
In Silico Clinical Trials Market Report 2026
Global Outlook – By Phase (Phase I, Phase II, Phase III, Phase IV), By Therapeutic Area (Oncology, Infectious Disease, Hematology, Cardiology, Dermatology, Neurology, Diabetes, Other Therapeutic Areas), By Industry (Medical Devices, Pharmaceutical) – Market Size, Trends, Strategies, and Forecast to 2035
In Silico Clinical Trials Market Overview
• In Silico Clinical Trials market size has reached to $3.74 billion in 2025 • Expected to grow to $6.14 billion in 2030 at a compound annual growth rate (CAGR) of 10.5% • Growth Driver: The In Silico Clinical Trials Market Surges In Response To The Urgency To Curb Clinical Trial Costs • Market Trend: Groundbreaking Technology Innovations Transforming The Silico Clinical Trials Market • North America was the largest region in 2025 and Asia-Pacific is the fastest growing region.What Is Covered Under In Silico Clinical Trials Market?
In silico clinical trials also refer as virtual clinical trial is a computer-simulations used to develop or evaluate the safety and efficacy of a medicinal product, device, or intervention. It assesses the safety and effectiveness of medical devices and imaging systems. The main phases of in silico clinical trials are Phase I, Phase II, Phase III, and Phase IV. Phase I refers to the first time a drug or treatment is assessed on a small group of people and is used to assess the safety and tolerability of new drugs in humans. These are conducted for oncology, infectious disease, hematology, cardiology, dermatology, neurology, diabetes, and others, and are used by medical devices and pharmaceutical industries.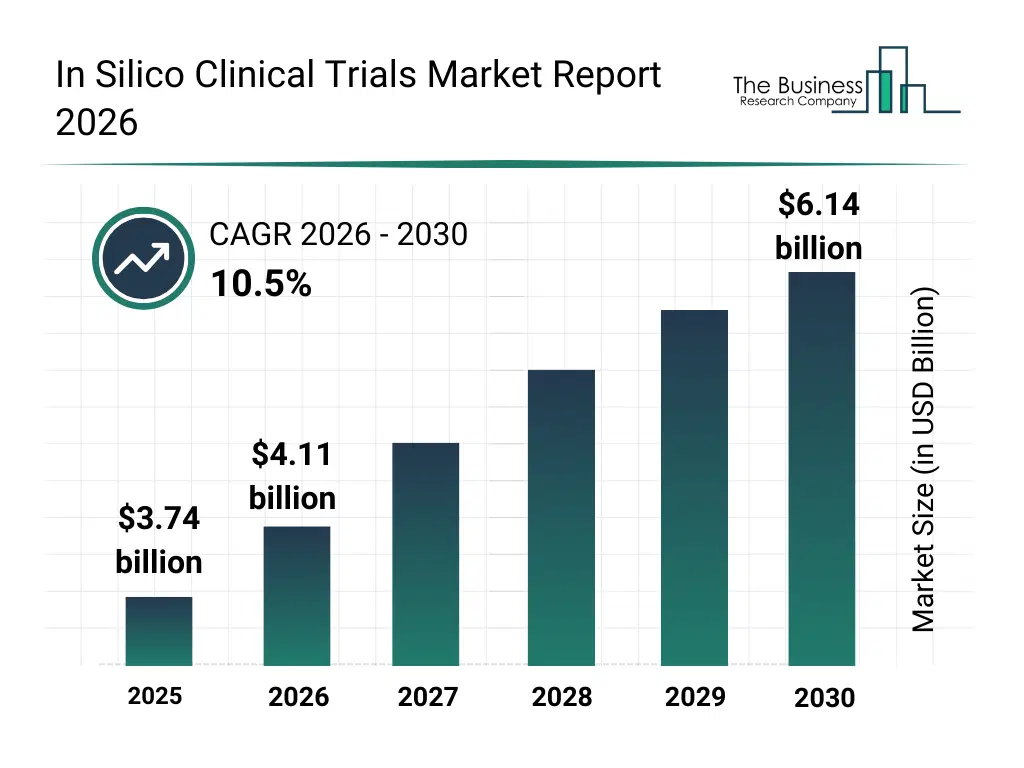
What Is The In Silico Clinical Trials Market Size and Share 2026?
The in silico clinical trials market size has grown rapidly in recent years. It will grow from $3.74 billion in 2025 to $4.11 billion in 2026 at a compound annual growth rate (CAGR) of 10.1%. The growth in the historic period can be attributed to high cost of traditional clinical trials, long drug development timelines, increasing use of computational modeling, need for improved trial efficiency, growth of medical device innovation.What Is The In Silico Clinical Trials Market Growth Forecast?
The in silico clinical trials market size is expected to see rapid growth in the next few years. It will grow to $6.14 billion in 2030 at a compound annual growth rate (CAGR) of 10.5%. The growth in the forecast period can be attributed to rising adoption of AI driven simulations, regulatory acceptance of virtual evidence, demand for faster clinical validation, expansion of personalized medicine, increasing R&D investment by pharma companies. Major trends in the forecast period include virtual patient modeling, simulation based trial design, digital twin clinical testing, predictive safety assessment, computational drug evaluation.Global In Silico Clinical Trials Market Segmentation
1) By Phase: Phase I, Phase II, Phase III, Phase IV 2) By Therapeutic Area: Oncology, Infectious Disease, Hematology, Cardiology, Dermatology, Neurology, Diabetes, Other Therapeutic Areas 3) By Industry: Medical Devices, Pharmaceutical Subsegments: 1) By Phase I: Safety And Dosage Trials, Pharmacokinetics And Pharmacodynamics Studies 2) By Phase II: Efficacy Trials, Dose-Response Studies 3) By Phase III: Comparative Effectiveness Trials, Large-Scale Efficacy Studies 4) By Phase IV: Post-Marketing Surveillance, Long-Term Safety StudiesWhat Is The Driver Of The In Silico Clinical Trials Market?
The growing need to curb clinical trial costs is expected to propel the growth of the in silico clinical trials market going forward. Clinical trial costs refer to the expenses associated with planning, conducting, and managing a clinical trial to test the safety, efficacy, and potential benefits of a new medical intervention, such as a drug, medical device, or treatment protocol. Traditional clinical trials are a lengthy and expensive process, often taking 10-15 years and costing billions of dollars to bring a new drug to market. In silico clinical trials offer a promising alternative, using computer simulations to test the safety and efficacy of drugs and medical devices. For instance, in February 2023, according to Genetic Engineering and Biotechnology News, a US-based platform that provides the latest information on genetic engineering and biotechnology, developing a new drug among the top 20 global biopharma companies rose by 15%, from $298 million in 2022 to approximately $2.3 billion in 2023, and this includes the costs incurred from developing a drug through clinical trials. Therefore, the growing need to curb clinical trial costs will drive the in silico clinical trial industry.Key Players In The Global In Silico Clinical Trials Market
Major companies operating in the in silico clinical trials market are Dassault Systemes SE; Evotec A.G; Evidera; Certara Inc.; Simulations Plus Inc.; Insilico Medicine Inc.; AnyLogic Company; Biomax Informatics; GNS Healthcare Inc.; 4P-Pharma; Nuventra Pharma Sciences; Archimedes; Novadiscovery Sas; Rosa & Co.; In Silico Biosciences; Leadscope; Biognos Ab; BioNova; Immunetrics Inc.; InSilicoTrials; Physiomics plc; Entelos; InhibOx; Selvita; Abzena LimitedGlobal In Silico Clinical Trials Market Trends and Insights
Major companies operating in the in silico clinical trials market are focused on innovative technologies, such as clinical trial platforms, to sustain their position in the market. A clinical trial platform is a special type of adaptive, disease-focused, randomized clinical trial (RCT) designed to evaluate multiple, simultaneous interventions against a single, constant control group. For instance, in October 2023, Advarra, a US-based provider of regulatory review solutions and clinical research technology for sites and sponsors, launched Longboat v2.2, a new technology functionality designed to improve all stakeholders’ clinical trial experiences. Longboat, an established platform used in more than 70 countries and by nearly 20,000 sites, now features an online patient portal with a comprehensive set of engagement tools, including a simple document exchange that enables transparent collaboration between patients, sites, and sponsors or clinical research organizations (CROs) without adding a technology burden.What Are Latest Mergers And Acquisitions In The In Silico Clinical Trials Market?
In May 2023, Recursion, a US-based clinical-stage TechBio company, announced the acquisition of Cyclica and Valence for an undisclosed amount. The strategic acquisitions of Cyclica and Valence add industry-leading capabilities in digital chemistry, as well as machine learning and artificial intelligence, which are combined with Recursion's large-scale automated wet laboratories and supercomputing capabilities and enable Recursion to deploy the most comprehensive, technology-enabled drug discovery solution in the biopharma industry. Cyclica is a Canada-based biotechnology company, and Valence is a Canada-based deep learning research institute.Regional Insights
North America was the largest region in the in silico clinical trials market in 2025. Asia-Pacific is expected to be the fastest-growing region in the in silico clinical trials market report during the forecast period. The regions covered in this market report are Asia-Pacific, South East Asia, Western Europe, Eastern Europe, North America, South America, Middle East, Africa. The countries covered in this market report are Australia, Brazil, China, France, Germany, India, Indonesia, Japan, Taiwan, Russia, South Korea, UK, USA, Canada, Italy, SpainWhat Defines the In Silico Clinical Trials Market?
The in silico clinical trials market includes revenues earned by entities by providing services such as data management, trial design, information compilation and site support. The market value includes the value of related goods sold by the service provider or included within the service offering. Only goods and services traded between entities or sold to end consumers are included.How is Market Value Defined and Measured?
The market value is defined as the revenues that enterprises gain from the sale of goods and/or services within the specified market and geography through sales, grants, or donations in terms of the currency (in USD unless otherwise specified). The revenues for a specified geography are consumption values that are revenues generated by organizations in the specified geography within the market, irrespective of where they are produced. It does not include revenues from resales along the supply chain, either further along the supply chain or as part of other products.What Key Data and Analysis Are Included in the In Silico Clinical Trials Market Report 2026?
The in silico clinical trials market research report is one of a series of new reports from The Business Research Company that provides market statistics, including industry global market size, regional shares, competitors with the market share, detailed market segments, market trends and opportunities, and any further data you may need to thrive in the in silico clinical trials industry. The market research report delivers a complete perspective of everything you need, with an in-depth analysis of the current and future state of the industry.In Silico Clinical Trials Market Report Forecast Analysis
| Report Attribute | Details |
|---|---|
| Market Size Value In 2026 | $4.11 billion |
| Revenue Forecast In 2035 | $6.14 billion |
| Growth Rate | CAGR of 10.1% from 2026 to 2035 |
| Base Year For Estimation | 2025 |
| Actual Estimates/Historical Data | 2020-2025 |
| Forecast Period | 2026 - 2030 - 2035 |
| Market Representation | Revenue in USD Billion and CAGR from 2026 to 2035 |
| Segments Covered | Phase, Therapeutic Area, Industry |
| Regional Scope | Asia-Pacific, Western Europe, Eastern Europe, North America, South America, Middle East, Africa |
| Country Scope | The countries covered in the report are Australia, Brazil, China, France, Germany, India, ... |
| Key Companies Profiled | Dassault Systemes SE; Evotec A.G; Evidera; Certara Inc.; Simulations Plus Inc.; Insilico Medicine Inc.; AnyLogic Company; Biomax Informatics; GNS Healthcare Inc.; 4P-Pharma; Nuventra Pharma Sciences; Archimedes; Novadiscovery Sas; Rosa & Co.; In Silico Biosciences; Leadscope; Biognos Ab; BioNova; Immunetrics Inc.; InSilicoTrials; Physiomics plc; Entelos; InhibOx; Selvita; Abzena Limited |
| Customization Scope | Request for Customization |
| Pricing And Purchase Options | Explore Purchase Options |
