
Interatrial Shunt Devices Market Report 2026
Global Outlook – By Device Type (Implantable Shunt Devices, Temporary Shunt Devices), By Delivery Method (Surgical Delivery, Transcatheter Delivery), By Material (Nitinol, Polyester, Polyproplene), By Application (Heart Failure With Preserved Ejection Fraction HFpEF, Heart Failure, Left Atrial Pressure Reduction), By End-User (Hospitals, Ambulatory Surgical Centers, Cardiac Catheterization Laboratories, Other Users) – Market Size, Trends, Strategies, and Forecast to 2035
Interatrial Shunt Devices Market Overview
• Interatrial Shunt Devices market size has reached to $2.9 billion in 2025 • Expected to grow to $4.71 billion in 2030 at a compound annual growth rate (CAGR) of 10.1% • Growth Driver: Rising Prevalence Of Heart Failure Fueling The Growth Of The Market Due To Increasing Demand For Advanced Cardiac Interventions • Market Trend: Innovative Clinical Advancements In First-In-Human Trials Drive Next-Gen Interatrial Shunt Development For Heart Failure Management • North America was the largest region in 2025 and Asia-Pacific is the fastest growing region.What Is Covered Under Interatrial Shunt Devices Market?
Interatrial shunt devices are implantable medical devices designed to create a controlled opening between the left and right atria of the heart to reduce pressure and improve symptoms in patients with heart failure. Interatrial shunt devices are utilized to divert blood from the left atrium to the right atrium, thereby reducing elevated left atrial pressure in patients with heart failure. The main device types of interatrial shunt devices are implantable shunt devices and temporary shunt devices. Implantable shunt devices are medical implants designed to establish a controlled channel between body compartments, commonly used to divert fluid or pressure for therapeutic benefit. These devices can be delivered through surgical procedures and transcatheter delivery. The various materials used in their construction include nitinol, polyester, and polypropylene. They are mainly applied in the management of congenital heart disease, heart failure, and stroke prevention and are utilized across end users, including hospitals, ambulatory surgical centers, cardiac catheterization laboratories, and others.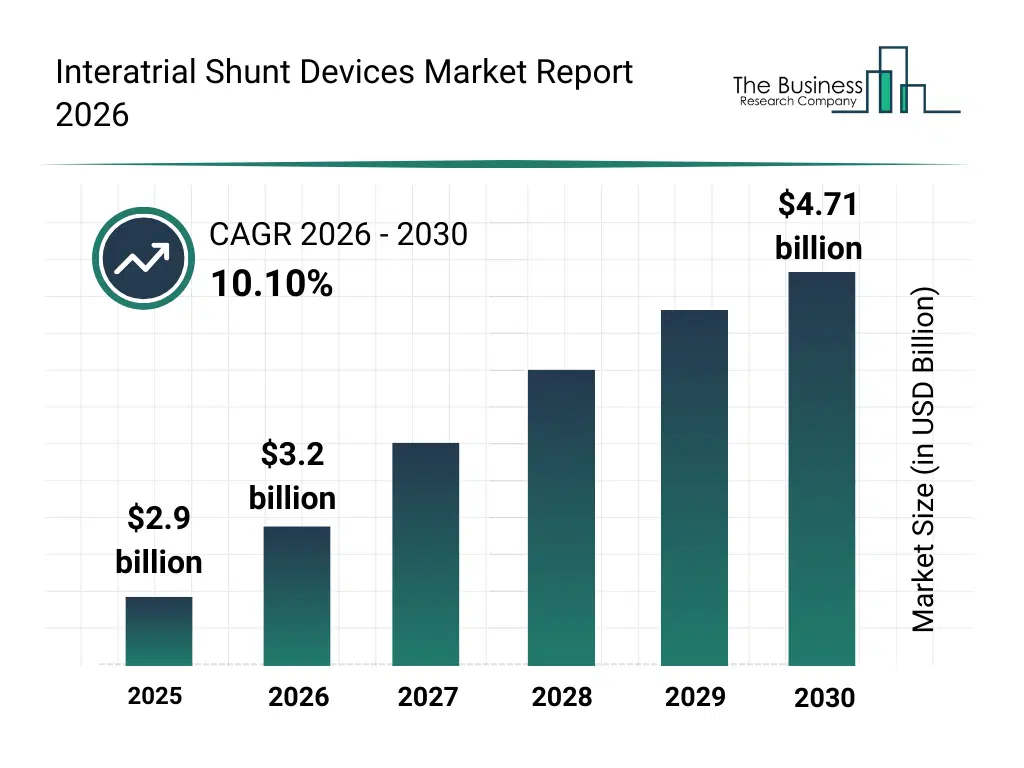
What Is The Interatrial Shunt Devices Market Size and Share 2026?
The interatrial shunt devices market size has grown rapidly in recent years. It will grow from $2.9 billion in 2025 to $3.2 billion in 2026 at a compound annual growth rate (CAGR) of 10.3%. The growth in the historic period can be attributed to rising prevalence of heart failure, growing awareness of interatrial shunt therapies, advancements in cardiac imaging, increasing cardiac catheterization procedures, favorable reimbursement policies.What Is The Interatrial Shunt Devices Market Growth Forecast?
The interatrial shunt devices market size is expected to see rapid growth in the next few years. It will grow to $4.71 billion in 2030 at a compound annual growth rate (CAGR) of 10.1%. The growth in the forecast period can be attributed to technological innovation in shunt devices, expansion of transcatheter delivery methods, aging population with cardiovascular diseases, rising adoption of minimally invasive procedures, increased focus on personalized heart failure management. Major trends in the forecast period include minimally invasive cardiac procedures, personalized heart failure management, device miniaturization, expansion of outpatient cardiac care, advanced hemodynamic monitoring.Global Interatrial Shunt Devices Market Segmentation
1) By Device Type: Implantable Shunt Devices, Temporary Shunt Devices 2) By Delivery Method: Surgical Delivery, Transcatheter Delivery 3) By Material: Nitinol, Polyester, Polyproplene 4) By Application: Heart Failure With Preserved Ejection Fraction HFpEF, Heart Failure, Left Atrial Pressure Reduction 5) By End-User: Hospitals, Ambulatory Surgical Centers, Cardiac Catheterization Laboratories, Other Users Subsegments: 1) By Implantable Shunt Devices: Single-Lumen Shunt Devices, Expandable Shunt Devices, Multi-Lumen Shunt Devices 2) By Temporary Shunt Devices: Balloon-Assisted Shunt Devices, Percutaneous Temporary Shunt Devices, Catheter-Based Shunt DevicesWhat Is The Driver Of The Interatrial Shunt Devices Market?
The rising prevalence of heart failure is expected to propel the growth of the interatrial shunt devices market going forward. Heart failure refers to the heart’s inability to circulate blood effectively, resulting to inadequate oxygen and nutrient delivery to tissues. Heart failure is increasing due to the growing prevalence of hypertension, gradually weakening the heart's ability to pump effectively. Interatrial shunt devices help in heart failure by creating a small opening between the left and right atria, allowing excess pressure from the overloaded left side of the heart to be offloaded to the right side, thereby reducing pulmonary congestion and improving symptoms. For instance, in September 2024, according to the Heart Failure Society of America, a US-based professional organization, reported that as of 2024, about 6.7 million Americans aged 20 and above live with heart failure. This number is projected to rise to 11.4 million by 2050. Therefore, the rising prevalence of heart failure is driving the growth of the interatrial shunt devices industry.Key Players In The Global Interatrial Shunt Devices Market
Major companies operating in the interatrial shunt devices market are Abbott Laboratories, Medtronic plc, Terumo Corporation, Edwards Lifesciences Corporation, Cook Medical, Lepu Medical Technology Beijing Co. Ltd., W. L. Gore & Associates Inc., Venus Medtech Inc., Occlutech Holding AG, Wuhan Vickor Medical Technology Co. Ltd., Corvia Medical Inc., Xeltis BV, Alleviant Medical Inc., Adona Medical Inc., Carag AG, InterShunt Technologies Inc., V-Wave Ltd., MicroPort CardioFlow Group Corp., Medinol Ltd., Lifetech Scientific Corp.Global Interatrial Shunt Devices Market Trends and Insights
Major companies operating in the interatrial shunt devices market are focusing on developing innovative solutions, such as the first-in-human (fih) clinical trial to evaluate the safety and efficacy of novel shunt technologies for reducing left atrial pressure in heart failure patients. First-in-human (FIH) clinical trial refers to the initial stage of testing a new medical device or drug in humans, aiming to assess its safety, tolerability, and preliminary efficacy after successful preclinical studies. For instance, in January 2025, Adona Medical Inc., a US-based medical technology company, successfully completed enrollment in its first-in-human (FIH) clinical trial, ATHENS-HF, for a novel interatrial shunt designed to treat patients with heart failure. This device is among the first to combine therapeutic shunting with integrated bi-atrial pressure monitoring, aiming to deliver a more adaptive and personalized approach to heart failure management.What Are Latest Mergers And Acquisitions In The Interatrial Shunt Devices Market?
In October 2024, Johnson & Johnson, a US-based pharmaceutical industry company, acquired V-Wave Ltd. for an undisclosed amount. With this acquisition, Johnson & Johnson aims to enhance its position in cardiovascular care by broadening its portfolio with advanced heart failure therapies and targeting critical unmet medical needs in the sector. V-Wave Ltd. is an Israel-based medical device company that develops innovative interatrial shunt devices.Regional Outlook
North America was the largest region in the interatrial shunt devices market in 2025. Asia-Pacific is expected to be the fastest-growing region in the forecast period. The regions covered in this market report are Asia-Pacific, South East Asia, Western Europe, Eastern Europe, North America, South America, Middle East, Africa. The countries covered in this market report are Australia, Brazil, China, France, Germany, India, Indonesia, Japan, Taiwan, Russia, South Korea, UK, USA, Canada, Italy, SpainWhat Defines the Interatrial Shunt Devices Market?
The interatrial shunt devices market consists of sales of catheter-based delivery tools, transseptal access kits, monitoring accessories, and procedure-specific consumables. Values in this market are ‘factory gate’ values, that is the value of goods sold by the manufacturers or creators of the goods, whether to other entities (including downstream manufacturers, wholesalers, distributors and retailers) or directly to end customers. The value of goods in this market includes related services sold by the creators of the goods.How is Market Value Defined and Measured?
The market value is defined as the revenues that enterprises gain from the sale of goods and/or services within the specified market and geography through sales, grants, or donations in terms of the currency (in USD unless otherwise specified). The revenues for a specified geography are consumption values that are revenues generated by organizations in the specified geography within the market, irrespective of where they are produced. It does not include revenues from resales along the supply chain, either further along the supply chain or as part of other products.Interatrial Shunt Devices Market Report Forecast Analysis
| Report Attribute | Details |
|---|---|
| Market Size Value In 2026 | $3.2 billion |
| Revenue Forecast In 2035 | $4.71 billion |
| Growth Rate | CAGR of 10.3% from 2026 to 2035 |
| Base Year For Estimation | 2025 |
| Actual Estimates/Historical Data | 2020-2025 |
| Forecast Period | 2026 - 2030 - 2035 |
| Market Representation | Revenue in USD Billion and CAGR from 2026 to 2035 |
| Segments Covered | Device Type, Delivery Method, Material, Application, End-User |
| Regional Scope | Asia-Pacific, Western Europe, Eastern Europe, North America, South America, Middle East, Africa |
| Country Scope | The countries covered in the report are Australia, Brazil, China, France, Germany, India, ... |
| Key Companies Profiled | Abbott Laboratories, Medtronic plc, Terumo Corporation, Edwards Lifesciences Corporation, Cook Medical, Lepu Medical Technology Beijing Co. Ltd., W. L. Gore & Associates Inc., Venus Medtech Inc., Occlutech Holding AG, Wuhan Vickor Medical Technology Co. Ltd., Corvia Medical Inc., Xeltis BV, Alleviant Medical Inc., Adona Medical Inc., Carag AG, InterShunt Technologies Inc., V-Wave Ltd., MicroPort CardioFlow Group Corp., Medinol Ltd., Lifetech Scientific Corp. |
| Customization Scope | Request for Customization |
| Pricing And Purchase Options | Explore Purchase Options |
