
Home Spirometry Bluetooth Device Market Report 2026
Global Outlook – By Product Type (Handheld Home Spirometry Devices, Compact Tabletop Home Spirometry Devices), By Connectivity (Bluetooth-Enabled Devices, Bluetooth + Wi-Fi Enabled Devices), By Distribution Channel (Online, Offline), By Application (Chronic Obstructive Pulmonary Disease (COPD), Asthma, Cystic Fibrosis, Other Applications), By End-User (Homecare Settings, Patients, Hospitals and Clinics, Other End-Users) – Market Size, Trends, Strategies, and Forecast to 2035
Home Spirometry Bluetooth Device Market Overview
• Home Spirometry Bluetooth Device market size has reached to $1.28 billion in 2025 • Expected to grow to $2.08 billion in 2030 at a compound annual growth rate (CAGR) of 10.2% • Growth Driver: Increasing Prevalence Of Chronic Respiratory Diseases Fuelling The Growth Of The Market Amid Growing Respiratory Health Challenges • Market Trend: Advancing Home-Based, Bluetooth-Enabled Lung Health Monitoring Boosting Lung Care • North America was the largest region in 2025 and Asia-Pacific is the fastest growing region.What Is Covered Under Home Spirometry Bluetooth Device Market?
A home spirometry Bluetooth device is a medical instrument that measures lung function and airflow capacity. It captures respiratory data in real time and transmits it wirelessly via Bluetooth to connected devices. The device enables continuous monitoring of breathing patterns, enhancing proactive respiratory health management. Its core advantage lies in providing accurate, instant, and easily accessible pulmonary measurements at home. The main types of home spirometry Bluetooth devices are handheld devices and tabletop devices. Handheld devices are portable spirometers designed for at-home monitoring of lung function with Bluetooth connectivity. The devices are available with several connectivity options, including Bluetooth, wireless fidelity (Wi-Fi), and others, and are distributed through multiple distribution channels, such as online and offline. It is used for various applications, including chronic obstructive pulmonary disease (COPD), asthma, cystic fibrosis, and others, and the end-users are hospitals and clinics, home care settings, ambulatory surgical centers, and others.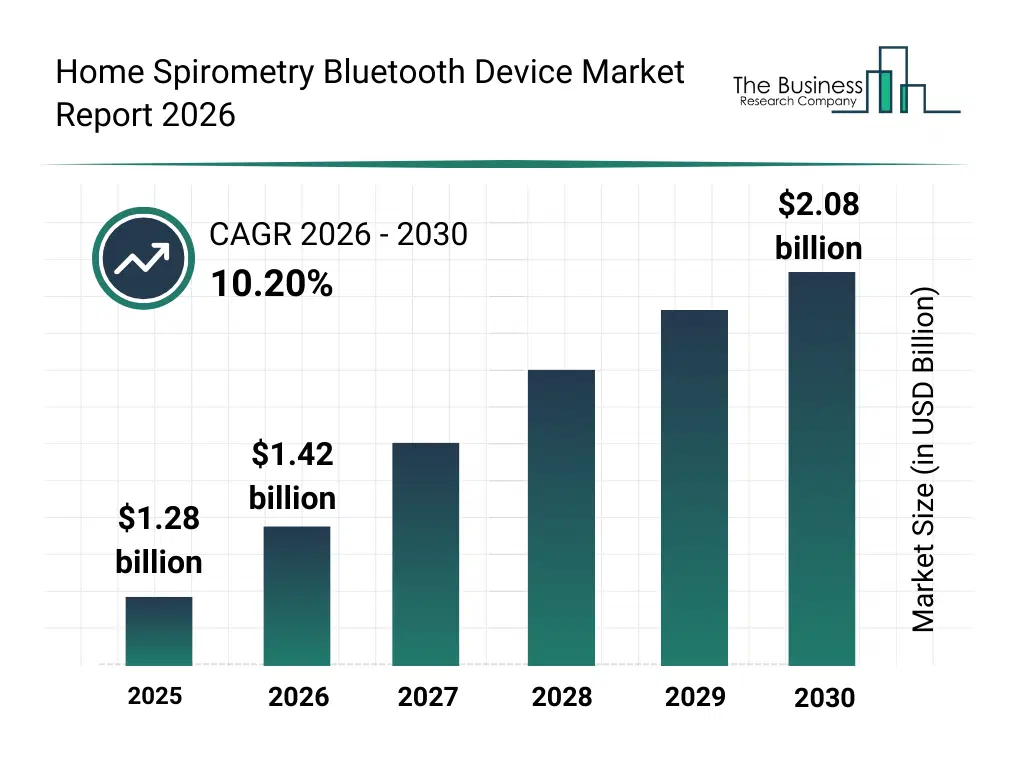
What Is The Home Spirometry Bluetooth Device Market Size and Share 2026?
The home spirometry bluetooth device market size has grown rapidly in recent years. It will grow from $1.28 billion in 2025 to $1.42 billion in 2026 at a compound annual growth rate (CAGR) of 10.5%. The growth in the historic period can be attributed to rise in chronic respiratory diseases, early adoption of home health devices, advancement in Bluetooth connectivity, increasing patient self-monitoring awareness, integration with hospital systems.What Is The Home Spirometry Bluetooth Device Market Growth Forecast?
The home spirometry bluetooth device market size is expected to see rapid growth in the next few years. It will grow to $2.08 billion in 2030 at a compound annual growth rate (CAGR) of 10.2%. The growth in the forecast period can be attributed to AI-powered predictive respiratory analytics, expansion of remote healthcare programs, integration with telemedicine platforms, adoption by homecare providers, growing demand for continuous pulmonary monitoring. Major trends in the forecast period include wireless pulmonary data transmission, portable home monitoring devices, real-time lung function tracking, integration with mobile health apps, patient-adherence monitoring.Global Home Spirometry Bluetooth Device Market Segmentation
1) By Product Type: Handheld Home Spirometry Devices, Compact Tabletop Home Spirometry Devices 2) By Connectivity: Bluetooth-Enabled Devices, Bluetooth + Wi-Fi Enabled Devices 3) By Distribution Channel: Online, Offline 4) By Application: Chronic Obstructive Pulmonary Disease (COPD), Asthma, Cystic Fibrosis, Other Applications 5) By End-User: Homecare Settings, Patients, Hospitals and Clinics, Other End-Users Subsegments: 1) By Handheld Home Spirometry Devices: Portable Bluetooth Spirometers, Pocket-Size Spirometers, Smartphone-Integrated Spirometers, Rechargeable Handheld Spirometers, Battery-Operated Handheld Spirometers 2) By Compact Tabletop Home Spirometry Devices: Wireless Bluetooth Tabletop Spirometers, Digital Home Diagnostic Spirometers, Multi-Parameter Home Monitoring SpirometersWhat Is The Driver Of The Home Spirometry Bluetooth Device Market?
The increasing prevalence of chronic respiratory diseases is expected to propel the growth of the home spirometry Bluetooth device market going forward. Chronic respiratory diseases refer to long-term conditions that affect the airways and other structures of the lungs, leading to reduced lung function and decreased overall respiratory health. The prevalence of chronic respiratory diseases is increasing mainly due to the aging population, as older individuals face a higher risk of respiratory conditions from natural declines in lung function and long-term exposure to environmental irritants. Home spirometry Bluetooth devices allow real-time remote monitoring of lung function in patients with chronic respiratory diseases, providing early detection of worsening conditions to enable timely medical interventions and better long-term management. For instance, in May 2025, according to the Centers for Disease Control and Prevention, a US-based government public health agency, chronic obstructive pulmonary disease (COPD) was reported as the fifth leading cause of death in the United States in 2023, resulting in 141,733 deaths. In the same year, the age-adjusted prevalence of diagnosed COPD in adults aged 18 years and older was 3.8 percent. Therefore, the increasing prevalence of chronic respiratory diseases is driving the growth of the home spirometry Bluetooth device industry.Key Players In The Global Home Spirometry Bluetooth Device Market
Major companies operating in the home spirometry bluetooth device market are Contec Medical Systems Co. Ltd., Vitalograph Ltd., Medcaptain Medical Technology Co. Ltd., Medical International Research S.p.A., CareSimple Inc., Geratherm Medical AG, SDI Diagnostics Inc., NDD Medical Technologies, Futuremed America Inc., Sibelmed S.L., Uscom Ltd., e-LinkCare Meditech Co. Ltd., Monitored Therapeutics Inc. (MTI), Aluna Health Inc., Jones Medical Instrument Company, CMI Health, Healthup SA, Motustech Inc., Medikro Oy, Icen Technology Company LimitedGlobal Home Spirometry Bluetooth Device Market Trends and Insights
Major companies operating in the home spirometry Bluetooth device market are focusing on developing innovative solutions, such as Air Next Spirometer supporting remote patient management and early detection of respiratory conditions such as asthma and COPD. The Air Next Spirometer is a Bluetooth-enabled, smartphone-connected home spirometry device designed to accurately measure lung function and transmit real-time respiratory data for remote monitoring and management. For instance, in January 2024, NuvoAir Medical, a US-based company that offers personalized heart and lung care announced that its Air Next Spirometer has received formal 510(k) clearance from the U.S. Food and Drug Administration (FDA) for in-home use as a full spirometer. This clearance means that the device is approved for use by patients at home, providing a comprehensive lung function assessment similar to those conducted in clinical settings. It enables patients to conduct various lung function tests such as forced expiratory volume (FEV1) and forced vital capacity (FVC) at home, with data transmitted to healthcare providers for ongoing management. This approval also supports clinical trials, research, and regular monitoring of lung health outside traditional healthcare facilities, promoting more accessible and continuous respiratory care.What Are Latest Mergers And Acquisitions In The Home Spirometry Bluetooth Device Market?
In January 2023, CAIRE Inc., a US-based manufacturer of oxygen therapy and respiratory care solutions, acquired MGC Diagnostics Holdings, Inc. for an undisclosed amount. The acquisition aims to expand CAIRE’s portfolio into cardiorespiratory diagnostic technologies and enhance patient care across awareness, diagnosis, therapy, and monitoring. This move also leverages CAIRE’s global network to improve access and strengthen its presence in the pulmonary and cardiorespiratory clinical market. MGC Diagnostics Holdings, Inc. is a US-based manufacturer of Bluetooth-enabled home spirometers such as the GoSpiro.Regional Outlook
North America was the largest region in the home spirometry bluetooth device market in 2025. Asia-Pacific is expected to be the fastest-growing region in the forecast period. The regions covered in this market report are Asia-Pacific, South East Asia, Western Europe, Eastern Europe, North America, South America, Middle East, Africa. The countries covered in this market report are Australia, Brazil, China, France, Germany, India, Indonesia, Japan, Taiwan, Russia, South Korea, UK, USA, Canada, Italy, SpainWhat Defines the Home Spirometry Bluetooth Device Market?
The home spirometry Bluetooth device market consists of sales of spirometers with Bluetooth, peak expiratory flow meters (PEF) with Bluetooth, portable spirometers, and lung function monitoring devices. Values in this market are ‘factory gate’ values, that is, the value of goods sold by the manufacturers or creators of the goods, whether to other entities (including downstream manufacturers, wholesalers, distributors, and retailers) or directly to end customers. The value of goods in this market includes related services sold by the creators of the goods.How is Market Value Defined and Measured?
The market value is defined as the revenues that enterprises gain from the sale of goods and/or services within the specified market and geography through sales, grants, or donations in terms of the currency (in USD unless otherwise specified). The revenues for a specified geography are consumption values that are revenues generated by organizations in the specified geography within the market, irrespective of where they are produced. It does not include revenues from resales along the supply chain, either further along the supply chain or as part of other products.Home Spirometry Bluetooth Device Market Report Forecast Analysis
| Report Attribute | Details |
|---|---|
| Market Size Value In 2026 | $1.42 billion |
| Revenue Forecast In 2035 | $2.08 billion |
| Growth Rate | CAGR of 10.5% from 2026 to 2035 |
| Base Year For Estimation | 2025 |
| Actual Estimates/Historical Data | 2020-2025 |
| Forecast Period | 2026 - 2030 - 2035 |
| Market Representation | Revenue in USD Billion and CAGR from 2026 to 2035 |
| Segments Covered | Product Type, Connectivity, Distribution Channel, Application, End-User |
| Regional Scope | Asia-Pacific, Western Europe, Eastern Europe, North America, South America, Middle East, Africa |
| Country Scope | The countries covered in the report are Australia, Brazil, China, France, Germany, India, ... |
| Key Companies Profiled | Contec Medical Systems Co. Ltd., Vitalograph Ltd., Medcaptain Medical Technology Co. Ltd., Medical International Research S.p.A., CareSimple Inc., Geratherm Medical AG, SDI Diagnostics Inc., NDD Medical Technologies, Futuremed America Inc., Sibelmed S.L., Uscom Ltd., e-LinkCare Meditech Co. Ltd., Monitored Therapeutics Inc. (MTI), Aluna Health Inc., Jones Medical Instrument Company, CMI Health, Healthup SA, Motustech Inc., Medikro Oy, Icen Technology Company Limited |
| Customization Scope | Request for Customization |
| Pricing And Purchase Options | Explore Purchase Options |
