
Influenza Diagnostic Market Report 2026
Global Outlook – By Traditional Diagnostic Tests (Rapid Influenza Diagnostic Tests (RIDT), Viral Culture, DFA, Serological Assays, Other Traditional Diagnostic Tests ), By Molecular Diagnostic Tests (Reverse Transcription Polymerase Chain Reaction (RT-PCR), Isothermal Nucleic Acid Amplification Tests (INAAT), Loop Mediated Isothermal Based Amplification Assays, Transcription Mediated Isothermal Based Amplification Assays, Other Isothermal Nucleic Acid Amplification Tests, Other Molecular Diagnostic Tests ), By End User (Hospitals And Clinical Laboratories, Diagnostic Reference Laboratories, Academic/Research Institutes, Other End Users) - Market Size, Trends, And Global Forecast 2026-2035
Influenza Diagnostic Market Overview
• Influenza Diagnostic market size has reached to $2.87 billion in 2025 • Expected to grow to $4.19 billion in 2030 at a compound annual growth rate (CAGR) of 7.8% • Growth Driver: Increased Research Funding Boosts Influenza Diagnostics • Market Trend: Innovative Products In Influenza Diagnostic Market Development • North America was the largest region in 2025 and Western Europe is the fastest growing region.What Is Covered Under Influenza Diagnostic Market?
Influenza diagnostic is the test used to diagnose influenza including viral culture, serology, rapid antigen testing, reverse transcription-polymerase chain reaction (RT-PCR), immunofluorescence assays, and rapid molecular assays. Influenza, commonly known as flu, is a contagious illness caused by the influenza virus. Three varieties of this virus affect people: type A, type B, and type C. Common symptoms include fever, sore throat, cough, body ache, fatigue, and headache. The market consists of revenues generated by establishments conducting diagnostics tests for influenza virus detection. The main traditional diagnostic test types of influenza diagnostics are rapid influenza diagnostic tests (RIDT), viral culture, DFA, serological assays and others. Rapid influenza diagnostic tests (RIDTs) are immunoassays that can detect the presence of influenza A and B viral nucleoprotein antigens in respiratory specimens and provide a qualitative result (positive vs. negative). The various molecular diagnostic tests include Reverse Transcription Polymerase Chain Reaction (RT-PCR), Isothermal Nucleic Acid Amplification Tests (INAAT), loop mediated isothermal based amplification assays, transcription mediated isothermal based amplification assays, transcription mediated isothermal based amplification assays, other isothermal nucleic acid amplification tests and are used by hospitals and clinical laboratories, diagnostic reference laboratories, academic/research institutes, other end users.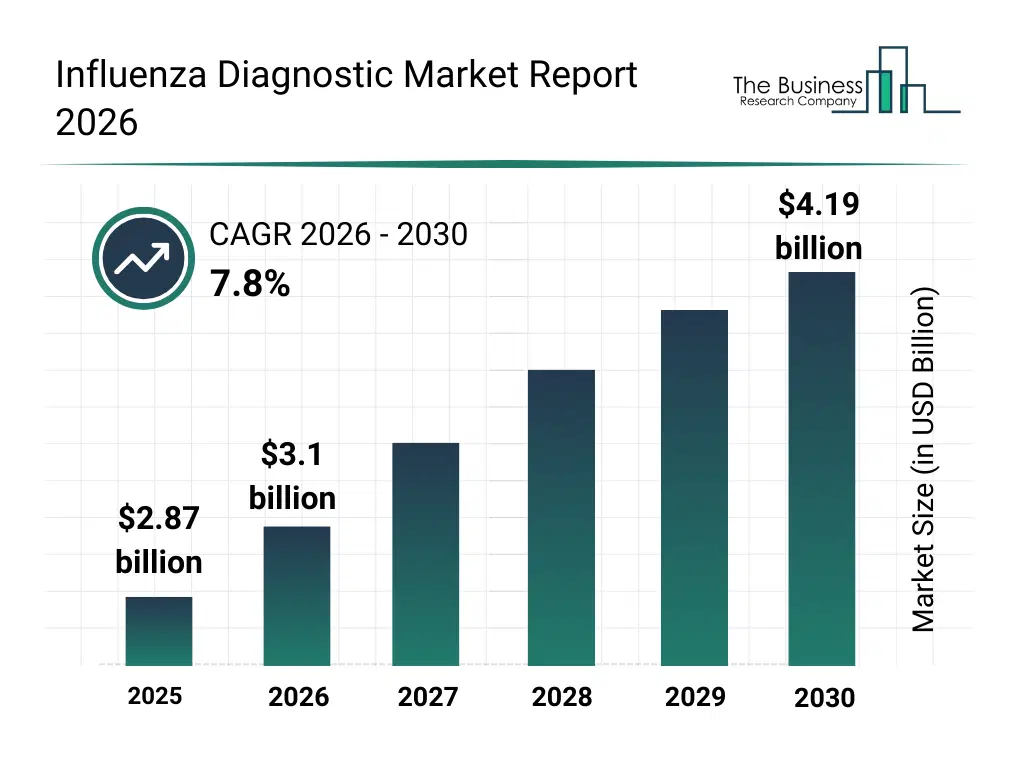
What Is The Influenza Diagnostic Market Size and Share 2026?
The influenza diagnostic market size has grown strongly in recent years. It will grow from $2.87 billion in 2025 to $3.1 billion in 2026 at a compound annual growth rate (CAGR) of 8.3%. The growth in the historic period can be attributed to seasonal influenza outbreaks, widespread use of rapid antigen tests, growth of hospital laboratory infrastructure, government influenza surveillance programs, increased awareness of early flu diagnosis.What Is The Influenza Diagnostic Market Growth Forecast?
The influenza diagnostic market size is expected to see strong growth in the next few years. It will grow to $4.19 billion in 2030 at a compound annual growth rate (CAGR) of 7.8%. The growth in the forecast period can be attributed to rising frequency of influenza epidemics, growing demand for accurate molecular diagnostics, expansion of point-of-care testing facilities, increased public health preparedness initiatives, technological advancements in rapid diagnostic assays. Major trends in the forecast period include rising demand for rapid influenza testing, increased adoption of molecular diagnostic methods, growing preference for point-of-care flu diagnostics, expansion of seasonal and pandemic surveillance testing, continuous improvement in test sensitivity and specificity.Global Influenza Diagnostic Market Segmentation
1) By Traditional Diagnostic Tests: Rapid Influenza Diagnostic Tests (RIDT), Viral Culture, DFA, Serological Assays, Other Traditional Diagnostic Tests 2) By Molecular Diagnostic Tests: Reverse Transcription Polymerase Chain Reaction (RT-PCR), Isothermal Nucleic Acid Amplification Tests (INAAT), Loop Mediated Isothermal Based Amplification Assays, Transcription Mediated Isothermal Based Amplification Assays, Other Isothermal Nucleic Acid Amplification Tests, Other Molecular Diagnostic Tests 3) By End User: Hospitals And Clinical Laboratories, Diagnostic Reference Laboratories, Academic/Research Institutes, Other End Users Subsegments: 1) By Rapid Influenza Diagnostic Tests (RIDT): Immunoassay-based Tests, Lateral Flow Assays, Enzyme Immunoassays (EIA) 2) By Viral Culture: Cell Culture, Egg-based Culture 3) By Serological Assays: Hemagglutination Inhibition (HI) Assay, Enzyme-Linked Immunosorbent Assay (ELISA), Western Blotting Assay 4) By Other Traditional Diagnostic Tests: Polymerase Chain Reaction (PCR), Immunofluorescence AssaysWhat Is The Driver Of The Influenza Diagnostic Market?
The increasing funding for research on influenza diagnostics is expected to propel the growth of the influenza diagnostic market going forward. Funding for research on influenza diagnostics refers to financial resources allocated by government agencies, institutions, and organizations to support scientific investigations aimed at developing, improving, and validating methods and technologies for detecting and identifying influenza viruses. The rising funding for influenza research is driven by lessons learned from the COVID-19 pandemic and concerns about future respiratory disease outbreaks, as health authorities recognize the critical importance of rapid and accurate diagnostic capabilities to prevent widespread transmission and enable timely public health interventions. Growing research funding supports the development and commercialization of advanced testing technologies, molecular assays, point-of-care devices, and improved laboratory procedures that enable faster and more accurate identification of influenza strains in clinical settings. For instance, in October 2024, according to the Department of Health and Aged Care, a Australia-based government organization responsible for health research, funding, promotion and regulation, the Australian Government is allocating $95 million to combat avian influenza, protecting agriculture, wildlife, and the economy. Therefore, increasing funding for research on influenza diagnostics is driving the growth of the influenza diagnostic industry.Key Players In The Global Influenza Diagnostic Market
Major companies operating in the influenza diagnostic market are F. Hoffmann-La Roche Ltd., Quidel Corporation, Thermo Fisher Scientific Inc., Abbott Laboratories, Hologic Inc., Becton Dickinson and Company, DiaSorin SpA, Luminex Corporation, Meridian Bioscience Inc., GenMark Diagnostics Inc., SA Scientific Ltd., Sekisui Diagnostics LLC, bioMérieux Inc., Danaher Corporation, Siemens Healthineers AG, Coris Bioconcept Sprl, Mast Group, Elitech Technology lnc., Analytik Jena AG, altona Diagnostics GmbH, Tecan Trading AG, Genome Diagnostics Pvt Ltd., Germaine Laboratories Inc., Response Biomedical Corporation, TAUNS Laboratories Inc., TRUPCR Europe Ltd., Quest Diagnostics Inc., Labcorp Employer Services Inc.Global Influenza Diagnostic Market Trends and Insights
Major companies operating in the influenza diagnostic market are developing innovative products such as Lucira COVID-19 & Flu Home Test for the simultaneous rapid qualitative detection. The Lucira COVID-19 and Flu Home Test is a single-use at-home test kit that provides results from self-collected nasal swab samples in roughly 30 minutes. For instance, in February 2023, Pfizer Inc., a US-based pharmaceutical company received approval for the Lucira COVID-19 & Flu Home Test. The test can detect influenza A and B, commonly known as the flu, and SARS-CoV-2, the virus that causes COVID-19.What Are Latest Mergers And Acquisitions In The Influenza Diagnostic Market?
In February 2024, Thyrocare Technologies Limited, a India-based healthcare company, acquired Think Health Diagnostic Private Limited for an undisclosed amount. This acquisition enhances Thyrocare’s service offerings, allowing them to reach more customers and improve market share in the diagnostic sector. Additionally, by integrating at-home ECG testing with blood diagnostics, thereby positioning itself as a comprehensive solution for premedical and annual health checkups. Think Health Diagnostic Private Limited is a India-based home health care services company specializes in providing serves such as telehealth, laboratory testing, and pharmacy.Regional Outlook
North America was the largest region in the influenza diagnostics market in 2025. Western Europe was the second largest region in the influenza diagnostics market. The regions covered in this market report are Asia-Pacific, South East Asia, Western Europe, Eastern Europe, North America, South America, Middle East, Africa. The countries covered in this market report are Australia, Brazil, China, France, Germany, India, Indonesia, Japan, Taiwan, Russia, South Korea, UK, USA, Canada, Italy, Spain.What Defines the Influenza Diagnostic Market?
The influenza diagnostic market includes revenue earned by test kit and reagents, and other products. The market value includes the value of related goods sold by the service provider or included within the service offering. Only goods and services traded between entities or sold to end consumers are included.How is Market Value Defined and Measured?
The market value is defined as the revenues that enterprises gain from the sale of goods and/or services within the specified market and geography through sales, grants, or donations in terms of the currency (in USD unless otherwise specified). The revenues for a specified geography are consumption values that are revenues generated by organizations in the specified geography within the market, irrespective of where they are produced. It does not include revenues from resales along the supply chain, either further along the supply chain or as part of other products.What Key Data and Analysis Are Included in the Influenza Diagnostic Market Report 2026?
The influenza diagnostic market research report is one of a series of new reports from The Business Research Company that provides market statistics, including industry global market size, regional shares, competitors with the market share, detailed market segments, market trends and opportunities, and any further data you may need to thrive in the influenza diagnostic industry. The market research report delivers a complete perspective of everything you need, with an in-depth analysis of the current and future state of the industry.Influenza Diagnostic Market Report Forecast Analysis
| Report Attribute | Details |
|---|---|
| Market Size Value In 2026 | $3.1 billion |
| Revenue Forecast In 2035 | $4.19 billion |
| Growth Rate | CAGR of 8.3% from 2026 to 2035 |
| Base Year For Estimation | 2025 |
| Actual Estimates/Historical Data | 2020-2025 |
| Forecast Period | 2026 - 2030 - 2035 |
| Market Representation | Revenue in USD Billion and CAGR from 2026 to 2035 |
| Segments Covered | Traditional Diagnostic Tests, Molecular Diagnostic Tests, End User |
| Regional Scope | Asia-Pacific, Western Europe, Eastern Europe, North America, South America, Middle East, Africa |
| Country Scope | The countries covered in the report are Australia, Brazil, China, France, Germany, India, ... |
| Key Companies Profiled | F. Hoffmann-La Roche Ltd., Quidel Corporation, Thermo Fisher Scientific Inc., Abbott Laboratories, Hologic Inc., Becton Dickinson and Company, DiaSorin SpA, Luminex Corporation, Meridian Bioscience Inc., GenMark Diagnostics Inc., SA Scientific Ltd., Sekisui Diagnostics LLC, bioMérieux Inc., Danaher Corporation, Siemens Healthineers AG, Coris Bioconcept Sprl, Mast Group, Elitech Technology lnc., Analytik Jena AG, altona Diagnostics GmbH, Tecan Trading AG, Genome Diagnostics Pvt Ltd., Germaine Laboratories Inc., Response Biomedical Corporation, TAUNS Laboratories Inc., TRUPCR Europe Ltd., Quest Diagnostics Inc., Labcorp Employer Services Inc. |
| Customization Scope | Request for Customization |
| Pricing And Purchase Options | Explore Purchase Options |
