
Human Papillomavirus (HPV) Cytomegalovirus (CMV) Therapeutic Market Report 2026
Global Outlook – By Therapeutic Type (Vaccines, Antiviral Drugs, Immunotherapy), By Route of Administration (Intramuscular, Oral), By Indications (Cervical Cancer, Genital Warts, Head And Neck Cancer, Anal Cancer), By Distribution Channel (Hospitals, Retail Pharmacies, Specialty Clinics) - Market Size, Trends, And Global Forecast 2026-2035
Human Papillomavirus (HPV) Cytomegalovirus (CMV) Therapeutic Market Overview
• Human Papillomavirus (HPV) Cytomegalovirus (CMV) Therapeutic market size has reached to $4.9 billion in 2025 • Expected to grow to $7.32 billion in 2030 at a compound annual growth rate (CAGR) of 8.4% • Growth Driver: Increasing Prevalence Of Cervical Cancer Fueling the Growth Of The Market Due To Low HPV Vaccination Coverage • Market Trend: Takeda’s Approval of LIVTENCITY Highlights Shift Toward Targeted Post-Transplant CMV Therapies In The HPV And CMV Therapeutic Market • North America was the largest region in 2025 and Asia-Pacific is the fastest growing region.What Is Covered Under Human Papillomavirus (HPV) Cytomegalovirus (CMV) Therapeutic Market?
Human papillomavirus (HPV) cytomegalovirus (CMV) therapeutic refers to medical treatments designed to manage, reduce, or eliminate infections caused by human papillomavirus (HPV) and cytomegalovirus (CMV) viruses. These therapeutics aim to control viral replication, prevent disease progression, and alleviate symptoms associated with conditions such as cervical cancer (HPV) and congenital infections or immunocompromised complications (CMV). The main therapeutic types included in the human papillomavirus (HPV) cytomegalovirus (CMV) therapeutics are vaccines, antiviral drugs, and immunotherapy. Vaccines are biological formulations intended to induce immunity against specific infectious diseases. The different routes of administration include intramuscular, oral, and topical. The various indications comprise cervical cancer, genital warts, head and neck cancer, and anal cancer. The different distribution channels include hospitals, retail pharmacies, online pharmacies, and specialty clinics.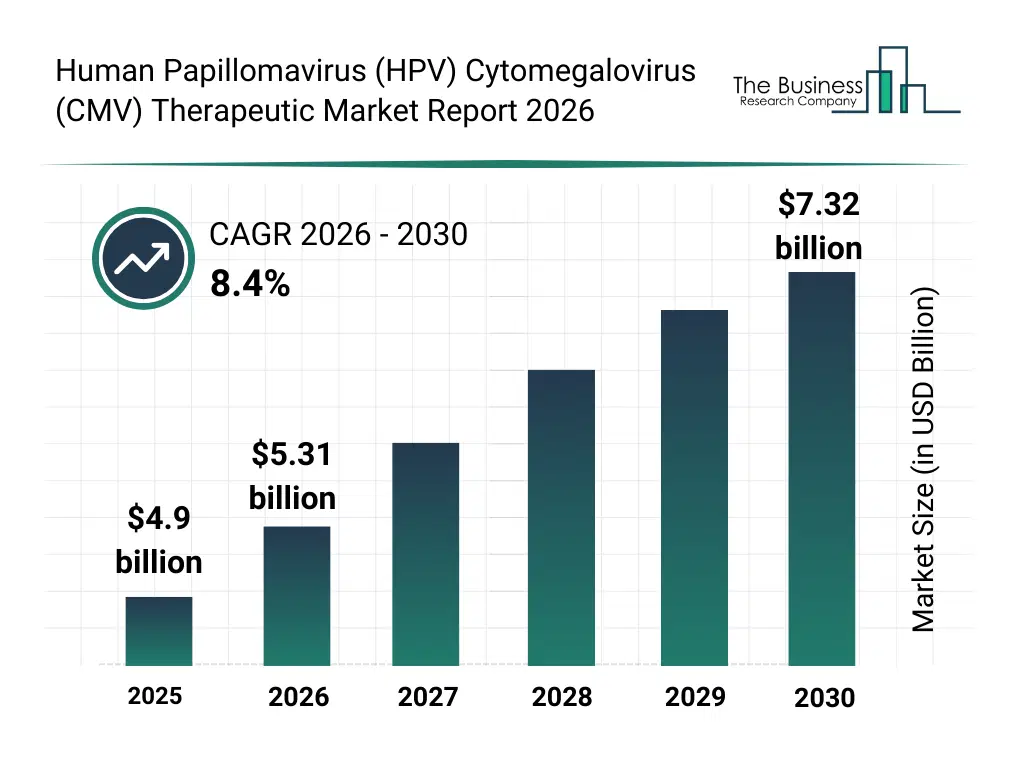
What Is The Human Papillomavirus (HPV) Cytomegalovirus (CMV) Therapeutic Market Size and Share 2026?
The human papillomavirus (hpv) cytomegalovirus (cmv) therapeutic market size has grown strongly in recent years. It will grow from $4.9 billion in 2025 to $5.31 billion in 2026 at a compound annual growth rate (CAGR) of 8.3%. The growth in the historic period can be attributed to high global prevalence of hpv and cmv infections, rising incidence of virus-associated cancers, widespread adoption of prophylactic hpv vaccination programs, increasing burden of cmv infections in immunocompromised patients, advancements in antiviral drug development.What Is The Human Papillomavirus (HPV) Cytomegalovirus (CMV) Therapeutic Market Growth Forecast?
The human papillomavirus (hpv) cytomegalovirus (cmv) therapeutic market size is expected to see strong growth in the next few years. It will grow to $7.32 billion in 2030 at a compound annual growth rate (CAGR) of 8.4%. The growth in the forecast period can be attributed to growing demand for therapeutic vaccines, increasing focus on cancer immunotherapy, rising investments in antiviral r&d, expanding screening and early intervention programs, increasing awareness of congenital and chronic viral infections. Major trends in the forecast period include increasing development of therapeutic vaccines for hpv and cmv, growing adoption of immunotherapy-based antiviral treatments, rising focus on combination therapies for viral infection management, expansion of targeted treatments for virus-associated cancers, increasing clinical trials for next-generation antiviral drugs.Global Human Papillomavirus (HPV) Cytomegalovirus (CMV) Therapeutic Market Segmentation
1) By Therapeutic Type: Vaccines, Antiviral Drugs, Immunotherapy 2) By Route of Administration: Intramuscular, Oral 3) By Indications: Cervical Cancer, Genital Warts, Head And Neck Cancer, Anal Cancer 4) By Distribution Channel: Hospitals, Retail Pharmacies, Specialty Clinics Subsegments: 1) By Vaccines: Prophylactic Vaccines, Therapeutic Vaccines, Recombinant Vaccines, Virus-Like Particle (VLP) Based Vaccines 2) By Antiviral Drugs: Nucleoside Analogues, Deoxyribonucleic Acid Polymerase Inhibitors 3) By Immunotherapy: Monoclonal Antibodies, Immune Checkpoint Inhibitors, Adoptive T-cell TherapyWhat Are The Drivers Of The Human Papillomavirus (HPV) Cytomegalovirus (CMV) Therapeutic Market?
The increasing prevalence of cervical cancer is expected to propel the growth of the human papillomavirus (HPV) and cytomegalovirus (CMV) therapeutic market going forward. Cervical cancer refers to a malignant tumor that develops in the cells of the cervix, which is the lower part of the uterus that connects to the vagina. The cervical cancer is increasing mainly due to persistent infection with high-risk strains of human papillomavirus (HPV) that causes abnormal cervical cell changes that can progress to cancer if not detected and treated early. Human papillomavirus (HPV) cytomegalovirus (CMV) therapeutics support cervical cancer management by targeting virus-driven disease progression, improving patient outcomes. They reduce the risk of cancer development by offering antiviral treatments and immunotherapies, enhancing long-term health and quality of life. For instance, in April 2024, according to the report by the American Cancer Society, a US-based non-profit organization, approximately 20 million new cervical cancer cases were diagnosed globally in 2022, which is projected to rise to 35 million by 2050. Therefore, the increasing prevalence of cervical cancer is driving the growth of the human papillomavirus (HPV) and cytomegalovirus (CMV) therapeutic market. The rising healthcare expenditure is expected to drive the growth of the human papillomavirus (HPV) cytomegalovirus (CMV) therapeutic market going forward. Healthcare expenditure is the total spending on medical services, treatments, and healthcare resources, funded by governments, insurers, and individuals to ensure accessible and quality care. Human papillomavirus (HPV) and cytomegalovirus (CMV) therapeutics benefit from rising healthcare expenditure that enables broader access to advanced antiviral treatments and preventive care. For instance, in May 2024, according to the Office for National Statistics, a UK-based government department, reported that total healthcare expenditure saw a 5.6% increase in nominal terms from 2022 to 2023, marking a significant acceleration compared to the 0.9% growth observed in 2022. Therefore, the rising healthcare expenditure is driving the growth of human papillomavirus (HPV) cytomegalovirus (CMV) therapeutic industry.Key Players In The Global Human Papillomavirus (HPV) Cytomegalovirus (CMV) Therapeutic Market
Major companies operating in the human papillomavirus (hpv) cytomegalovirus (cmv) therapeutic market are Merck & Co. Inc., Pfizer Inc., GlaxoSmithKline plc, Moderna Inc., BioNTech SE, Gilead Sciences Inc., F. Hoffmann-La Roche AG, Sanofi S.A., AstraZeneca plc, Novartis AG, AbbVie Inc., Bristol Myers Squibb Company, Takeda Pharmaceutical Company Limited, Amgen Inc., Vir Biotechnology Inc., Inovio Pharmaceuticals Inc., Dynavax Technologies Corporation, Hookipa Pharma Inc., Vaxart Inc., Bavarian Nordic A/SGlobal Human Papillomavirus (HPV) Cytomegalovirus (CMV) Therapeutic Market Trends and Insights
Major companies operating in the human papillomavirus (HPV) and cytomegalovirus (CMV) therapeutics market are focusing on developing innovative technologies, such as post-transplant anti-cytomegalovirus (CMV) treatment, to address unmet medical needs, enhance treatment efficacy, reduce resistance to existing therapies, and improve outcomes for immunocompromised patients. Post-transplant anti-cytomegalovirus (CMV) treatment refers to therapeutic interventions designed to prevent or manage cytomegalovirus (CMV) infections in organ or stem cell transplant recipients at high risk due to immunosuppression. For instance, in June 2024, Takeda Pharmaceutical Company Limited, a Japan-based pharmaceutical company, received approval from the Japanese Ministry of Health, Labour, and Welfare (MHLW) for LIVTENCITY (maribavir) for the treatment of post-transplant cytomegalovirus (CMV) infection or disease that is resistant to current anti-CMV medications. This innovative antiviral agent is the only treatment in Japan that specifically targets and inhibits the cytomegalovirus (CMV)-specific UL97 protein kinase, offering a novel mechanism of action distinct from traditional therapies.Regional Outlook
North America was the largest region in the human papillomavirus (HPV) cytomegalovirus (CMV) therapeutic market in 2025. Asia-Pacific is expected to be the fastest-growing region in the forecast period. The regions covered in this market report are Asia-Pacific, South East Asia, Western Europe, Eastern Europe, North America, South America, Middle East, Africa. The countries covered in this market report are Australia, Brazil, China, France, Germany, India, Indonesia, Japan, Taiwan, Russia, South Korea, UK, USA, Canada, Italy, Spain.What Defines the Human Papillomavirus (HPV) Cytomegalovirus (CMV) Therapeutic Market?
The human papillomavirus (HPV) cytomegalovirus (CMV) therapeutic market consists of revenues earned by entities providing specific services such as antiviral drug infusion and dispensing, therapeutic vaccine administration, genotypic resistance testing, viral load monitoring, patient counselling and follow-up care. The market value includes the value of related goods sold by the service provider or included within the service offering. The human papillomavirus (HPV) and cytomegalovirus (CMV) therapeutic market also includes sales of antiviral medications, therapeutic vaccines targeting human papillomavirus strains, immunomodulators, monoclonal antibodies, and gene therapy-based treatments. Values in this market are ‘factory gate’ values, that is, the value of goods sold by the manufacturers or creators of the goods, whether to other entities (including downstream manufacturers, wholesalers, distributors, and retailers) or directly to end customers. The value of goods in this market includes related services sold by the creators of the goods.How is Market Value Defined and Measured?
The market value is defined as the revenues that enterprises gain from the sale of goods and/or services within the specified market and geography through sales, grants, or donations in terms of the currency (in USD unless otherwise specified). The revenues for a specified geography are consumption values that are revenues generated by organizations in the specified geography within the market, irrespective of where they are produced. It does not include revenues from resales along the supply chain, either further along the supply chain or as part of other products.What Key Data and Analysis Are Included in the Human Papillomavirus (HPV) Cytomegalovirus (CMV) Therapeutic Market Report 2026?
The human papillomavirus (hpv) cytomegalovirus (cmv) therapeutic market research report is one of a series of new reports from The Business Research Company that provides market statistics, including industry global market size, regional shares, competitors with the market share, detailed market segments, market trends and opportunities, and any further data you may need to thrive in the human papillomavirus (hpv) cytomegalovirus (cmv) therapeutic industry. The market research report delivers a complete perspective of everything you need, with an in-depth analysis of the current and future state of the industry.Human Papillomavirus (HPV) Cytomegalovirus (CMV) Therapeutic Market Report Forecast Analysis
| Report Attribute | Details |
|---|---|
| Market Size Value In 2026 | $5.31 billion |
| Revenue Forecast In 2035 | $7.32 billion |
| Growth Rate | CAGR of 8.3% from 2026 to 2035 |
| Base Year For Estimation | 2025 |
| Actual Estimates/Historical Data | 2020-2025 |
| Forecast Period | 2026 - 2030 - 2035 |
| Market Representation | Revenue in USD Billion and CAGR from 2026 to 2035 |
| Segments Covered | Therapeutic Type, Route of Administration, Indications, Distribution Channel |
| Regional Scope | Asia-Pacific, Western Europe, Eastern Europe, North America, South America, Middle East, Africa |
| Country Scope | The countries covered in the report are Australia, Brazil, China, France, Germany, India, ... |
| Key Companies Profiled | Merck & Co. Inc., Pfizer Inc., GlaxoSmithKline plc, Moderna Inc., BioNTech SE, Gilead Sciences Inc., F. Hoffmann-La Roche AG, Sanofi S.A., AstraZeneca plc, Novartis AG, AbbVie Inc., Bristol Myers Squibb Company, Takeda Pharmaceutical Company Limited, Amgen Inc., Vir Biotechnology Inc., Inovio Pharmaceuticals Inc., Dynavax Technologies Corporation, Hookipa Pharma Inc., Vaxart Inc., Bavarian Nordic A/S |
| Customization Scope | Request for Customization |
| Pricing And Purchase Options | Explore Purchase Options |
