Human Respiratory Syncytial Virus Treatment Market Report 2026

Human Respiratory Syncytial Virus Treatment Market Report 2026
Global Outlook – By Treatment Type (Antiviral Medications, Monoclonal Antibodies, Vaccines, Supportive Care), By Patient Type (Pediatric Patients, Adult Patients), By Route Of Administration (Oral, Parenteral), By End User (Hospitals, Clinics, Home Care) - Market Size, Trends, And Global Forecast 2026-2035
Human Respiratory Syncytial Virus Treatment Market Overview
• Human Respiratory Syncytial Virus Treatment market size has reached to $1.83 billion in 2025 • Expected to grow to $2.76 billion in 2030 at a compound annual growth rate (CAGR) of 8.5% • Growth Driver: Expansion Of Telehealth Solutions Fueling The Growth Of The Market Due To Increasing Adoption Of Digital Healthcare Technologies • Market Trend: Advancements In Monoclonal Antibodies And Antiviral Drugs Driving Innovation In Disease Prevention And Treatment • North America was the largest region in 2025 and Asia-Pacific is the fastest growing region.What Is Covered Under Human Respiratory Syncytial Virus Treatment Market?
Human respiratory syncytial virus (RSV) treatment focuses on managing symptoms and preventing complications, particularly in vulnerable populations. It involves supportive care to ease breathing difficulties, reduce fever, and maintain hydration. In severe cases, hospitalization may be required for oxygen therapy and monitoring. Prevention strategies also play a crucial role in reducing infection risk. The main treatment types of human respiratory syncytial virus treatment are antiviral medications, monoclonal antibodies, vaccines, and supportive care. Antiviral medications are drugs designed to treat viral infections by inhibiting the replication of viruses, reducing the severity and duration of illness, and preventing complications. The various patient types include pediatric patients and adult patients. The multiple routes of administration include oral and parenteral and are used by several end users such as hospitals, clinics, and home care.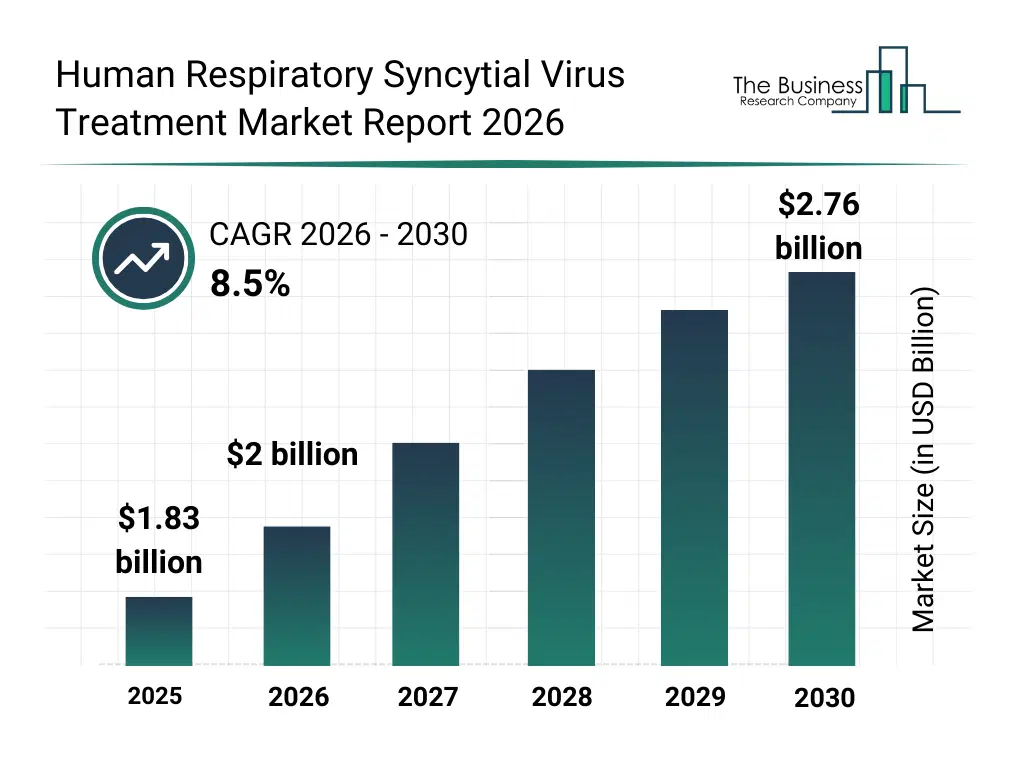
What Is The Human Respiratory Syncytial Virus Treatment Market Size and Share 2026?
The human respiratory syncytial virus treatment market size has grown strongly in recent years. It will grow from $1.83 billion in 2025 to $2 billion in 2026 at a compound annual growth rate (CAGR) of 8.8%. The growth in the historic period can be attributed to high global incidence of rsv infections, increased hospitalization rates among infants and elderly, reliance on supportive care treatments, limited availability of approved antiviral therapies, growing awareness of rsv-related complications.What Is The Human Respiratory Syncytial Virus Treatment Market Growth Forecast?
The human respiratory syncytial virus treatment market size is expected to see strong growth in the next few years. It will grow to $2.76 billion in 2030 at a compound annual growth rate (CAGR) of 8.5%. The growth in the forecast period can be attributed to increasing approval of rsv vaccines and monoclonal antibodies, rising demand for preventive therapies, expanding immunization programs for vulnerable populations, growing investment in respiratory virus r&d, increasing focus on reducing pediatric hospitalizations. Major trends in the forecast period include increasing adoption of monoclonal antibodies for rsv prevention and treatment, growing development of rsv vaccines across pediatric and adult populations, rising focus on early intervention for high-risk infants and elderly patients, expansion of supportive care protocols in severe rsv cases, increasing clinical research into novel antiviral therapies for rsv.Global Human Respiratory Syncytial Virus Treatment Market Segmentation
1) By Treatment Type: Antiviral Medications, Monoclonal Antibodies, Vaccines, Supportive Care 2) By Patient Type: Pediatric Patients, Adult Patients 3) By Route Of Administration: Oral, Parenteral 4) By End User: Hospitals, Clinics, Home Care Subsegments: 1) By Antiviral Medications: Fusion Inhibitors, Nucleoside Analogues, RNA Polymerase Inhibitors 2) By Monoclonal Antibodies: Palivizumab, Nirsevimab, Other RSV-Specific Monoclonal Antibodies 3) By Vaccines: Live Attenuated Vaccines, Subunit Vaccines, mRNA-Based Vaccines, Vector-Based Vaccines 4) By Supportive Care: Oxygen Therapy, Mechanical Ventilation, Hydration Therapy, Fever and Pain ManagementWhat Is The Driver Of The Human Respiratory Syncytial Virus Treatment Market?
The expanding telehealth solutions are expected to fuel the growth of the human respiratory syncytial virus treatment market going forward. Telehealth solutions refer to digital healthcare services that enable remote patient consultations, diagnosis, monitoring, and treatment through telecommunication technologies. Telehealth solutions are increasing due to the rising adoption of digital healthcare technologies, enabling remote patient care and reducing the burden on traditional healthcare facilities. Telehealth solutions for human respiratory syncytial virus treatment facilitate remote monitoring, early detection, and virtual consultations, enhancing patient care while minimizing hospital visits. For instance, according to Telehealth.HHS.gov, a US-based government official information provider, in quarter four of 2023, telehealth usage climbed to approximately 21,883,731 users, reflecting a growth of 2,783,159 users or a 13% increase from the third quarter. Therefore, the expanding telehealth solutions are driving the growth of the human respiratory syncytial virus treatment industry.Key Players In The Global Human Respiratory Syncytial Virus Treatment Market
Major companies operating in the human respiratory syncytial virus treatment market are Pfizer Inc., Merck & Co, Sanofi S.A., AstraZeneca plc, GSK plc, Johnson & Johnson, Moderna Inc., Kyowa Kirin Co. Ltd., Novavax Inc., Alnylam Pharmaceuticals Inc., AdVaccine Biotechnology Co. Ltd., Enanta Pharmaceuticals Inc., Aridis Pharmaceuticals Inc., Synairgen plc, VBI Vaccines Inc., SAB Biotherapeutics Inc., Meissa Vaccines Inc., Osivax SAS, Codagenix Inc., BioVaxys Technology Corp.Global Human Respiratory Syncytial Virus Treatment Market Trends and Insights
Major companies operating in the human respiratory syncytial virus treatment market are focusing on innovative therapies, such as monoclonal antibodies and antiviral drugs, to gain a competitive edge in the industry. Monoclonal antibodies are lab-made proteins that help the body fight infections, while antiviral drugs are medicines that stop viruses from growing or spreading. For instance, in July 2023, AstraZeneca plc, a UK-based biopharmaceutical company, partnered with Sanofi S.A., a France-based pharmaceutical company, to announce that the U.S. Food and Drug Administration (FDA) approved Beyfortus (nirsevimab-alip), a monoclonal antibody designed to prevent RSV lower respiratory tract disease in infants and high-risk children. This approval represented a major advancement in RSV prevention, as Beyfortus provides a long-acting, single-dose option that delivers passive immunity throughout the RSV season. It is particularly beneficial for newborns and infants, who are at the highest risk of severe RSV infections, helping to reduce hospitalizations and complications.What Are Latest Mergers And Acquisitions In The Human Respiratory Syncytial Virus Treatment Market?
In December 2023, AstraZeneca plc, a UK-based biopharmaceutical company, acquired Icosavax Inc. for an undisclosed amount. With this acquisition, AstraZeneca aimed to strengthen its late-stage respiratory infectious disease portfolio by adding Icosavax’s potential first-in-class protein VLP vaccine targeting both respiratory syncytial virus (RSV) and human metapneumovirus (hMPV). Icosavax is a US-based clinical-stage biotech company focused on developing virus-like particle (VLP) vaccines for RSV/hMPV combination vaccine leveraging an innovative VLP platform.Regional Outlook
North America was the largest region in the human respiratory syncytial virus treatment market in 2025. Asia-Pacific is expected to be the fastest-growing region in the forecast period. The regions covered in this market report are Asia-Pacific, South East Asia, Western Europe, Eastern Europe, North America, South America, Middle East, Africa. The countries covered in this market report are Australia, Brazil, China, France, Germany, India, Indonesia, Japan, Taiwan, Russia, South Korea, UK, USA, Canada, Italy, Spain.What Defines the Human Respiratory Syncytial Virus Treatment Market?
The human respiratory syncytial virus treatment market consists of revenues earned by entities by providing services such as oxygen therapy, mechanical ventilation, hydration therapy, and fever and pain management. The market value includes the value of related goods sold by the service provider or included within the service offering. The human respiratory syncytial virus treatment market also includes sales of medications, monoclonal antibodies, and vaccines. Values in this market are ‘factory gate’ values, that is the value of goods sold by the manufacturers or creators of the goods, whether to other entities (including downstream manufacturers, wholesalers, distributors and retailers) or directly to end customers. The value of goods in this market includes related services sold by the creators of the goods.How is Market Value Defined and Measured?
The market value is defined as the revenues that enterprises gain from the sale of goods and/or services within the specified market and geography through sales, grants, or donations in terms of the currency (in USD unless otherwise specified). The revenues for a specified geography are consumption values that are revenues generated by organizations in the specified geography within the market, irrespective of where they are produced. It does not include revenues from resales along the supply chain, either further along the supply chain or as part of other products.What Key Data and Analysis Are Included in the Human Respiratory Syncytial Virus Treatment Market Report 2026?
The human respiratory syncytial virus treatment market research report is one of a series of new reports from The Business Research Company that provides market statistics, including industry global market size, regional shares, competitors with the market share, detailed market segments, market trends and opportunities, and any further data you may need to thrive in the human respiratory syncytial virus treatment industry. The market research report delivers a complete perspective of everything you need, with an in-depth analysis of the current and future state of the industry.Human Respiratory Syncytial Virus Treatment Market Report Forecast Analysis
| Report Attribute | Details |
|---|---|
| Market Size Value In 2026 | $2 billion |
| Revenue Forecast In 2035 | $2.76 billion |
| Growth Rate | CAGR of 8.8% from 2026 to 2035 |
| Base Year For Estimation | 2025 |
| Actual Estimates/Historical Data | 2020-2025 |
| Forecast Period | 2026 - 2030 - 2035 |
| Market Representation | Revenue in USD Billion and CAGR from 2026 to 2035 |
| Segments Covered | Treatment Type, Patient Type, Route Of Administration, End User |
| Regional Scope | Asia-Pacific, Western Europe, Eastern Europe, North America, South America, Middle East, Africa |
| Country Scope | The countries covered in the report are Australia, Brazil, China, France, Germany, India, ... |
| Key Companies Profiled | Pfizer Inc., Merck & Co, Sanofi S.A., AstraZeneca plc, GSK plc, Johnson & Johnson, Moderna Inc., Kyowa Kirin Co. Ltd., Novavax Inc., Alnylam Pharmaceuticals Inc., AdVaccine Biotechnology Co. Ltd., Enanta Pharmaceuticals Inc., Aridis Pharmaceuticals Inc., Synairgen plc, VBI Vaccines Inc., SAB Biotherapeutics Inc., Meissa Vaccines Inc., Osivax SAS, Codagenix Inc., BioVaxys Technology Corp. |
| Customization Scope | Request for Customization |
| Pricing And Purchase Options | Explore Purchase Options |
Frequently Asked Questions
The Human Respiratory Syncytial Virus Treatment market was valued at $1.83 billion in 2025, increased to $2 billion in 2026, and is projected to reach $2.76 billion by 2030.
The global Human Respiratory Syncytial Virus Treatment market is expected to grow at a CAGR of 8.5% from 2026 to 2035 to reach $2.76 billion by 2035.
Some Key Players in the Human Respiratory Syncytial Virus Treatment market Include, Pfizer Inc., Merck & Co, Sanofi S.A., AstraZeneca plc, GSK plc, Johnson & Johnson, Moderna Inc., Kyowa Kirin Co. Ltd., Novavax Inc., Alnylam Pharmaceuticals Inc., AdVaccine Biotechnology Co. Ltd., Enanta Pharmaceuticals Inc., Aridis Pharmaceuticals Inc., Synairgen plc, VBI Vaccines Inc., SAB Biotherapeutics Inc., Meissa Vaccines Inc., Osivax SAS, Codagenix Inc., BioVaxys Technology Corp. .
Major trend in this market includes: Advancements In Monoclonal Antibodies And Antiviral Drugs Driving Innovation In Disease Prevention And Treatment. For further insights on this market.
Request for SampleNorth America was the largest region in the human respiratory syncytial virus treatment market in 2025. Asia-Pacific is expected to be the fastest-growing region in the forecast period. The regions covered in the human respiratory syncytial virus treatment market report are Asia-Pacific, South East Asia, Western Europe, Eastern Europe, North America, South America, Middle East, Africa.
