
In-Vitro Diagnostics Instruments Market Report 2026
Global Outlook – By Product Type (Clinical Chemistry, Immunoassay, Molecular Diagnostics, Hematology, Microbiology, Coagulation, Other Product Types), By Application (Infectious Diseases, Cancer, Cardiology, Nephrology, Autoimmune Diseases, Other Applications), By End User (Hospitals And Diagnostic Laboratories, Academic And Research Institutes, Point-Of-Care Settings, Blood Banks, Pharmaceutical And Biotechnology Companies, Other Healthcare Facilities) – Market Size, Trends, Strategies, and Forecast to 2035
In-Vitro Diagnostics Instruments Market Overview
• In-Vitro Diagnostics Instruments market size has reached to $61.09 billion in 2025 • Expected to grow to $99.44 billion in 2030 at a compound annual growth rate (CAGR) of 10.3% • Growth Driver: The Rising Prevalence Of Chronic And Lifestyle-Related Diseases Is Driving The In Vitro Diagnostics Instruments Market Through Early Detection • Market Trend: Advancing Precision Diagnostics With Innovative Digital PCR Technology • North America was the largest region in 2025 and Asia-Pacific is the fastest growing region.What Is Covered Under In-Vitro Diagnostics Instruments Market?
In-vitro diagnostics instruments are medical devices used to analyze biological samples, such as blood, urine, or tissue, outside the human body. Their primary purpose is to detect, diagnose, monitor, and manage diseases or medical conditions by providing accurate and timely laboratory test results. These instruments support clinical decision-making and personalized patient care. The main product types of in-vitro diagnostic instruments are clinical chemistry, immunoassay, molecular diagnostics, hematology, microbiology, coagulation, and others. Clinical chemistry refers to digital tools that use AI to analyze blood, urine, and other body fluid samples, helping detect biochemical abnormalities, improve diagnostic accuracy, and support treatment planning. These instruments are primarily applied in areas such as infectious diseases, cancer, cardiology, nephrology, autoimmune diseases, and other applications, and cater to a wide range of end users, including hospitals and diagnostic laboratories, academic and research institutes, point-of-care settings, blood banks, pharmaceutical and biotechnology companies, and others.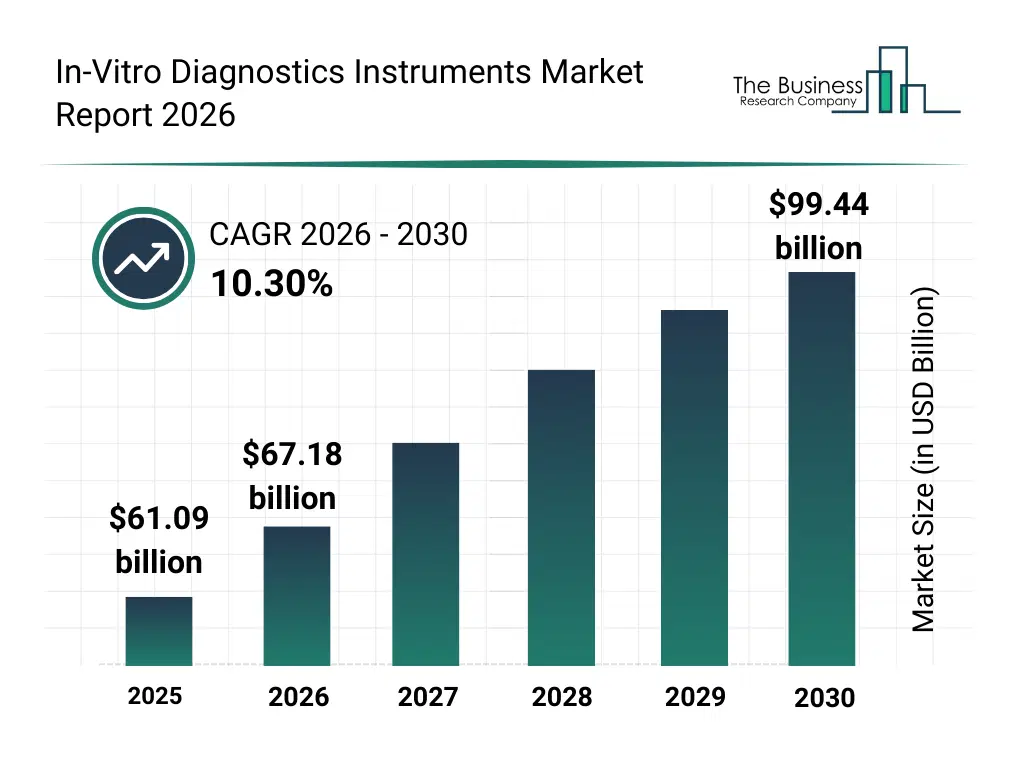
What Is The In-Vitro Diagnostics Instruments Market Size and Share 2026?
The in-vitro diagnostics instruments market size has grown strongly in recent years. It will grow from $61.09 billion in 2025 to $67.18 billion in 2026 at a compound annual growth rate (CAGR) of 10.0%. The growth in the historic period can be attributed to rising prevalence of chronic diseases, increasing hospital lab infrastructure, advances in diagnostic reagents, growing awareness for early disease detection, government healthcare initiatives.What Is The In-Vitro Diagnostics Instruments Market Growth Forecast?
The in-vitro diagnostics instruments market size is expected to see rapid growth in the next few years. It will grow to $99.44 billion in 2030 at a compound annual growth rate (CAGR) of 10.3%. The growth in the forecast period can be attributed to technological advancements in molecular diagnostics, growth of personalized medicine, integration of AI in diagnostics, expansion of emerging markets healthcare, demand for home-based diagnostic devices. Major trends in the forecast period include point-of-care testing expansion, personalized diagnostics, rapid molecular testing adoption, remote patient monitoring integration, miniaturized lab instruments.Global In-Vitro Diagnostics Instruments Market Segmentation
1) By Product Type: Clinical Chemistry, Immunoassay, Molecular Diagnostics, Hematology, Microbiology, Coagulation, Other Product Types 2) By Application: Infectious Diseases, Cancer, Cardiology, Nephrology, Autoimmune Diseases, Other Applications 3) By End User: Hospitals And Diagnostic Laboratories, Academic And Research Institutes, Point-Of-Care Settings, Blood Banks, Pharmaceutical And Biotechnology Companies, Other Healthcare Facilities Subsegments: 1) By Clinical Chemistry: Automated Clinical Chemistry Analyzers, Semi-Automated Clinical Chemistry Analyzers, Point-Of-Care Clinical Chemistry Devices 2) By Immunoassay: Enzyme-Linked Immunosorbent Assay (ELISA) Systems, Chemiluminescence Immunoassay (CLIA) Analyzers, Radioimmunoassay (RIA) Systems 3) By Molecular Diagnostics: Polymerase Chain Reaction (PCR) Systems, Next-Generation Sequencing (NGS) Platforms, Deoxyribonucleic Acid Microarrays, Isothermal Amplification Devices 4) By Hematology: Automated Hematology Analyzers, Manual Hematology Analyzers, Hemostasis Analyzers, Coagulation Analyzers 5) By Microbiology: Microbial Identification Systems, Antibiotic Susceptibility Testing (AST) Systems, Blood Culture Systems, Automated Microbial Analyzers. 6) By Coagulation: Coagulometers, Platelet Function Analyzers, Thromboelastography (TEG) Systems, Point-Of-Care Coagulation Devices 7) By Other Product Types: Point-Of-Care Diagnostic Devices, Miscellaneous Laboratory InstrumentsWhat Is The Driver Of The In-Vitro Diagnostics Instruments Market?
The rising prevalence of chronic and lifestyle-related diseases is expected to propel the growth of the in vitro diagnostics instruments market going forward. Chronic and lifestyle-related diseases refer to long-term health conditions that are primarily influenced by daily habits, behaviors, and environmental factors, such as heart disease, diabetes, obesity, and certain cancers. Chronic and lifestyle-related diseases are rising due to sedentary lifestyles and unhealthy dietary habits. The in vitro diagnostics instruments solutions are witnessing increased demand as the rapid expansion of chronic and lifestyle-related diseases pushes companies to adopt early detection and monitoring tools for better disease management. For instance, in 2024, according to the American Action Forum, a US-based non-profit, center-right think tank, chronic diseases remain a significant public health concern in the U.S., with nearly 60 percent of adults living with at least one condition, 40 percent managing multiple conditions, and prevalence expected to nearly double by 2050. Further, the chronic disease treatment market is expected to reach approximately USD 38.02 billion by 2034, indicating a significant rise from USD 9.74 billion in 2025. Therefore, the rising prevalence of chronic and lifestyle-related diseases is driving the demand for in vitro diagnostics instruments market.Key Players In The Global In-Vitro Diagnostics Instruments Market
Major companies operating in the in-vitro diagnostics instruments market are F. Hoffmann-La Roche Ltd, Danaher Corporation, Abbott Laboratories, Thermo Fisher Scientific, Siemens Healthineers AG, Becton Dickinson and Company, Sysmex Corporation, Bio-Rad Laboratories Inc, Agilent Technologies, Hologic Inc, Qiagen N.V., Illumina Inc, Ortho Clinical Diagnostics, Beckman Coulter Inc, Mindray Medical International Limited, Horiba Ltd, Randox Laboratories Ltd, DiaSorin S.p.A., Werfen S.A., Trinity Biotech plcGlobal In-Vitro Diagnostics Instruments Market Trends and Insights
Major companies operating in the in-vitro diagnostics instruments market are focusing on developing advanced products, such as digital polymerase chain reaction (dPCR) systems, to enable highly precise and sensitive nucleic acid quantification for advanced diagnostics. Digital polymerase chain reaction (dPCR) systems are advanced diagnostic tools that precisely quantify DNA or RNA molecules by partitioning samples into thousands of individual reactions for highly accurate and sensitive detection. For instance, in September 2024, QIAGEN N.V., a Netherlands-based biotechnology company, launched the QIAcuityDx Digital PCR System, expanding its digital PCR portfolio into clinical diagnostics with a focus on oncology testing. It is a fully integrated, automated system designed specifically for clinical testing in oncology, featuring a cartridge-based workflow that minimizes hands-on time. The QIAcuityDx includes automated analysis and reporting software, enabling rapid and accurate detection of genetic biomarkers for cancer diagnosis and therapy monitoring without extensive operator intervention.What Are Latest Mergers And Acquisitions In The In-Vitro Diagnostics Instruments Market?
In January 2025, Fremman Ltd., a UK-based investment company, acquired a majority stake in DIESSE Diagnostica Senese S.p.A. for an undisclosed amount. Through this acquisition, Fremman Ltd aims to further internationalize DIESSE Diagnostica Senese's business, enhance its market leadership in specialty in vitro diagnostics, and support ongoing innovation to improve global diagnostic capabilities and patient outcomes. DIESSE Diagnostica Senese S.p.A. is an Italy-based medical equipment company that offers in vitro diagnostic instruments.Regional Outlook
North America was the largest region in the in-vitro diagnostics instruments market in 2025. Asia-Pacific is expected to be the fastest-growing region in the forecast period. The regions covered in this market report are Asia-Pacific, South East Asia, Western Europe, Eastern Europe, North America, South America, Middle East, Africa. The countries covered in this market report are Australia, Brazil, China, France, Germany, India, Indonesia, Japan, Taiwan, Russia, South Korea, UK, USA, Canada, Italy, SpainWhat Defines the In-Vitro Diagnostics Instruments Market?
The in-vitro diagnostics instruments market consists of sales of point-of-care testing devices, flow cytometry instruments, histopathology and cytology instruments, and molecular point-of-care systems. Values in this market are ‘factory gate’ values, that is, the value of goods sold by the manufacturers or creators of the goods, whether to other entities (including downstream manufacturers, wholesalers, distributors, and retailers) or directly to end customers. The value of goods in this market includes related services sold by the creators of the goods.How is Market Value Defined and Measured?
The market value is defined as the revenues that enterprises gain from the sale of goods and/or services within the specified market and geography through sales, grants, or donations in terms of the currency (in USD unless otherwise specified). The revenues for a specified geography are consumption values that are revenues generated by organizations in the specified geography within the market, irrespective of where they are produced. It does not include revenues from resales along the supply chain, either further along the supply chain or as part of other products.In-Vitro Diagnostics Instruments Market Report Forecast Analysis
| Report Attribute | Details |
|---|---|
| Market Size Value In 2026 | $67.18 billion |
| Revenue Forecast In 2035 | $99.44 billion |
| Growth Rate | CAGR of 10.0% from 2026 to 2035 |
| Base Year For Estimation | 2025 |
| Actual Estimates/Historical Data | 2020-2025 |
| Forecast Period | 2026 - 2030 - 2035 |
| Market Representation | Revenue in USD Billion and CAGR from 2026 to 2035 |
| Segments Covered | Product Type, Application, End User |
| Regional Scope | Asia-Pacific, Western Europe, Eastern Europe, North America, South America, Middle East, Africa |
| Country Scope | The countries covered in the report are Australia, Brazil, China, France, Germany, India, ... |
| Key Companies Profiled | F. Hoffmann-La Roche Ltd, Danaher Corporation, Abbott Laboratories, Thermo Fisher Scientific, Siemens Healthineers AG, Becton Dickinson and Company, Sysmex Corporation, Bio-Rad Laboratories Inc, Agilent Technologies, Hologic Inc, Qiagen N.V., Illumina Inc, Ortho Clinical Diagnostics, Beckman Coulter Inc, Mindray Medical International Limited, Horiba Ltd, Randox Laboratories Ltd, DiaSorin S.p.A., Werfen S.A., Trinity Biotech plc |
| Customization Scope | Request for Customization |
| Pricing And Purchase Options | Explore Purchase Options |
