical innovation. Major trends in the forecast period include increasing adoption of lightweight and composite mesh materials, rising preference for minimally invasive hernia repair solutions, growing use of biologic mesh in complex hernia cases, expansion of laparoscopic and robotic hernia procedures, enhanced focus on reduced recurrence and infection rates.Global Hernia Mesh Devices Market Segmentation
1) By Mesh Type: Biologic Mesh, Synthetic Mesh 2) By Procedure: Open Surgeries, Laparoscopic Surgeries, Robotic Surgeries 3) By Hernia Type: Incisional Hernia, Umbilical Hernia, Inguinal Hernia, Femoral Hernia, Other Hernia Types 4) By End User: Hospitals, Clinics, Ambulatory Surgical Centers, Other End Users Subsegments: 1) By Biologic Mesh: Human-Derived Biologic Mesh, Animal-Derived Biologic Mesh 2) By Synthetic Mesh: Polyester Mesh, Polypropylene Mesh, ePTFE (Expanded Polytetrafluoroethylene) Mesh, Composites MeshWhat Is The Driver Of The Hernia Mesh Devices Market?
The growing obese population is expected to propel the growth of the hernia mesh devices market going forward. Obesity refers to the abnormal or excessive accumulation of fat that impairs the health of individuals and increases the likelihood of abdominal wall weakness. The prevalence of obesity is rising as more people adopt sedentary lifestyles and unhealthy dietary habits, contributing to higher hernia risks. Hernia mesh devices support obese individuals by offering stronger, more durable reinforcement, reducing recurrence rates and improving recovery compared to traditional suture repairs. For instance, in May 2025, according to the Office for Health Improvement & Disparities, a UK-based government department, between 2023 and 2024, an estimated 64.5% of adults aged 18 and over in England were overweight or living with obesity, up slightly from 64.0% in 2022 to 2023. Therefore, the growing obese population drives the growth of the hernia mesh devices industry.Key Players In The Global Hernia Mesh Devices Market
Major companies operating in the hernia mesh devices market are Johnson & Johnson Services Inc., Medtronic plc, Betatech Medical, Ethicon Inc., B. Braun Melsungen AG, W.L. Gore & Associates Inc., Cook Medical Inc., Samyang Corporation, Integra Life Sciences Pvt. Ltd., LifeCell Corporation, Meril Life Sciences Pvt Ltd., Sinolinks Medical Innovation Inc, Aspide Medical S.A.S, C.R. Bard Inc., Bard Inc., Deep Blue Medical Inc., Changzhou Medical Equipment General Factory Co. Ltd., Katsan Katgut Sanayi ve Tic. A.S., BioCer Entwicklungs-GmbH, Dipromed S.r.l., Herniamesh S.r.l., Novus Scientific AB, Gem S.r.l., Primequal SAGlobal Hernia Mesh Devices Market Trends and Insights
Major companies operating in the hernia mesh devices market are focusing on technological innovation, such as tri-layer hybrid preperitoneal biomaterials, which combine permanent synthetic strength with biointegrative design to enhance reinforcement, reduce recurrence risk, and improve handling during minimally invasive and open hernia repair procedures. Tri-layer hybrid preperitoneal biomaterials integrate multiple material layers to provide durable support, fluid absorption, and conformability, enabling surgeons to achieve secure placement and effective bridging in complex hernia repairs. For instance, in April 2025, W. L. Gore & Associates, a US-based medical materials and device company, launched its GORE SYNECOR Preperitoneal Biomaterial across Europe, the Middle East, and South Africa. The product offers flexible, conformable construction with material memory for easy unrolling, requires no pre-soaking, and supports laparoscopic, robotic, and open procedures—expanding the company’s portfolio with a unique option designed to minimize hernia recurrence and streamline surgical handling.What Are Latest Mergers And Acquisitions In The Hernia Mesh Devices Market?
In May 2024, Kohlberg Kravis Roberts & Co. Inc. (KKR), a US-based investment firm, has acquired Healthium MedTech for an undisclosed sum. This acquisition allows KKR to utilize its extensive network and expertise in healthcare to enhance Healthium's product offerings and expand their global presence. The strategic move aims to support the development of advanced hernia repair devices, ensuring they meet high standards of safety and efficacy through significant investment in research and development. Healthium, an Indian-based medical devices company, known for its wide range of surgical products, including hernia mesh devices, which are crucial for hernia repair surgeries.Regional Outlook
North America was the largest region in the hernia mesh devices market in 2025. Asia-Pacific is expected to be the fastest-growing region in the forecast period. The regions covered in this market report are Asia-Pacific, South East Asia, Western Europe, Eastern Europe, North America, South America, Middle East, Africa. The countries covered in this market report are Australia, Brazil, China, France, Germany, India, Indonesia, Japan, Taiwan, Russia, South Korea, UK, USA, Canada, Italy, SpainWhat Defines the Hernia Mesh Devices Market?
The hernia mesh devices market consists of sales of synthetic mesh, composite mesh, absorbable mesh, mesh with coating, 3D meshes, and mesh kits. Values in this market are ‘factory gate’ values, that is the value of goods sold by the manufacturers or creators of the goods, whether to other entities (including downstream manufacturers, wholesalers, distributors, and retailers) or directly to end customers. The value of goods in this market includes related services sold by the creators of the goods.How is Market Value Defined and Measured?
The market value is defined as the revenues that enterprises gain from the sale of goods and/or services within the specified market and geography through sales, grants, or donations in terms of the currency (in USD unless otherwise specified). The revenues for a specified geography are consumption values that are revenues generated by organizations in the specified geography within the market, irrespective of where they are produced. It does not include revenues from resales along the supply chain, either further along the supply chain or as part of other products.Hernia Mesh Devices Market Report Forecast Analysis
| Report Attribute | Details |
|---|---|
| Market Size Value In 2026 | $4.73 billion |
| Revenue Forecast In 2035 | $5.73 billion |
| Growth Rate | CAGR of 4.9% from 2026 to 2035 |
| Base Year For Estimation | 2025 |
| Actual Estimates/Historical Data | 2020-2025 |
| Forecast Period | 2026 - 2030 - 2035 |
| Market Representation | Revenue in USD Billion and CAGR from 2026 to 2035 |
| Segments Covered | Mesh Type, Procedure, Hernia Type, End User |
| Regional Scope | Asia-Pacific, Western Europe, Eastern Europe, North America, South America, Middle East, Africa |
| Country Scope | The countries covered in the report are Australia, Brazil, China, France, Germany, India, ... |
| Key Companies Profiled | Johnson & Johnson Services Inc., Medtronic plc, Betatech Medical, Ethicon Inc., B. Braun Melsungen AG, W.L. Gore & Associates Inc., Cook Medical Inc., Samyang Corporation, Integra Life Sciences Pvt. Ltd., LifeCell Corporation, Meril Life Sciences Pvt Ltd., Sinolinks Medical Innovation Inc, Aspide Medical S.A.S, C.R. Bard Inc., Bard Inc., Deep Blue Medical Inc., Changzhou Medical Equipment General Factory Co. Ltd., Katsan Katgut Sanayi ve Tic. A.S., BioCer Entwicklungs-GmbH, Dipromed S.r.l., Herniamesh S.r.l., Novus Scientific AB, Gem S.r.l., Primequal SA |
| Customization Scope | Request for Customization |
| Pricing And Purchase Options | Explore Purchase Options |
