
Herpes Simplex Virus (HSV) Polymerase Chain Reaction (PCR) Testing Market Report 2026
Global Outlook – By Test Type (Polymerase Chain Reaction Testing, Serology Testing, Culture Testing), By Product Type (Instruments, Reagents And Kits, Software), By Sample Type (Vesicular Swabs or Fluid Samples, Cerebrospinal Fluid Samples, Blood or Serum Samples), By Application (Clinical Diagnostics, Research, Blood Screening, Other Applications), By End User (Hospitals And Clinics, Diagnostic Laboratories, Research Institutes, Other End Users) – Market Size, Trends, Strategies, and Forecast to 2035
Herpes Simplex Virus (HSV) Polymerase Chain Reaction (PCR) Testing Market Overview
• Herpes Simplex Virus (HSV) Polymerase Chain Reaction (PCR) Testing market size has reached to $1.18 billion in 2025 • Expected to grow to $1.75 billion in 2030 at a compound annual growth rate (CAGR) of 8.2% • Growth Driver: Surge In Adoption Of Precision Medicine Fueling The Growth Of The Market Due To Improved Treatment Outcomes And Personalized Therapy Selection • Market Trend: Innovative Point-Of-Care Molecular Diagnostics For Rapid HSV Detection • North America was the largest region in 2025 and Asia-Pacific is the fastest growing region.What Is Covered Under Herpes Simplex Virus (HSV) Polymerase Chain Reaction (PCR) Testing Market?
Herpes simplex virus (HSV) polymerase chain reaction (PCR) testing is a molecular diagnostic method that detects HSV DNA in clinical samples such as blood, swabs, or cerebrospinal fluid. It is used to provide rapid, highly sensitive, and specific identification of HSV infections, including genital, oral, or neonatal cases. It enables early diagnosis, guides antiviral treatment decisions, and helps prevent the spread of infection. The main test types of herpes simplex virus (HSV) polymerase chain reaction (PCR) testing include polymerase chain reaction testing, serology testing, and culture testing. Polymerase chain reaction (PCR) testing is a diagnostic method that amplifies and detects specific DNA or RNA sequences to identify pathogens or genetic material with high sensitivity and accuracy. The various product types include instruments, reagents and kits, and software and are used for sample types that include vesicular swabs or fluid samples, cerebrospinal fluid samples, and blood or serum samples. They find application in clinical diagnostics, research, blood screening, and other applications, serving end users including hospitals and clinics, diagnostic laboratories, research institutes, and other end users.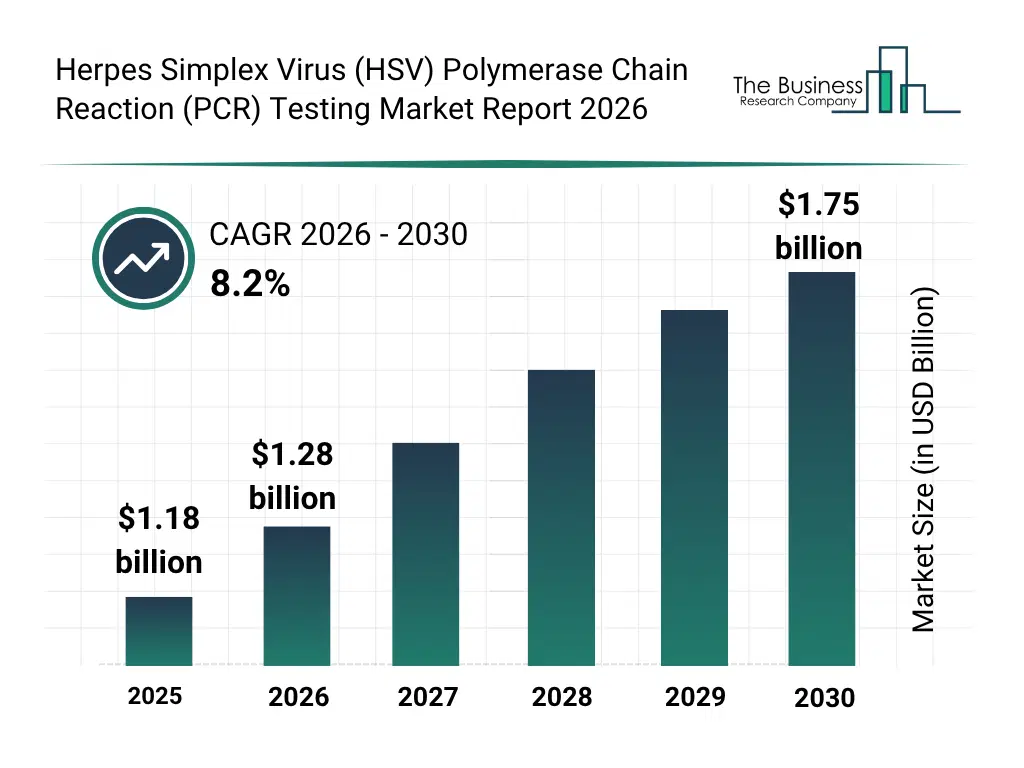
What Is The Herpes Simplex Virus (HSV) Polymerase Chain Reaction (PCR) Testing Market Size and Share 2026?
The herpes simplex virus (HSV) polymerase chain reaction (PCR) testing market size has grown strongly in recent years. It will grow from $1.18 billion in 2025 to $1.28 billion in 2026 at a compound annual growth rate (CAGR) of 8.0%. The growth in the historic period can be attributed to rising global prevalence of hsv infections, increasing awareness of sexually transmitted infections, expansion of molecular diagnostic laboratories, growing adoption of nucleic acid amplification tests, increasing availability of antiviral therapies .What Is The Herpes Simplex Virus (HSV) Polymerase Chain Reaction (PCR) Testing Market Growth Forecast?
The herpes simplex virus (HSV) polymerase chain reaction (PCR) testing market size is expected to see strong growth in the next few years. It will grow to $1.75 billion in 2030 at a compound annual growth rate (CAGR) of 8.2%. The growth in the forecast period can be attributed to growing implementation of routine hsv screening programs, rising demand for rapid point-of-care molecular testing, expansion of precision medicine approaches in infectious diseases, increasing healthcare investments in emerging markets, technological advancements in multiplex and quantitative pcr platforms. Major trends in the forecast period include increasing adoption of real-time pcr for rapid hsv detection, rising demand for early diagnosis in neonatal and genital hsv cases, growing use of Multiplex PCR Assays for differential diagnosis, expansion of high-sensitivity quantitative pcr testing, increasing integration of automated sample-to-result platforms.Global Herpes Simplex Virus (HSV) Polymerase Chain Reaction (PCR) Testing Market Segmentation
1) By Test Type: Polymerase Chain Reaction Testing, Serology Testing, Culture Testing 2) By Product Type: Instruments, Reagents And Kits, Software 3) By Sample Type: Vesicular Swabs or Fluid Samples, Cerebrospinal Fluid Samples, Blood or Serum Samples 4) By Application: Clinical Diagnostics, Research, Blood Screening, Other Applications 5) By End User: Hospitals And Clinics, Diagnostic Laboratories, Research Institutes, Other End Users Subsegments: 1) By Polymerase Chain Reaction Testing: Real Time Polymerase Chain Reaction, Nested Polymerase Chain Reaction, Multiplex Polymerase Chain Reaction, Quantitative Polymerase Chain Reaction 2) By Serology Testing: IgM Antibody Testing, IgG Antibody Testing, Western Blot Testing, Enzyme Linked Immunosorbent Assay 3) By Culture Testing: Cell Culture Isolation, Plaque Assay Culture, Shell Vial Culture, Tissue Culture Infection AssayWhat Is The Driver Of The Herpes Simplex Virus (HSV) Polymerase Chain Reaction (PCR) Testing Market?
The rising incidence of infections is expected to propel the growth of the herpes simplex virus (HSV) polymerase chain reaction (PCR) testing market going forward. Infections are the invasion and growth of harmful microorganisms in the body that can cause illness. Infections are increasing due to urbanization, as higher population density in cities facilitates faster spread of pathogens through close human contact and shared resources. Herpes simplex virus (HSV) polymerase chain reaction (PCR) testing helps by detecting the presence of HSV DNA quickly and accurately, enabling early diagnosis and treatment, which prevents the infection from worsening or spreading to others. For instance, in 2023, according to the UK Health Security Agency, a UK-based government agency, the rate of yeast-related bloodstream infections across England rose by 4% compared to 2022. Therefore, the rising incidence of infections is driving the growth of the herpes simplex virus (HSV) polymerase chain reaction (PCR) testing industry.Key Players In The Global Herpes Simplex Virus (HSV) Polymerase Chain Reaction (PCR) Testing Market
Major companies operating in the herpes simplex virus (HSV) polymerase chain reaction (PCR) testing market are F. Hoffmann-La Roche Ltd., Thermo Fisher Scientific Inc., Abbott Laboratories, Danaher Corporation, Becton, Dickinson Company, Quest Diagnostics Incorporated, Hologic Inc., bioMérieux SA, Bio-Rad Laboratories Inc., QIAGEN N.V., DiaSorin S.p.A., Promega Corporation, Meridian Bioscience Inc., Seegene Inc., BioPerfectus Technologies Co. Ltd., altona Diagnostics GmbH, Xi’an Tianlong Science & Technology Co. Ltd., 3B BlackBio Dx Limited, GeneProof s.r.o., Primerdesign Ltd., BioHelix Corporation.Global Herpes Simplex Virus (HSV) Polymerase Chain Reaction (PCR) Testing Market Trends and Insights
Major companies operating in the herpes simplex virus (HSV) polymerase chain reaction (PCR) testing market are focusing on developing innovative solutions such as, point-of-care molecular diagnostics to enable rapid, accurate, and decentralized detection of HSV infections for improved patient management. Point-of-care molecular diagnostics refers to tests that detect pathogens rapidly and accurately at the site of patient care without requiring a central laboratory. For instance, in January 2023, Molbio Diagnostics Pvt. Ltd., an India-based medical technology company, launched a Truenat HSV 1/2 chip-based RT-PCR test for diagnosing herpes simplex virus types 1 and 2. This portable, point-of-care assay delivers results in under one hour from genital swabs or cerebrospinal fluid (CSF) using the battery-operated Truelab platform, requiring no additional equipment and holding central drugs standard control organisation (CDSCO) approval in India. It supports rapid detection of active herpes simplex virus (HSV) infections to boost awareness and early treatment amid rising cases.What Are Latest Mergers And Acquisitions In The Herpes Simplex Virus (HSV) Polymerase Chain Reaction (PCR) Testing Market?
In May 2024, Bruker Corporation, a US-based scientific instruments and diagnostics company, acquired ELITech Group for an undisclosed amount. Through this acquisition, Bruker aims to expand its presence in polymerase chain reaction (PCR) molecular diagnostics and strengthen its clinical laboratory diagnostic product portfolio by leveraging ELITech Group’s molecular diagnostic systems, infectious disease assays, and microbiology solutions to enhance Bruker’s market position and service offerings for clinical laboratories. ELITech Group is a France-based molecular diagnostic company that offers herpes simplex virus (HSV) polymerase chain reaction (PCR) testing solutions.Regional Insights
North America was the largest region in the herpes simplex virus (HSV) polymerase chain reaction (PCR) testing market in 2025. Asia-Pacific is expected to be the fastest-growing region in the forecast period. The regions covered in this market report are Asia-Pacific, South East Asia, Western Europe, Eastern Europe, North America, South America, Middle East, Africa. The countries covered in this market report are Australia, Brazil, China, France, Germany, India, Indonesia, Japan, Taiwan, Russia, South Korea, UK, USA, Canada, Italy, Spain.What Defines the Herpes Simplex Virus (HSV) Polymerase Chain Reaction (PCR) Testing Market?
The herpes simplex virus (HSV) polymerase chain reaction (PCR) testing market consists of revenues earned by entities by providing services such as quantitative viral load analysis, confirmation of suspected cases, prenatal and neonatal screening, and monitoring of treatment efficacy. The market value includes the value of related goods sold by the service provider or included within the service offering. The herpes simplex virus (HSV) polymerase chain reaction (PCR) testing market also includes sales of control and calibration materials, viral transport media, swabs and sample collection devices, automated nucleic acid extraction systems, microfluidic cartridges, and laboratory automation accessories. Values in this market are ‘factory gate’ values, that is the value of goods sold by the manufacturers or creators of the goods, whether to other entities (including downstream manufacturers, wholesalers, distributors and retailers) or directly to end customers. The value of goods in this market includes related services sold by the creators of the goods.How is Market Value Defined and Measured?
The market value is defined as the revenues that enterprises gain from the sale of goods and/or services within the specified market and geography through sales, grants, or donations in terms of the currency (in USD unless otherwise specified). The revenues for a specified geography are consumption values that are revenues generated by organizations in the specified geography within the market, irrespective of where they are produced. It does not include revenues from resales along the supply chain, either further along the supply chain or as part of other products.What Key Data And Analysis Are Included In The Herpes Simplex Virus (HSV) Polymerase Chain Reaction (PCR) Testing Market Report 2026?
The herpes simplex virus (hsv) polymerase chain reaction (pcr) testing market research report is one of a series of new reports from The Business Research Company that provides market statistics, including industry global market size, regional shares, competitors with the market share, detailed market segments, market trends and opportunities, and any further data you may need to thrive in the herpes simplex virus (hsv) polymerase chain reaction (pcr) testing industry. The market research report delivers a complete perspective of everything you need, with an in-depth analysis of the current and future state of the industry.Herpes Simplex Virus (HSV) Polymerase Chain Reaction (PCR) Testing Market Report Forecast Analysis
| Report Attribute | Details |
|---|---|
| Market Size Value In 2026 | $1.28 billion |
| Revenue Forecast In 2035 | $1.75 billion |
| Growth Rate | CAGR of 8.0% from 2026 to 2035 |
| Base Year For Estimation | 2025 |
| Actual Estimates/Historical Data | 2020-2025 |
| Forecast Period | 2026 - 2030 - 2035 |
| Market Representation | Revenue in USD Billion and CAGR from 2026 to 2035 |
| Segments Covered | Test Type, Product Type, Sample Type, Application, End User |
| Regional Scope | Asia-Pacific, Western Europe, Eastern Europe, North America, South America, Middle East, Africa |
| Country Scope | The countries covered in the report are Australia, Brazil, China, France, Germany, India, Indonesia, Japan, Taiwan, Russia, South Korea, UK, USA, Canada, Italy, Spain. |
| Key Companies Profiled | F. Hoffmann-La Roche Ltd., Thermo Fisher Scientific Inc., Abbott Laboratories, Danaher Corporation, Becton, Dickinson Company, Quest Diagnostics Incorporated, Hologic Inc., bioMérieux SA, Bio-Rad Laboratories Inc., QIAGEN N.V., DiaSorin S.p.A., Promega Corporation, Meridian Bioscience Inc., Seegene Inc., BioPerfectus Technologies Co. Ltd., altona Diagnostics GmbH, Xi’an Tianlong Science & Technology Co. Ltd., 3B BlackBio Dx Limited, GeneProof s.r.o., Primerdesign Ltd., BioHelix Corporation. |
| Customization Scope | Request for Customization |
| Pricing And Purchase Options | Explore Purchase Options |
