
Inactivated Influenza Vaccine Market Report 2026
Global Outlook – By Type Of Vaccine (Trivalent Inactivated Influenza Vaccine (TIIV), Quadrivalent Inactivated Influenza Vaccine (QIIV)), By Application (Children, Adolescence, Adults, Elderly), By Distribution Channel (Hospital Pharmacies, Retail Pharmacies, Online Pharmacies, Public Health Clinics), By End-User (Healthcare Providers, Individuals, Government Organizations, Pharmaceutical Companies) – Market Size, Trends, Strategies, and Forecast to 2035
Inactivated Influenza Vaccine Market Overview
• Inactivated Influenza Vaccine market size has reached to $7.14 billion in 2025 • Expected to grow to $10.55 billion in 2030 at a compound annual growth rate (CAGR) of 8.1% • Growth Driver: Expansion Of Government Vaccination Programs Driving Growth Of The Market Due To Enhancing Public Health Awareness • Market Trend: Focus On Quadrivalent Inactivated Influenza Vaccines To Enhance Protection And Vaccine Efficacy In The Market • North America was the largest region in 2025 and Asia-Pacific is the fastest growing region.What Is Covered Under Inactivated Influenza Vaccine Market?
Inactivated influenza vaccine refers to a type of flu vaccine that contains viruses that have been killed or inactivated. It works by prompting the immune system to produce antibodies against the influenza virus. This vaccine is administered via injection to help prevent seasonal influenza infections. The main types of vaccines in the inactivated influenza vaccine market are trivalent inactivated influenza vaccine (TIIV) and quadrivalent inactivated influenza vaccine (QIIV). Trivalent inactivated influenza vaccine refers to a flu shot containing three killed virus strains, two influenza A and one influenza B, to protect against seasonal influenza. The various applications include children, adolescence, adults, and the elderly and are distributed through various channels, including hospital pharmacies, retail pharmacies, online pharmacies, and public health clinics. The different end-users encompass healthcare providers, individuals, government organizations, and pharmaceutical companies.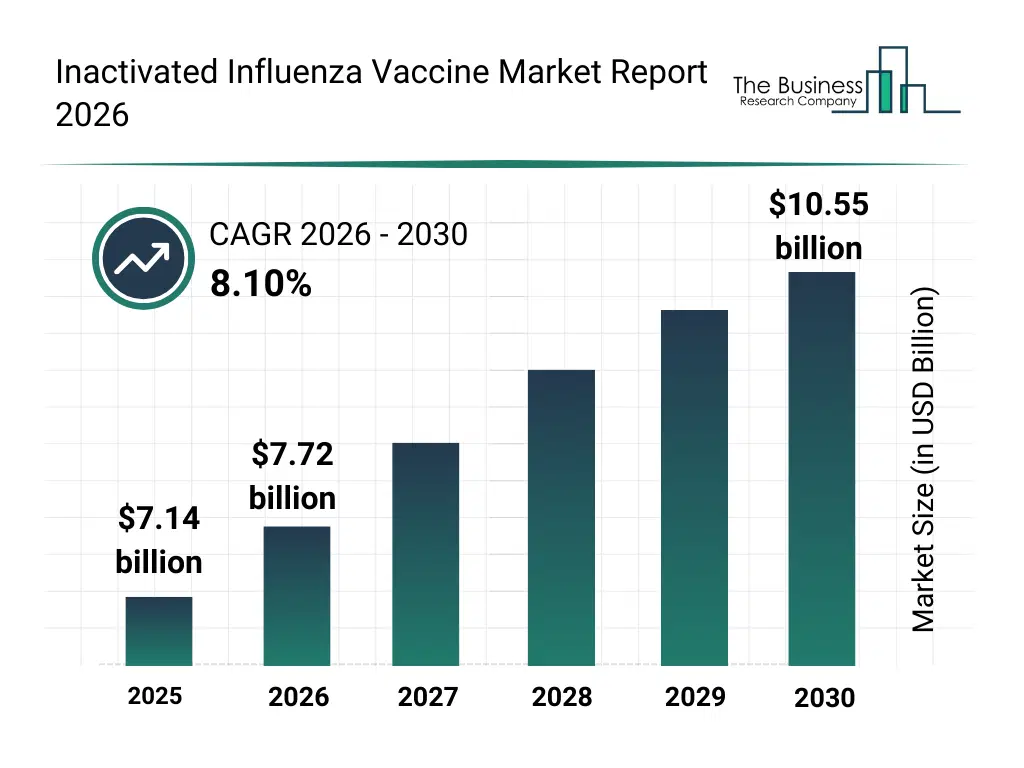
What Is The Inactivated Influenza Vaccine Market Size and Share 2026?
The inactivated influenza vaccine market size has grown strongly in recent years. It will grow from $7.14 billion in 2025 to $7.72 billion in 2026 at a compound annual growth rate (CAGR) of 8.2%. The growth in the historic period can be attributed to increasing seasonal influenza incidence, expansion of immunization programs, rising awareness of preventive healthcare, availability of egg-based vaccine production, growth of public health initiatives.What Is The Inactivated Influenza Vaccine Market Growth Forecast?
The inactivated influenza vaccine market size is expected to see strong growth in the next few years. It will grow to $10.55 billion in 2030 at a compound annual growth rate (CAGR) of 8.1%. The growth in the forecast period can be attributed to increasing focus on pandemic preparedness, rising investments in cell-based and recombinant vaccines, expansion of adult and elderly immunization coverage, growing government procurement of vaccines, increasing innovation in vaccine manufacturing technologies. Major trends in the forecast period include increasing adoption of quadrivalent influenza vaccines, rising focus on high-dose and adjuvanted vaccines, expansion of seasonal vaccination programs, growing emphasis on cold-chain optimization, enhanced focus on vaccine manufacturing scalability.Global Inactivated Influenza Vaccine Market Segmentation
1) By Type Of Vaccine: Trivalent Inactivated Influenza Vaccine (TIIV), Quadrivalent Inactivated Influenza Vaccine (QIIV) 2) By Application: Children, Adolescence, Adults, Elderly 3) By Distribution Channel: Hospital Pharmacies, Retail Pharmacies, Online Pharmacies, Public Health Clinics 4) By End-User: Healthcare Providers, Individuals, Government Organizations, Pharmaceutical Companies Subsegments: 1) Trivalent Inactivated Influenza Vaccine (TIIV): Egg-based Trivalent Inactivated Influenza Vaccine, Cell-based Trivalent Inactivated Influenza Vaccine, Adjuvanted Trivalent Inactivated Influenza Vaccine, High-dose Trivalent Inactivated Influenza Vaccine 2) Quadrivalent Inactivated Influenza Vaccine (QIIV): Egg-based Quadrivalent Inactivated Influenza Vaccine, Cell-based Quadrivalent Inactivated Influenza Vaccine, Adjuvanted Quadrivalent Inactivated Influenza Vaccine, Recombinant Quadrivalent Inactivated Influenza Vaccine, Pediatric Quadrivalent Inactivated Influenza VaccineWhat Is The Driver Of The Inactivated Influenza Vaccine Market?
The rising government vaccination programs are expected to propel the growth of the inactivated influenza vaccine market going forward. ?Government vaccination programs are expanding due to increasing public health awareness among populations, which leads to higher demand for immunization services and encourages governments to enhance vaccine accessibility and outreach initiatives. Government vaccination programs facilitate the administration of inactivated influenza vaccines by providing free or subsidized access to high-risk groups, ensuring equitable immunization coverage, and reducing the burden of seasonal influenza. For instance, in December 2023, the National Health Service, a UK-based government agency, initiated an NHS vaccine strategy for immunization goals. The strategy includes plans for community-based vaccination services, particularly targeting underserved and marginalized groups. Therefore, the rising government vaccination programs are driving the growth of the inactivated influenza vaccine industry.Key Players In The Global Inactivated Influenza Vaccine Market
Major companies operating in the inactivated influenza vaccine market are Sanofi S.A., GlaxoSmithKline plc, CSL Limited, Viatris Inc., Zydus Lifesciences Limited, Sinovac Biotech Ltd., Bharat Biotech International Limited, Hualan Biological Bacterin Co. Ltd., Changchun Biological Products Co. Ltd., Dalian Aleph Biomedical Co. Ltd., Jiangsu GDK Biotechnology Co. Ltd., KM Biologics Co. Ltd., JoinHub Pharma, Seqirus (CSL Influenza Division), Serum Institute of India Pvt. Ltd., Biological E. Limited, Shenzhen Sanofi Pasteur Biologicals Co. Ltd., Beijing Tiantan Biological Products Co. Ltd., Wuhan Institute of Biological Products Co. Ltd., Lanzhou Institute of Biological Products Co. Ltd., Chengdu Institute of Biological Products Co. Ltd., Sinopharm CNBG, Takeda Pharmaceutical Company Limited, PfizerGlobal Inactivated Influenza Vaccine Market Trends and Insights
Major companies operating in the inactivated influenza vaccine market are focusing on developing quadrivalent inactivated influenza vaccines to provide broader protection against four strains of the influenza virus. A quadrivalent inactivated influenza vaccine is a flu shot that protects against four different flu viruses. For instance, in February 2025, Zydus Lifesciences, an India-based pharmaceutical company, launched VaxiFlu-4, a quadrivalent inactivated influenza vaccine designed to offer seasonal protection against four new virus strains. VaxiFlu-4 provides broader protection by covering both influenza A and B strains, reducing the risk of vaccine mismatch. The vaccine aims to address seasonal flu outbreaks with improved immune response tailored to regional epidemiological patterns.What Are Latest Mergers And Acquisitions In The Inactivated Influenza Vaccine Market?
In July 2024, GSK plc, a UK-based pharmaceutical company specializing in vaccines and biopharmaceuticals, acquired full rights to develop, manufacture, and commercialize mRNA-based influenza and COVID-19 vaccines from CureVac N.V. for an undisclosed amount. With this acquisition, GSK aims to accelerate its mRNA vaccine development, leveraging CureVac’s research to enhance seasonal flu and pandemic preparedness strategies. CureVac N.V. is a Germany-based biotechnology company focused on mRNA-based therapeutics, including vaccines for influenza and cancer immunotherapy.Regional Outlook
North America was the largest region in the inactivated influenza vaccine market in 2025. Asia-Pacific is expected to be the fastest-growing region in the forecast period. The regions covered in this market report are Asia-Pacific, South East Asia, Western Europe, Eastern Europe, North America, South America, Middle East, Africa. The countries covered in this market report are Australia, Brazil, China, France, Germany, India, Indonesia, Japan, Taiwan, Russia, South Korea, UK, USA, Canada, Italy, SpainWhat Defines the Inactivated Influenza Vaccine Market?
The inactivated influenza vaccine market consists of sales of adjuvanted inactivated influenza vaccines, preservative-free inactivated influenza vaccines, cell-based inactivated influenza vaccines, egg-based inactivated influenza vaccines, pediatric inactivated influenza vaccines, high-dose inactivated influenza vaccines for older adults, prefilled syringe vaccines, and multidose vial vaccines. Values in this market are ‘factory gate’ values, that is, the value of goods sold by the manufacturers or creators of the goods, whether to other entities (including downstream manufacturers, wholesalers, distributors, and retailers) or directly to end customers. The value of goods in this market includes related services sold by the creators of the goods.How is Market Value Defined and Measured?
The market value is defined as the revenues that enterprises gain from the sale of goods and/or services within the specified market and geography through sales, grants, or donations in terms of the currency (in USD unless otherwise specified). The revenues for a specified geography are consumption values that are revenues generated by organizations in the specified geography within the market, irrespective of where they are produced. It does not include revenues from resales along the supply chain, either further along the supply chain or as part of other products.Inactivated Influenza Vaccine Market Report Forecast Analysis
| Report Attribute | Details |
|---|---|
| Market Size Value In 2026 | $7.72 billion |
| Revenue Forecast In 2035 | $10.55 billion |
| Growth Rate | CAGR of 8.2% from 2026 to 2035 |
| Base Year For Estimation | 2025 |
| Actual Estimates/Historical Data | 2020-2025 |
| Forecast Period | 2026 - 2030 - 2035 |
| Market Representation | Revenue in USD Billion and CAGR from 2026 to 2035 |
| Segments Covered | Type Of Vaccine, Application, Distribution Channel, End-User |
| Regional Scope | Asia-Pacific, Western Europe, Eastern Europe, North America, South America, Middle East, Africa |
| Country Scope | The countries covered in the report are Australia, Brazil, China, France, Germany, India, ... |
| Key Companies Profiled | Sanofi S.A., GlaxoSmithKline plc, CSL Limited, Viatris Inc., Zydus Lifesciences Limited, Sinovac Biotech Ltd., Bharat Biotech International Limited, Hualan Biological Bacterin Co. Ltd., Changchun Biological Products Co. Ltd., Dalian Aleph Biomedical Co. Ltd., Jiangsu GDK Biotechnology Co. Ltd., KM Biologics Co. Ltd., JoinHub Pharma, Seqirus (CSL Influenza Division), Serum Institute of India Pvt. Ltd., Biological E. Limited, Shenzhen Sanofi Pasteur Biologicals Co. Ltd., Beijing Tiantan Biological Products Co. Ltd., Wuhan Institute of Biological Products Co. Ltd., Lanzhou Institute of Biological Products Co. Ltd., Chengdu Institute of Biological Products Co. Ltd., Sinopharm CNBG, Takeda Pharmaceutical Company Limited, Pfizer |
| Customization Scope | Request for Customization |
| Pricing And Purchase Options | Explore Purchase Options |
