
Human Immunodeficiency Viruses (HIV) Clinical Trials Market Report 2026
Global Outlook – By Phase (Phase I, Phase II, Phase III, Phase IV, Preclinical), By Drug Type (Antiretroviral Therapy (ART), Pre-Exposure Prophylaxis (PrEP), Post-Exposure Prophylaxis (PEP), Long-Acting Injectable Antiretrovirals, Experimental Drugs), By Study Design (Interventional Studies, Observational Studies, Expanded Access Studies), By Patient Population (Adults, Pediatric Patients, Seniors, Pregnant Women, High-Risk Populations), By End User (Pharmaceutical Companies, Research Institutes, Hospitals, Other End-Users) - Market Size, Trends, And Global Forecast 2026-2035
Human Immunodeficiency Viruses (HIV) Clinical Trials Market Overview
• Human Immunodeficiency Viruses (HIV) Clinical Trials market size has reached to $1.94 billion in 2025 • Expected to grow to $2.97 billion in 2030 at a compound annual growth rate (CAGR) of 8.9% • Growth Driver: Rising HIV Infections Driving Growth In HIV Clinical Trials Market Due To Increasing Disease Burden And Need For Effective Treatments • Market Trend: Long-Acting Injectable Solutions In HIV Prevention Boosting Market • North America was the largest region in 2025.What Is Covered Under Human Immunodeficiency Viruses (HIV) Clinical Trials Market?
Human immunodeficiency virus (HIV) clinical trials refer to research studies conducted on humans to evaluate the safety, efficacy, and potential outcomes of new drugs, therapies, vaccines, or treatment strategies aimed at preventing, managing, or curing HIV infection. These trials follow structured phases, from early safety testing to large-scale effectiveness studies, and help generate clinical evidence needed for regulatory approval and improved patient care. The main phases of human immunodeficiency virus (HIV) clinical trials are phase I, phase II, phase III, phase IV, and preclinical. Phase I represents the initial stage of testing, conducted on a small group of healthy volunteers or patients. These trials assess drug types such as antiretroviral therapy (ART), pre-exposure prophylaxis (PrEP), post-exposure prophylaxis (PEP), long-acting injectable antiretrovirals, and experimental drugs, using study designs that include interventional studies, observational studies, and expanded access studies. Patient populations targeted in these trials include adults, pediatric patients, seniors, pregnant women, and high-risk populations, and the results are utilized by end users, including pharmaceutical companies, research institutes, hospitals, and others.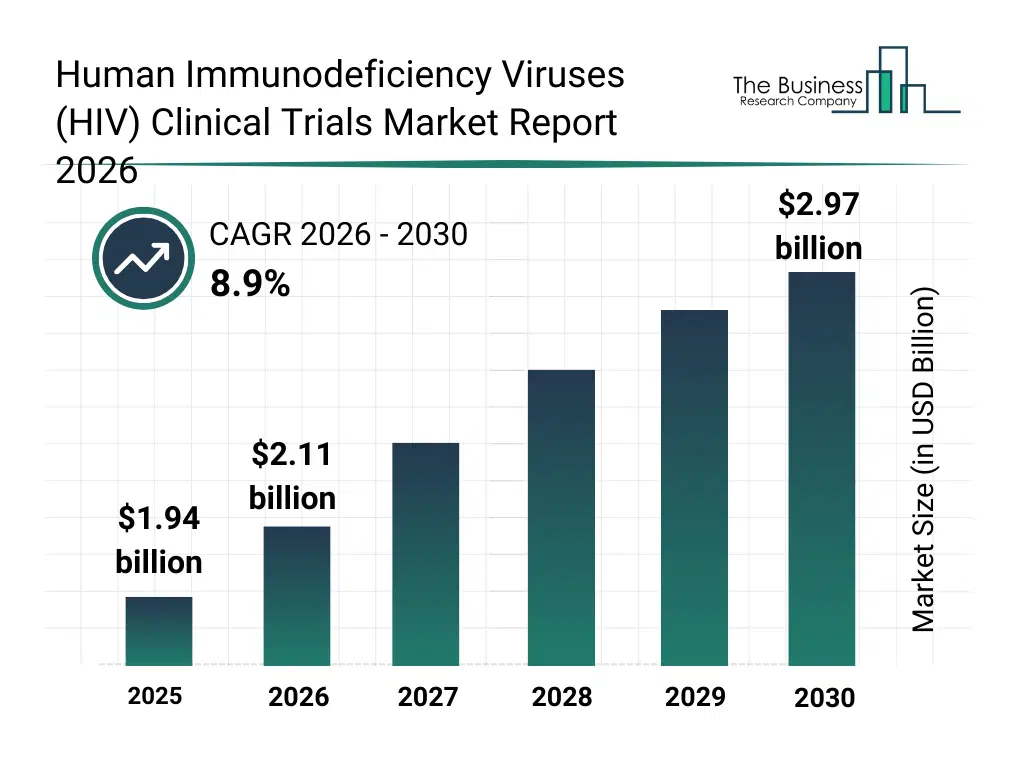
What Is The Human Immunodeficiency Viruses (HIV) Clinical Trials Market Size and Share 2026?
The human immunodeficiency viruses (hiv) clinical trials market size has grown strongly in recent years. It will grow from $1.94 billion in 2025 to $2.11 billion in 2026 at a compound annual growth rate (CAGR) of 9.2%. The growth in the historic period can be attributed to high global hiv disease burden, expansion of antiretroviral drug research, increased public and private funding for hiv studies, growth of clinical research infrastructure, regulatory support for hiv drug development.What Is The Human Immunodeficiency Viruses (HIV) Clinical Trials Market Growth Forecast?
The human immunodeficiency viruses (hiv) clinical trials market size is expected to see strong growth in the next few years. It will grow to $2.97 billion in 2030 at a compound annual growth rate (CAGR) of 8.9%. The growth in the forecast period can be attributed to increasing development of curative and preventive hiv therapies, rising adoption of adaptive trial designs, expansion of vaccine-focused hiv trials, growing use of ai in clinical research, increased focus on long-term safety and efficacy outcomes. Major trends in the forecast period include increasing adoption of decentralized clinical trial models, rising use of digital patient recruitment tools, growing focus on long-acting hiv therapies in trials, expansion of real-world evidence integration, enhanced use of data analytics in trial management.Global Human Immunodeficiency Viruses (HIV) Clinical Trials Market Segmentation
1) By Phase: Phase I, Phase II, Phase III, Phase IV, Preclinical 2) By Drug Type: Antiretroviral Therapy (ART), Pre-Exposure Prophylaxis (PrEP), Post-Exposure Prophylaxis (PEP), Long-Acting Injectable Antiretrovirals, Experimental Drugs 3) By Study Design: Interventional Studies, Observational Studies, Expanded Access Studies 4) By Patient Population: Adults, Pediatric Patients, Seniors, Pregnant Women, High-Risk Populations 5) By End User: Pharmaceutical Companies, Research Institutes, Hospitals, Other End-Users Subsegments: 1) By Phase I: First-In-Human Studies, Safety And Tolerability Studies 2) By Phase II: Dose-Finding Studies, Efficacy Studies 3) By Phase III: Pivotal Trials, Comparative Efficacy Studies 4) By Phase Iv: Post-Marketing Surveillance, Long-Term Safety Studies 5) By Preclinical: In Vitro Studies, Animal StudiesWhat Is The Driver Of The Human Immunodeficiency Viruses (HIV) Clinical Trials Market?
The increasing incidence of human immunodeficiency virus (HIV) infections is expected to propel the growth of the human immunodeficiency virus (HIV) clinical trials market going forward. Human immunodeficiency virus (HIV) infections refer to the condition caused by the HIV virus attacking and weakening the immune system, increasing susceptibility to infections and potentially leading to AIDS. The increase in HIV infections is largely due to lack of awareness about transmission, resulting to risky behaviors and delayed testing that facilitate the virus’s spread. Human immunodeficiency virus (HIV) clinical trials help patients suffering from HIV infections by testing new drugs, vaccines, and treatment strategies to improve prevention, manage the virus more effectively, and ultimately reduce disease progression and transmission. For instance, in August 2024, according to the Minority HIV/AIDS Fund (MHAF), a US-based government agency, an estimated 39.9 million individuals worldwide were living with HIV in 2023, consisting of 38.6 million adults and 1.4 million children, compared to previous years. Therefore, the increasing incidence of human immunodeficiency virus (HIV) infections is driving the growth of the human immunodeficiency virus (HIV) clinical trials market.Key Players In The Global Human Immunodeficiency Viruses (HIV) Clinical Trials Market
Major companies operating in the human immunodeficiency viruses (hiv) clinical trials market are Johnson & Johnson, F. Hoffmann-La Roche Ltd., Pfizer Inc., AbbVie Inc., Sanofi S.A., Thermo Fisher Scientific Inc., Amgen Inc., Gilead Sciences Inc., Merck & Co., IQVIA Holdings Inc., ICON plc, Moderna Inc., ViiV Healthcare Limited, HOOKIPA Pharma Inc., Excision BioTherapeutics Inc., American Gene Technologies International Inc., Bionor Holding AS, Aelix Therapeutics, Immuno Cure BioTech Private Limited, ImmunityBioGlobal Human Immunodeficiency Viruses (HIV) Clinical Trials Market Trends and Insights
Major companies operating in the human immunodeficiency virus (HIV) clinical trials market are focusing on developing innovative solutions such as HIV prevention injections to enhance treatment efficacy and reduce transmission rates. An HIV prevention injection refers to a long-acting injectable medication administered periodically to prevent HIV infection in high-risk individuals by blocking the virus from establishing infection in the body. For instance, in August 2025, Gilead Sciences Inc., a US-based pharmaceutical company, received an approval from the European Union for Yeytuo (lenacapavir) to treat HIV. It is a twice-yearly injectable HIV prevention treatment that offers high efficacy, nearly 100% in clinical trials, significantly reducing the risk of sexually acquired HIV-1 infection. Its long-acting dosing improves adherence and convenience compared to daily oral PrEP, addressing a major gap in HIV prevention. This is designed for adults and adolescents at increased risk, that aims to expand access and help reduce new HIV infections globally as part of comprehensive prevention strategies.What Are Latest Mergers And Acquisitions In The Human Immunodeficiency Viruses (HIV) Clinical Trials Market?
In July 2025, Elliott Investment Management L.P., a US-based investment company; Patient Square Capital LP, a US-based health investment company; and Veritas Capital Fund Management L.L.C., a US-based investment company, acquired Syneos Health for $7.1 billion. With this acquisition, investment groups aim to accelerate Syneos Health's growth, enhance customer delivery, and transform the company into a tech-enabled leader in biopharma solutions. Syneos Health is a US-based pharmaceutical commercial company that offers human immunodeficiency virus (HIV) clinical trials.Regional Outlook
North America was the largest region in the human immunodeficiency viruses (HIV) clinical trials market in 2025. Asia-Pacific is expected to be the fastest-growing region in the forecast period. The regions covered in this market report are Asia-Pacific, South East Asia, Western Europe, Eastern Europe, North America, South America, Middle East, Africa. The countries covered in this market report are Australia, Brazil, China, France, Germany, India, Indonesia, Japan, Taiwan, Russia, South Korea, UK, USA, Canada, Italy, Spain.What Defines the Human Immunodeficiency Viruses (HIV) Clinical Trials Market?
The human immunodeficiency virus (HIV) clinical trials market includes revenues earned by entities by providing services such as clinical study design and protocol development, patient recruitment and screening, and medical monitoring and care. The market value includes the value of related goods sold by the service provider or included within the service offering. Only goods and services traded between entities or sold to end consumers are included.How is Market Value Defined and Measured?
The market value is defined as the revenues that enterprises gain from the sale of goods and/or services within the specified market and geography through sales, grants, or donations in terms of the currency (in USD unless otherwise specified). The revenues for a specified geography are consumption values that are revenues generated by organizations in the specified geography within the market, irrespective of where they are produced. It does not include revenues from resales along the supply chain, either further along the supply chain or as part of other products.What Key Data and Analysis Are Included in the Human Immunodeficiency Viruses (HIV) Clinical Trials Market Report 2026?
The human immunodeficiency viruses (hiv) clinical trials market research report is one of a series of new reports from The Business Research Company that provides market statistics, including industry global market size, regional shares, competitors with the market share, detailed market segments, market trends and opportunities, and any further data you may need to thrive in the human immunodeficiency viruses (hiv) clinical trials industry. The market research report delivers a complete perspective of everything you need, with an in-depth analysis of the current and future state of the industry.Human Immunodeficiency Viruses (HIV) Clinical Trials Market Report Forecast Analysis
| Report Attribute | Details |
|---|---|
| Market Size Value In 2026 | $2.11 billion |
| Revenue Forecast In 2035 | $2.97 billion |
| Growth Rate | CAGR of 9.2% from 2026 to 2035 |
| Base Year For Estimation | 2025 |
| Actual Estimates/Historical Data | 2020-2025 |
| Forecast Period | 2026 - 2030 - 2035 |
| Market Representation | Revenue in USD Billion and CAGR from 2026 to 2035 |
| Segments Covered | Phase, Drug Type, Study Design, Patient Population, End User |
| Regional Scope | Asia-Pacific, Western Europe, Eastern Europe, North America, South America, Middle East, Africa |
| Country Scope | The countries covered in the report are Australia, Brazil, China, France, Germany, India, ... |
| Key Companies Profiled | Johnson & Johnson, F. Hoffmann-La Roche Ltd., Pfizer Inc., AbbVie Inc., Sanofi S.A., Thermo Fisher Scientific Inc., Amgen Inc., Gilead Sciences Inc., Merck & Co., IQVIA Holdings Inc., ICON plc, Moderna Inc., ViiV Healthcare Limited, HOOKIPA Pharma Inc., Excision BioTherapeutics Inc., American Gene Technologies International Inc., Bionor Holding AS, Aelix Therapeutics, Immuno Cure BioTech Private Limited, ImmunityBio |
| Customization Scope | Request for Customization |
| Pricing And Purchase Options | Explore Purchase Options |
