
In Vitro Cancer Diagnostics Market Report 2026
Global Outlook – By Product Type (Kits, Instruments, Reagents, Software, Consumables), By Technology (Polymerase Chain Reaction (PCR), Next-Generation Sequencing (NGS), Fluorescence In Situ Hybridization (FISH), Immunohistochemistry, Other Technologies), By Application (Breast Cancer, Lung Cancer, Colorectal Cancer, Prostate Cancer, Other Applications), By End User (Hospitals, Diagnostic Laboratories, Research Institutes, Other End Users) – Market Size, Trends, Strategies, and Forecast to 2035
In Vitro Cancer Diagnostics Market Overview
• In Vitro Cancer Diagnostics market size has reached to $11.49 billion in 2025 • Expected to grow to $16.78 billion in 2030 at a compound annual growth rate (CAGR) of 7.9% • Growth Driver: The Rising Adoption Of Liquid Biopsy Technologies Is Driving The In Vitro Cancer Diagnostics Market Through Non-Invasive Detection • Market Trend: Enhancing Cancer Diagnosis With AI-Driven Digital Pathology Solution • North America was the largest region in 2025 and Asia-Pacific is the fastest growing region.What Is Covered Under In Vitro Cancer Diagnostics Market?
In vitro cancer diagnostics refers to laboratory tests performed on samples such as blood, tissue, or other body fluids to detect, monitor, or evaluate cancer outside the human body. These diagnostics use techniques such as molecular assays, biomarkers, and genetic profiling to identify cancer presence or progression. They support early detection, accurate diagnosis, and informed treatment planning for cancer patients. The main product types of in vitro cancer diagnostics are kits, instruments, reagents, software, and consumables. Kits refer to ready-to-use sets of reagents and materials designed for musculoskeletal testing, enabling the detection of biomarkers, supporting diagnosis, monitoring disease progression, and assisting in treatment evaluation. The various technologies include polymerase chain reaction (PCR), next-generation sequencing (NGS), fluorescence in situ hybridization (FISH), immunohistochemistry, and others. These are used for various applications, including breast cancer, lung cancer, colorectal cancer, prostate cancer, and other applications, with end-users comprising hospitals, diagnostic laboratories, research institutes, home care settings, and others.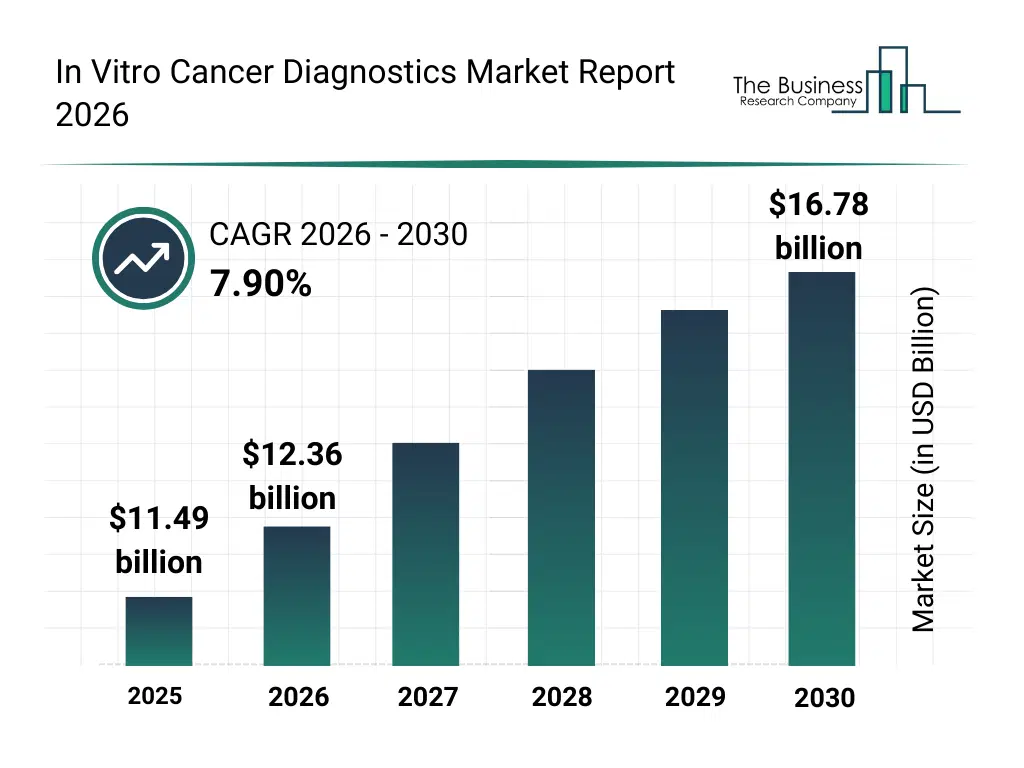
What Is The In Vitro Cancer Diagnostics Market Size and Share 2026?
The in vitro cancer diagnostics market size has grown strongly in recent years. It will grow from $11.49 billion in 2025 to $12.36 billion in 2026 at a compound annual growth rate (CAGR) of 7.6%. The growth in the historic period can be attributed to rising cancer prevalence, adoption of molecular diagnostics, development of immunoassay kits, expansion of diagnostic laboratories, advancements in reagent technologies.What Is The In Vitro Cancer Diagnostics Market Growth Forecast?
The in vitro cancer diagnostics market size is expected to see strong growth in the next few years. It will grow to $16.78 billion in 2030 at a compound annual growth rate (CAGR) of 7.9%. The growth in the forecast period can be attributed to AI-enabled predictive analytics, expansion in personalized oncology, development of high-throughput sequencing platforms, growth in point-of-care diagnostics, adoption of cloud-based diagnostic software. Major trends in the forecast period include biomarker-based detection, genetic profiling tools, multiplex assay development, integration with laboratory information systems (lims), rapid diagnostic kits.Global In Vitro Cancer Diagnostics Market Segmentation
1) By Product Type: Kits, Instruments, Reagents, Software, Consumables 2) By Technology: Polymerase Chain Reaction (PCR), Next-Generation Sequencing (NGS), Fluorescence In Situ Hybridization (FISH), Immunohistochemistry, Other Technologies 3) By Application: Breast Cancer, Lung Cancer, Colorectal Cancer, Prostate Cancer, Other Applications 4) By End User: Hospitals, Diagnostic Laboratories, Research Institutes, Other End Users Subsegments: 1) By Kits: Immunoassay Kits, Polymerase Chain Reaction (PCR) Kits, Enzyme-Linked Immunosorbent Assay (ELISA) Kits, Flow Cytometry Kits 2) By Instruments: Polymerase Chain Reaction (PCR) Instruments, Sequencers, Flow Cytometers, Imaging Systems 3) By Reagents: Enzymes, Buffers, Probes, Stains 4) By Software: Data Analysis Software, Imaging Software, Laboratory Information Management System (LIMS) Software, Diagnostic Reporting Software 5) By Consumables: Microplates, Pipette Tips, Tubes, SlidesWhat Is The Driver Of The In Vitro Cancer Diagnostics Market?
The rising adoption of liquid biopsy technologies is expected to propel the growth of the in vitro cancer diagnostics market going forward. Liquid biopsy technologies refer to minimally invasive diagnostic methods that detect and analyze biomarkers, such as circulating tumor DNA (ctDNA), circulating tumor cells (CTCs), or exosomes, present in body fluids like blood, urine, or saliva, to monitor disease presence, progression, or response to treatment, particularly in cancer. Liquid biopsy technologies are expanding due to advances in isolating and analyzing circulating tumor DNA and circulating tumor cells from blood and other body fluids. In vitro cancer diagnostic solutions are witnessing increased demand as the rapid expansion of liquid biopsy technologies pushes companies to adopt non-invasive and real-time molecular profiling tools for cancer detection and treatment guidance. For instance, in May 2025, according to NHS England, a UK-based public healthcare system, the new liquid biopsy blood test-first rollout in the UK is expected to benefit up to 15,000 patients with suspected lung cancer and 5,000 women with advanced breast cancer annually, scaling rapidly from approximately 2,200 patients tested during the early 2025 rollout to a projected 20,000 patients per year by 2028. Therefore, the rising adoption of liquid biopsy technologies is driving the demand for the in vitro cancer diagnostics industry.Key Players In The Global In Vitro Cancer Diagnostics Market
Major companies operating in the in vitro cancer diagnostics market are F. Hoffmann-La Roche Ltd, Danaher Corporation, Abbott Laboratories, Thermo Fisher Scientific, Siemens Healthineers AG, Illumina Inc, Hologic Inc, Qiagen N.V, Bio-Rad Laboratories Inc, Agilent Technologies, Becton, Dickinson and Company (BD), Sysmex Corporation, LabCorp Holding Corp., Guardant Health, Quest Diagnostics, Exact Sciences, Myriad Genetics Inc, Natera Inc., NeoGenomics Laboratories, NanoString Technologies Inc, Caris Life Sciences, Transasia Bio-Medicals Ltd, Autobio Diagnostics Co Ltd, QuidelOrtho Corporation, Cancer Diagnostics Inc, Predicine Inc, Strata Oncology, Biocept Inc, Lucence DiagnosticsGlobal In Vitro Cancer Diagnostics Market Trends and Insights
Major companies operating in the in vitro cancer diagnostics market are focusing on developing AI-driven cancer diagnostics to enhance patient care and advance personalized healthcare. AI-driven cancer diagnostics refers to the use of artificial intelligence to analyze medical and molecular data from patient samples, imaging, or genetic tests, enabling more accurate detection, diagnosis, and prediction of cancer. For instance, in September 2024, Roche, a Switzerland-based pharmaceutical company, launched the Digital Pathology Open Environment, a collaborative platform that securely integrates advanced AI-based image analysis tools from Roche and multiple third-party developers. The platform enables pathologists to access innovative algorithms within Roche’s Navify Digital Pathology enterprise software, improving diagnostic precision and workflow efficiency. By promoting collaboration and data sharing, the platform accelerates innovation in cancer diagnostics and research, supports precision medicine, and facilitates targeted treatments, ultimately broadening access to cutting-edge digital pathology tools worldwide.What Are Latest Mergers And Acquisitions In The In Vitro Cancer Diagnostics Market?
In February 2024, Veracyte Inc., a US-based molecular diagnostics company, acquired C2i Genomics, Inc. for an undisclosed amount. Through this acquisition, Veracyte aims to expand its role across the cancer care continuum by adding AI-driven whole-genome minimal residual disease testing to improve treatment monitoring and disease recurrence detection. C2i Genomics Inc. is a US-based biotechnology company that offers in vitro cancer diagnostics.Regional Outlook
North America was the largest region in the in vitro cancer diagnostics market in 2025. Asia-Pacific is expected to be the fastest-growing region in the forecast period. The regions covered in this market report are Asia-Pacific, South East Asia, Western Europe, Eastern Europe, North America, South America, Middle East, Africa. The countries covered in this market report are Australia, Brazil, China, France, Germany, India, Indonesia, Japan, Taiwan, Russia, South Korea, UK, USA, Canada, Italy, SpainWhat Defines the In Vitro Cancer Diagnostics Market?
The in vitro cancer diagnostics market consists of revenues earned by entities by providing services such as molecular diagnostic testing, biopsy and cytology analysis, companion diagnostic services, and next-generation sequencing (NGS) services. The market value includes the value of related goods sold by the service provider or included within the service offering. The in vitro cancer diagnostics market also includes sales of cancer biomarker kits, molecular diagnostic assays, flow cytometry kits, tissue-based diagnostic kits, and genomic testing panels. Values in this market are ‘factory gate’ values, that is the value of goods sold by the manufacturers or creators of the goods, whether to other entities (including downstream manufacturers, wholesalers, distributors and retailers) or directly to end customers. The value of goods in this market includes related services sold by the creators of the goods.How is Market Value Defined and Measured?
The market value is defined as the revenues that enterprises gain from the sale of goods and/or services within the specified market and geography through sales, grants, or donations in terms of the currency (in USD unless otherwise specified). The revenues for a specified geography are consumption values that are revenues generated by organizations in the specified geography within the market, irrespective of where they are produced. It does not include revenues from resales along the supply chain, either further along the supply chain or as part of other products.In Vitro Cancer Diagnostics Market Report Forecast Analysis
| Report Attribute | Details |
|---|---|
| Market Size Value In 2026 | $12.36 billion |
| Revenue Forecast In 2035 | $16.78 billion |
| Growth Rate | CAGR of 7.6% from 2026 to 2035 |
| Base Year For Estimation | 2025 |
| Actual Estimates/Historical Data | 2020-2025 |
| Forecast Period | 2026 - 2030 - 2035 |
| Market Representation | Revenue in USD Billion and CAGR from 2026 to 2035 |
| Segments Covered | Product Type, Technology, Application, End User |
| Regional Scope | Asia-Pacific, Western Europe, Eastern Europe, North America, South America, Middle East, Africa |
| Country Scope | The countries covered in the report are Australia, Brazil, China, France, Germany, India, ... |
| Key Companies Profiled | F. Hoffmann-La Roche Ltd, Danaher Corporation, Abbott Laboratories, Thermo Fisher Scientific, Siemens Healthineers AG, Illumina Inc, Hologic Inc, Qiagen N.V, Bio-Rad Laboratories Inc, Agilent Technologies, Becton, Dickinson and Company (BD), Sysmex Corporation, LabCorp Holding Corp., Guardant Health, Quest Diagnostics, Exact Sciences, Myriad Genetics Inc, Natera Inc., NeoGenomics Laboratories, NanoString Technologies Inc, Caris Life Sciences, Transasia Bio-Medicals Ltd, Autobio Diagnostics Co Ltd, QuidelOrtho Corporation, Cancer Diagnostics Inc, Predicine Inc, Strata Oncology, Biocept Inc, Lucence Diagnostics |
| Customization Scope | Request for Customization |
| Pricing And Purchase Options | Explore Purchase Options |
