
Infectious Disease Diagnostics Testing Market Report 2026
Global Outlook – By Type (Molecular Diagnostic Test, Point Of Care Tests (POCT) Infectious Disease Diagnostics Test), By Application (Respiratory, Human Immunodeficiency Virus (HIV), Healthcare-Associated Infections (HAIs), Sexual Health, Tropical Diseases), By End-User (Diagnostic Laboratories, Hospitals And Clinics) – Market Size, Trends, Strategies, and Forecast to 2035
Infectious Disease Diagnostics Testing Market Overview
• Infectious Disease Diagnostics Testing market size has reached to $42.24 billion in 2025 • Expected to grow to $65.49 billion in 2030 at a compound annual growth rate (CAGR) of 9% • Growth Driver: Impact Of Rising Respiratory Infections On The Infectious Disease Diagnostic Testing Market • Market Trend: Advanced Syndromic Testing Panels For Rapid Infectious Disease Diagnosis • North America was the largest region in 2025 and Asia-Pacific is the fastest growing region.What Is Covered Under Infectious Disease Diagnostics Testing Market?
Infectious disease diagnostics testing refers to the process of identifying the presence of pathogens, such as bacteria, viruses, fungi, or parasites, in a patient's body. These tests are crucial for diagnosing infectious diseases and guiding appropriate treatment decisions. Infectious disease diagnostics testing plays a vital role in controlling the spread of infectious diseases, guiding treatment decisions, and monitoring outbreaks. The main types of infectious disease diagnostics testing market molecular diagnostic tests, and point of care tests (POCT) infectious disease diagnostics tests. Molecular diagnostic testing refers to laboratory techniques that detect and analyze genetic material (DNA or RNA) to diagnose diseases, identify pathogens, and predict treatment responses. The various applications include respiratory, human immunodeficiency virus (HIV), healthcare-associated infections (HAIS), sexual health, tropical diseases, and liver, and are used by various end-users such as diagnostic laboratories, hospitals and clinics, academic research institutes, and other end-users.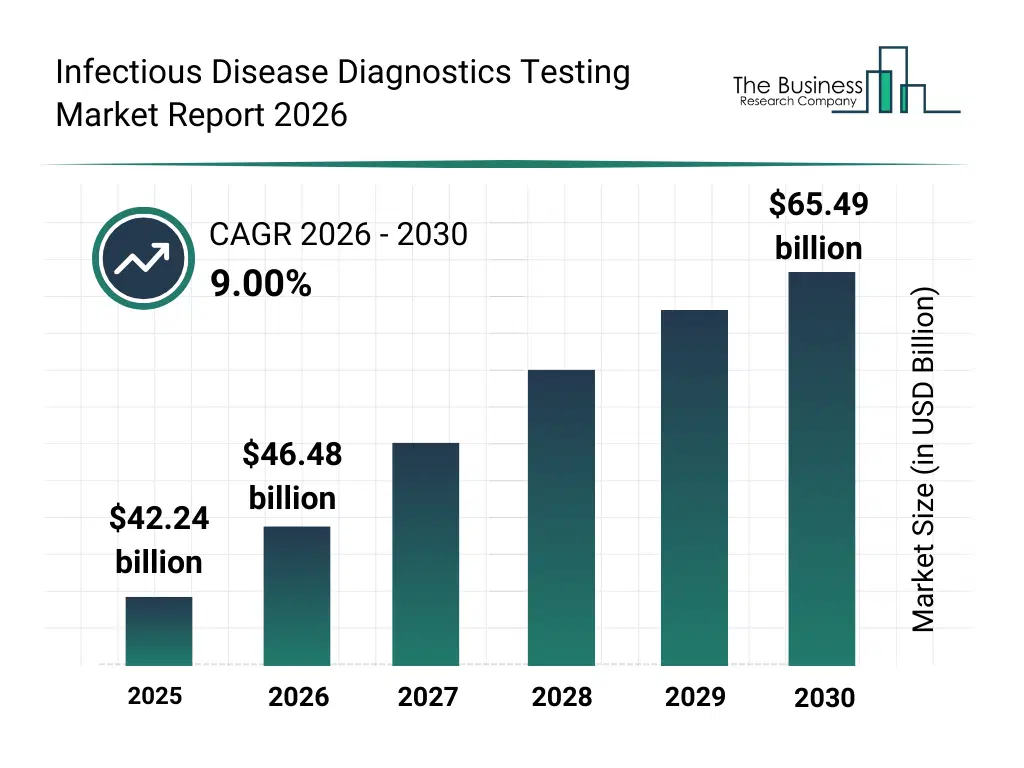
What Is The Infectious Disease Diagnostics Testing Market Size and Share 2026?
The infectious disease diagnostics testing market size has grown rapidly in recent years. It will grow from $42.24 billion in 2025 to $46.48 billion in 2026 at a compound annual growth rate (CAGR) of 10.0%. The growth in the historic period can be attributed to increasing prevalence of infectious diseases, rising incidence of respiratory infections, expansion of diagnostic laboratory infrastructure, growing awareness of early disease diagnosis, increased government focus on disease control.What Is The Infectious Disease Diagnostics Testing Market Growth Forecast?
The infectious disease diagnostics testing market size is expected to see strong growth in the next few years. It will grow to $65.49 billion in 2030 at a compound annual growth rate (CAGR) of 9.0%. The growth in the forecast period can be attributed to rising risk of global pandemics, increasing demand for rapid diagnostic solutions, growing investments in healthcare diagnostics, expanding disease surveillance programs, rising burden of tropical and viral diseases. Major trends in the forecast period include rising demand for rapid and early infectious disease detection, growing adoption of point of care infectious disease tests, increasing use of molecular diagnostics for pathogen identification, expansion of diagnostic testing for emerging and re-emerging infections, growing focus on large-scale disease screening and surveillance.Global Infectious Disease Diagnostics Testing Market Segmentation
1) By Type: Molecular Diagnostic Test, Point Of Care Tests (POCT) Infectious Disease Diagnostics Test 2) By Application: Respiratory, Human Immunodeficiency Virus (HIV), Healthcare-Associated Infections (HAIs), Sexual Health, Tropical Diseases 3) By End-User: Diagnostic Laboratories, Hospitals And Clinics Subsegments: 1) By Molecular Diagnostic Test: Polymerase Chain Reaction (PCR) Tests, Nucleic Acid Amplification Tests (NAATs) 2) By Point Of Care Tests (POCT) Infectious Disease Diagnostics Test: Lateral Flow Assays, Rapid Antigen Tests, Immunoassays, Portable PCRWhat Is The Driver Of The Infectious Disease Diagnostics Testing Market?
A rise in respiratory infections is expected to propel the growth of the infectious disease diagnostic testing market going forward. Respiratory infections affect the respiratory system, including the nose, throat, airways, and lungs. The increasing respiratory infections are due to viral and bacterial transmission through close contact with infected individuals, poor hygiene practices, crowded living conditions, and seasonal variations in weather patterns. Infectious disease diagnostic testing is essential for diagnosing and treating respiratory infections as it can precisely identify pathogens such as bacteria and viruses in patient samples. For instance, in February 2024, according to the Canadian Institute for Health Information, a Canada-based non-profit organization, hospitalizations for acute upper respiratory infections increased by 32% from 2,114 in 2022 to 2,797 cases in 2023. Therefore, the rise in respiratory infections is driving the infectious disease diagnostic testing market.Key Players In The Global Infectious Disease Diagnostics Testing Market
Major companies operating in the infectious disease diagnostics testing market are F. Hoffmann-La Roche Ltd., Thermo Fisher Scientific Inc., Abbott Laboratories, Danaher Corporation, Siemens Healthineers AG, Becton Dickinson And Company, Quest Diagnostics Incorporated, Grifols S.A., Hologic Inc., PerkinElmer Inc., bioMérieux SA, Quidel Corporation, Sysmex Corporation, Bio-Rad Laboratories Inc., QIAGEN N.V., DiaSorin SpA (Luminex), Seegene Inc., Ortho Clinical Diagnostics Inc., OraSure Technologies Inc., Meridian Bioscience Inc., Trinity Biotech PLCGlobal Infectious Disease Diagnostics Testing Market Trends and Insights
Major companies in the infectious disease diagnostic testing market are advancing innovative technologies to rapidly and reliably detect critical infections. Infectious disease diagnostics involves identifying pathogens, such as bacteria, viruses, or parasites, to guide diagnosis and treatment. For instance, in January 2025, QIAGEN, a Germany-based biotechnology company, launched new QIAstat-Dx syndromic testing panels in India, including the Gastrointestinal Panel 2 and Meningitis/Encephalitis Panel, alongside the previously authorized Respiratory SARS-CoV-2 Panel, all approved by India's Central Drugs Standard Control Organization (CDSCO). The QIAstat-Dx system uses multiplex real-time PCR with single-use cartridges to detect multiple pathogens simultaneously, delivering results in about an hour for gastrointestinal infections and 80 minutes for central nervous system infections. These panels enhance diagnostic speed and accuracy compared to traditional methods, enabling timely treatment, improving patient outcomes, and easing healthcare system burdens. The system also offers configurable setups and cloud connectivity for remote monitoring and efficient data management.What Are Latest Mergers And Acquisitions In The Infectious Disease Diagnostics Testing Market?
In July 2024, Roche, a Switzerland-based company, acquired the point-of-care technology business of LumiraDx for an undisclosed amount. With this acquisition, Roche aimed to accelerate its expansion into decentralized and primary-care testing by integrating LumiraDx’s innovative multi-assay platform capable of performing immunoassay, clinical chemistry, and, with future potential, molecular diagnostics. LumiraDx is a UK-based provider of infectious disease diagnostics testing.Regional Outlook
North America was the largest region in the infectious disease diagnostics testing market in 2025. Asia-Pacific is expected to be the fastest-growing region in the forecast period. The regions covered in this market report are Asia-Pacific, South East Asia, Western Europe, Eastern Europe, North America, South America, Middle East, Africa. The countries covered in this market report are Australia, Brazil, China, France, Germany, India, Indonesia, Japan, Taiwan, Russia, South Korea, UK, USA, Canada, Italy, SpainWhat Defines the Infectious Disease Diagnostics Testing Market?
The infectious disease diagnostics testing market consists of revenues earned by entities by providing services such as laboratory testing, point-of-care testing, molecular diagnostics, immunoassays, and rapid diagnostic tests. The market value includes the value of related goods sold by the service provider or included within the service offering. The infectious disease diagnostics testing market also includes sales of diagnostic kits, reagents, instruments, and software used for detecting and identifying infectious agents in patients. Values in this market are ‘factory gate’ values, that is, the value of goods sold by the manufacturers or creators of the goods, whether to other entities (including downstream manufacturers, wholesalers, distributors, and retailers) or directly to end customers. The value of goods in this market includes related services sold by the creators of the goods.How is Market Value Defined and Measured?
The market value is defined as the revenues that enterprises gain from the sale of goods and/or services within the specified market and geography through sales, grants, or donations in terms of the currency (in USD unless otherwise specified). The revenues for a specified geography are consumption values that are revenues generated by organizations in the specified geography within the market, irrespective of where they are produced. It does not include revenues from resales along the supply chain, either further along the supply chain or as part of other products.Infectious Disease Diagnostics Testing Market Report Forecast Analysis
| Report Attribute | Details |
|---|---|
| Market Size Value In 2026 | $46.48 billion |
| Revenue Forecast In 2035 | $65.49 billion |
| Growth Rate | CAGR of 10.0% from 2026 to 2035 |
| Base Year For Estimation | 2025 |
| Actual Estimates/Historical Data | 2020-2025 |
| Forecast Period | 2026 - 2030 - 2035 |
| Market Representation | Revenue in USD Billion and CAGR from 2026 to 2035 |
| Segments Covered | Type, Application, End-User |
| Regional Scope | Asia-Pacific, Western Europe, Eastern Europe, North America, South America, Middle East, Africa |
| Country Scope | The countries covered in the report are Australia, Brazil, China, France, Germany, India, ... |
| Key Companies Profiled | F. Hoffmann-La Roche Ltd., Thermo Fisher Scientific Inc., Abbott Laboratories, Danaher Corporation, Siemens Healthineers AG, Becton Dickinson And Company, Quest Diagnostics Incorporated, Grifols S.A., Hologic Inc., PerkinElmer Inc., bioMérieux SA, Quidel Corporation, Sysmex Corporation, Bio-Rad Laboratories Inc., QIAGEN N.V., DiaSorin SpA (Luminex), Seegene Inc., Ortho Clinical Diagnostics Inc., OraSure Technologies Inc., Meridian Bioscience Inc., Trinity Biotech PLC |
| Customization Scope | Request for Customization |
| Pricing And Purchase Options | Explore Purchase Options |
