
Hypoprothrombinemia Treatment Market Report 2026
Global Outlook – By Type (Type I Deficiency Treatment, Type II Deficiency Treatment), By Drug Type (Vitamin K, Fresh Frozen Plasma, Prothrombin Complex Concentrates, Recombinant Prothrombin), By Indication Based (Vitamin K Deficiency, Liver Diseases, Cirrhosis, Hepatitis, Anticoagulant related Hypoprothrombinemia, Genetic Disorders, Malabsorption Syndromes), By Drug Delivery Method (Oral Delivery, Injectable Delivery, Intravenous, Intramuscular, Transfusion Based Delivery), By End User (Hospitals, Speciality Clinics, Homecare Settings) – Market Size, Trends, Strategies, and Forecast to 2035
Hypoprothrombinemia Treatment Market Overview
• Hypoprothrombinemia Treatment market size has reached to $1.52 billion in 2025 • Expected to grow to $2.3 billion in 2030 at a compound annual growth rate (CAGR) of 8.6% • Growth Driver: Rising Prevalence Of Bleeding Disorders Driving Growth Of The Market Due To Enhanced Diagnosis And Treatment Demand • Market Trend: Innovative Treatments Enhancing Patient Outcomes With Targeted Clotting Factor Replacement • North America was the largest region in 2025 and Asia-Pacific is the fastest growing region.What Is Covered Under Hypoprothrombinemia Treatment Market?
Hypoprothrombinemia treatment refers to medical interventions aimed at restoring adequate levels of prothrombin (Factor II), a key protein involved in blood clotting. The purpose is to prevent or control abnormal bleeding by correcting the underlying deficiency, whether due to genetic factors, vitamin K deficiency, liver disease, or anticoagulant overuse. The main types of hypoprothrombinemia treatment are Type I deficiency treatment and Type II deficiency treatment. Type I hypoprothrombinemia (true prothrombin deficiency) is typically treated with prothrombin complex concentrates (PCCs) or fresh frozen plasma (FFP) to restore adequate prothrombin levels and improve clotting ability. The treatment involves drug types such as vitamin K, fresh frozen plasma, prothrombin complex concentrates, and recombinant prothrombin. It is indicated for conditions including vitamin K deficiency, liver diseases, cirrhosis, hepatitis, anticoagulant-related hypoprothrombinemia, genetic disorders, and malabsorption syndromes. Drug delivery methods include oral delivery, injectable delivery, intravenous, intramuscular, and transfusion-based delivery. The key end users are hospitals, specialty clinics, and homecare settings.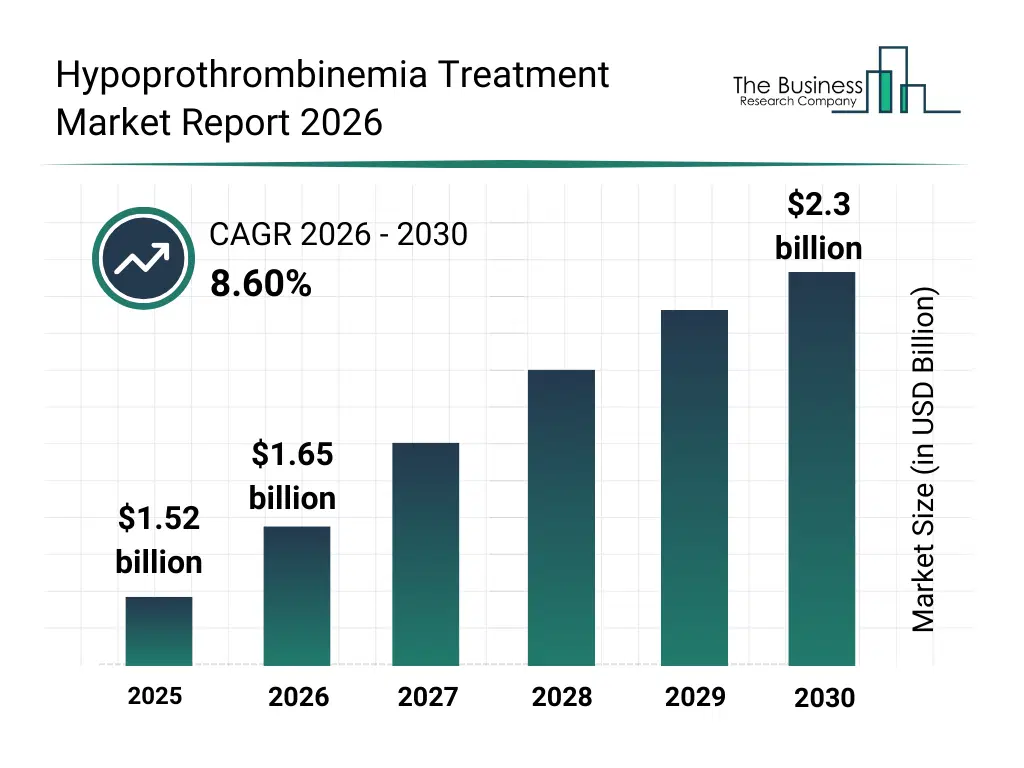
What Is The Hypoprothrombinemia Treatment Market Size and Share 2026?
The hypoprothrombinemia treatment market size has grown strongly in recent years. It will grow from $1.52 billion in 2025 to $1.65 billion in 2026 at a compound annual growth rate (CAGR) of 8.9%. The growth in the historic period can be attributed to vitamin k deficiency prevalence, liver disease complications, anticoagulant overuse, hospital transfusion protocols, inherited clotting disorders.What Is The Hypoprothrombinemia Treatment Market Growth Forecast?
The hypoprothrombinemia treatment market size is expected to see strong growth in the next few years. It will grow to $2.3 billion in 2030 at a compound annual growth rate (CAGR) of 8.6%. The growth in the forecast period can be attributed to advances in recombinant clotting factors, improved emergency care infrastructure, aging population bleeding risks, precision hematology adoption, outpatient coagulation management. Major trends in the forecast period include increased use of prothrombin complex concentrates, growing reliance on rapid coagulation correction therapies, expansion of vitamin k-based treatment protocols, improved emergency bleeding management standards, rising adoption of recombinant coagulation factors.Global Hypoprothrombinemia Treatment Market Segmentation
1) By Type: Type I Deficiency Treatment, Type II Deficiency Treatment 2) By Drug Type: Vitamin K, Fresh Frozen Plasma, Prothrombin Complex Concentrates, Recombinant Prothrombin 3) By Indication Based: Vitamin K Deficiency, Liver Diseases, Cirrhosis, Hepatitis, Anticoagulant related Hypoprothrombinemia, Genetic Disorders, Malabsorption Syndromes 4) By Drug Delivery Method: Oral Delivery, Injectable Delivery, Intravenous, Intramuscular, Transfusion Based Delivery 5) By End User: Hospitals, Speciality Clinics, Homecare Settings Subsegments: 1) By Type I Deficiency Treatment: Prothrombin Complex Concentrates (PCCs), Fresh Frozen Plasma (FFP), Recombinant Prothrombin, Plasma-derived Coagulation Factors 2) By Type II Deficiency Treatment: Vitamin K Therapy, Warfarin Reversal Agents, Liver Function Supportive Therapy, Anticoagulant Adjustment TherapiesWhat Are The Drivers Of The Hypoprothrombinemia Treatment Market?
The increasing prevalence of bleeding disorders is expected to drive the growth of the hypoprothrombinemia treatment market. Bleeding disorders are conditions in which the blood fails to clot properly, leading to excessive or prolonged bleeding. The rising prevalence of bleeding disorders is primarily due to improved diagnostic capabilities and greater awareness leading to more accurate identification and reporting of these conditions. Hypoprothrombinemia treatment is used to restore blood clotting ability in patients with bleeding disorders by replenishing deficient prothrombin levels. For instance, in November 2024, according to the BMC Health Services Research, a UK-based journal that publishes research on the organization, delivery, and evaluation of health care services, between April 2022 and March 2023 in the United Kingdom, a total of 6,662 males were identified with hemophilia A, of which 5,032 were adults (aged 18 and above), and 2,421 received treatment with either factor replacement therapy or emicizumab. During the same period, 1,342 males were recorded with hemophilia B, including 1,022 adults, of whom 479 underwent factor replacement treatment. Therefore, this increasing prevalence of bleeding disorders is expected to propel the growth of the hypoprothrombinemia treatment industry. The growing use of biosimilars is expected to drive the growth of the hypoprothrombinemia treatment market going forward. Biosimilars are biologic medical products highly similar to an already approved original biologic, with no clinically meaningful differences in safety or effectiveness. The rising use of biosimilars is driven by their potential to provide more affordable, accessible treatment options while maintaining comparable safety and efficacy to original biologics. Biosimilars in hypoprothrombinemia treatment are used to provide cost-effective alternatives to original clotting factor therapies, improving patient access to essential coagulation management. For instance, in January 2023, according to the Cardinal Health Inc., a US-based healthcare company, as of January 2022, the U.S. had 33 biosimilar products approved by the FDA, with 21 of them available on the market and by 2023, the number of FDA-approved biosimilars had risen to 40, with 25 commercially available. Therefore, the growing use of biosimilars is driving growth of the hypoprothrombinemia treatment industry.Key Players In The Global Hypoprothrombinemia Treatment Market
Major companies operating in the hypoprothrombinemia treatment market are Biotest AG, Kedrion Biopharma, LFB S.A., Hualan Biological Engineering Inc., China Biologic Products Holdings Inc., Bio Products Laboratory (BPL), Kamada Ltd., Green Cross Corporation, Shanghai RAAS Blood Products Co. Ltd., SK Plasma Co. Ltd., Emergent BioSolutions Inc., Intas Pharmaceuticals Ltd., Bharat Serums and Vaccines Ltd., ZLB Behring (CSL Behring), Mitsubishi Tanabe Pharma CorporationGlobal Hypoprothrombinemia Treatment Market Trends and Insights
Major companies operating in the hypoprothrombinemia treatment are focusing on innovative therapies, such as non-activated four-factor prothrombin complex concentrate, to rapidly restore clotting factor levels and reduce bleeding risks in patients requiring urgent reversal of anticoagulation. Non-activated four-factor prothrombin complex concentrate (4F-PCC) is a plasma-derived therapy containing clotting factors II, VII, IX, and X, used to rapidly reverse vitamin K antagonist effects and treat bleeding disorders like hypoprothrombinemia. For instance, in January 2024, Octapharma USA, a US-based healthcare company subsidiary of Octapharma AG, launched Balfaxar, a non-activated four-factor prothrombin complex concentrate (4F-PCC) recently approved by the FDA for the urgent reversal of vitamin K antagonist-induced coagulation deficiencies in adults requiring emergency surgery or invasive procedures. This therapy, containing key vitamin K-dependent clotting factors and antithrombotic proteins, addresses critical needs in hypoprothrombinemia management.Regional Insights
North America was the largest region in the hypoprothrombinemia treatment market in 2025. Asia-Pacific is expected to be the fastest-growing region in the forecast period. The regions covered in this market report are Asia-Pacific, South East Asia, Western Europe, Eastern Europe, North America, South America, Middle East, Africa. The countries covered in this market report are Australia, Brazil, China, France, Germany, India, Indonesia, Japan, Taiwan, Russia, South Korea, UK, USA, Canada, Italy, Spain.What Defines the Hypoprothrombinemia Treatment Market?
The hypoprothrombinemia treatment market consists of revenues earned by entities by providing services such as diagnostic testing, hospital care, specialized transfusion services, therapeutic drug monitoring, and patient management programs. The market value includes the value of related goods sold by the service provider or included within the service offering. The hypoprothrombinemia treatment market also includes sales of plasma-derived concentrates, synthetic clotting factors, oral anticoagulant reversal agents, blood products, and plasma derivatives. Values in this market are ‘factory gate’ values, that is, the value of goods sold by the manufacturers or creators of the goods, whether to other entities (including downstream manufacturers, wholesalers, distributors, and retailers) or directly to end customers. The value of goods in this market includes related services sold by the creators of the goods.How is Market Value Defined and Measured?
The market value is defined as the revenues that enterprises gain from the sale of goods and/or services within the specified market and geography through sales, grants, or donations in terms of the currency (in USD unless otherwise specified). The revenues for a specified geography are consumption values that are revenues generated by organizations in the specified geography within the market, irrespective of where they are produced. It does not include revenues from resales along the supply chain, either further along the supply chain or as part of other products.What Key Data and Analysis Are Included in the Hypoprothrombinemia Treatment Market Report 2026?
The hypoprothrombinemia treatment market research report is one of a series of new reports from The Business Research Company that provides market statistics, including industry global market size, regional shares, competitors with the market share, detailed market segments, market trends and opportunities, and any further data you may need to thrive in the hypoprothrombinemia treatment industry. The market research report delivers a complete perspective of everything you need, with an in-depth analysis of the current and future state of the industry.Hypoprothrombinemia Treatment Market Report Forecast Analysis
| Report Attribute | Details |
|---|---|
| Market Size Value In 2026 | $1.65 billion |
| Revenue Forecast In 2035 | $2.3 billion |
| Growth Rate | CAGR of 8.9% from 2026 to 2035 |
| Base Year For Estimation | 2025 |
| Actual Estimates/Historical Data | 2020-2025 |
| Forecast Period | 2026 - 2030 - 2035 |
| Market Representation | Revenue in USD Billion and CAGR from 2026 to 2035 |
| Segments Covered | Type, Drug Type, Indication Based, Drug Delivery Method, End User |
| Regional Scope | Asia-Pacific, Western Europe, Eastern Europe, North America, South America, Middle East, Africa |
| Country Scope | The countries covered in the report are Australia, Brazil, China, France, Germany, India, ... |
| Key Companies Profiled | Biotest AG, Kedrion Biopharma, LFB S.A., Hualan Biological Engineering Inc., China Biologic Products Holdings Inc., Bio Products Laboratory (BPL), Kamada Ltd., Green Cross Corporation, Shanghai RAAS Blood Products Co. Ltd., SK Plasma Co. Ltd., Emergent BioSolutions Inc., Intas Pharmaceuticals Ltd., Bharat Serums and Vaccines Ltd., ZLB Behring (CSL Behring), Mitsubishi Tanabe Pharma Corporation |
| Customization Scope | Request for Customization |
| Pricing And Purchase Options | Explore Purchase Options |
