
Intracranial Hemorrhage Devices Market Report 2026
Global Outlook – By Product Type (Diagnosis, Treatment), By Devices (Invasive, Non-Invasive), By End Users (Hospitals, Ambulatory Surgical Centres, Other End Users) – Market Size, Trends, Strategies, and Forecast to 2035
Intracranial Hemorrhage Devices Market Overview
• Intracranial Hemorrhage Devices market size has reached to $1.79 billion in 2025 • Expected to grow to $2.27 billion in 2030 at a compound annual growth rate (CAGR) of 4.9% • Growth Driver: Rising Brain Disorder Cases Driving Market Growth • Market Trend: Technological Advancements In Neuro Access Devices For Enhanced Stroke Care • North America was the largest region in 2025.What Is Covered Under Intracranial Hemorrhage Devices Market?
Intracranial hemorrhage devices refer to medical tools and equipment designed to diagnose, manage, and treat intracranial hemorrhages (ICH), which are bleeding events within the skull. These devices play a crucial role in addressing conditions such as subdural hematoma, epidural hematoma, subarachnoid hemorrhage, and intracerebral hemorrhage. The main product types of intracranial hemorrhage devices are diagnosis and treatment. Intracranial hemorrhage diagnosis refers to the medical process of identifying bleeding within the skull. It includes various devices, such as invasive and non-invasive, catering to various end users, including hospitals, ambulatory surgical centers, and others.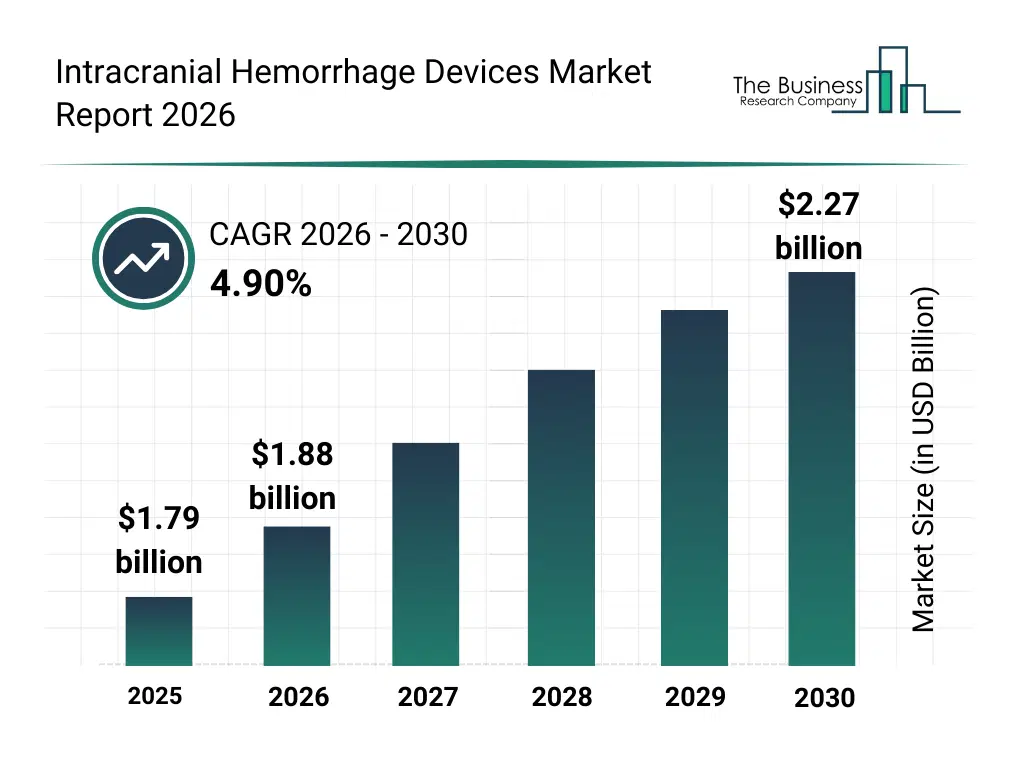
What Is The Intracranial Hemorrhage Devices Market Size and Share 2026?
The intracranial hemorrhage devices market size has grown strongly in recent years. It will grow from $1.79 billion in 2025 to $1.88 billion in 2026 at a compound annual growth rate (CAGR) of 5.1%. The growth in the historic period can be attributed to increasing incidence of traumatic brain injuries, rising prevalence of stroke cases, expansion of neurocritical care units, growth in emergency medical services, availability of advanced diagnostic devices.What Is The Intracranial Hemorrhage Devices Market Growth Forecast?
The intracranial hemorrhage devices market size is expected to see steady growth in the next few years. It will grow to $2.27 billion in 2030 at a compound annual growth rate (CAGR) of 4.9%. The growth in the forecast period can be attributed to increasing investments in neurotechnology, rising demand for real-time brain monitoring, expansion of minimally invasive neurosurgery, growing adoption of AI-assisted imaging analysis, increasing focus on early intervention protocols. Major trends in the forecast period include increasing use of advanced neuroimaging devices, rising adoption of minimally invasive treatment solutions, growing integration of intracranial pressure monitoring, expansion of emergency stroke and trauma care, enhanced focus on rapid diagnosis and intervention.Global Intracranial Hemorrhage Devices Market Segmentation
1) By Product Type: Diagnosis, Treatment 2) By Devices: Invasive, Non-Invasive 3) By End Users: Hospitals, Ambulatory Surgical Centres, Other End Users Subsegments: 1) By Diagnosis: MRI (Magnetic Resonance Imaging) Devices, CT (Computed Tomography) Scanners, Ultrasound Devices, EEG (Electroencephalogram) Devices, Intracranial Pressure Monitors, Blood Gas Analyzers 2) By Treatment: Surgical Instruments (Craniotomy Instruments), Drainage Devices (Ventriculostomy Catheters), Embolization Devices, Hemostatic Agents, Coagulation Devices, Neurostimulation DevicesWhat Is The Driver Of The Intracranial Hemorrhage Devices Market?
The rising cases of brain disorders are expected to propel the growth of the intracranial hemorrhage devices market going forward. Brain disorders refer to a wide range of conditions that affect the structure, function, or chemistry of the brain, leading to disruptions in normal brain processes. The cases of brain disorders are increasing due to factors such as improved diagnostics, aging populations, lifestyle changes, and greater awareness, along with rising stress. Intracranial hemorrhage devices are used to detect, monitor, and treat brain disorders involving bleeding by identifying hemorrhages, managing pressure, and removing blood clots to prevent further brain damage. For instance, in October 2025, according to the World Health Organization, a Switzerland-based Intergovernmental organization reported that The WHO warns that fewer than one in three countries have a national policy to tackle neurological disorders, which cause over 11 million deaths annually. Additionally, its new report shows these conditions affect over 3 billion people,more than 40% of the global population. Therefore, the rising cases of brain disorders drive the growth of the intracranial hemorrhage devices industry.Key Players In The Global Intracranial Hemorrhage Devices Market
Major companies operating in the intracranial hemorrhage devices market are Johnson & Johnson Services Inc., Medtronic Inc., Siemens Healthcare GmbH, Stryker Corporation, General Electric Company, Terumo Corporation, Canon Medical Systems Corporation, Penumbra Inc., MicroPort Scientific Corporation, Aidoc Medical Ltd., Viz.ai Inc., Phenox GmbH, RAPID AI Inc., Acandis GmbH, Kaneka Medix Corporation, Hyperfine Inc., ClearMind Biomedical Inc., InfraScan Inc., Schindler Endoskopie Technologie GmbH, Sense Neuro Diagnostics Inc., MaxQ AI Inc., Invamed Inc.Global Intracranial Hemorrhage Devices Market Trends and Insights
Major companies operating in the intracranial hemorrhage devices market are focusing on developing innovative products, such as advanced neuro access devices, to enhance the precision and efficiency of diagnosing and treating brain injuries. Advanced neuro access devices refer to specialized medical tools and technologies designed to facilitate precise access to the brain or nervous system for diagnostic, surgical, or therapeutic purposes. For instance, in June 2024, Penumbra Inc., a US-based medical device company, announced the European launch of its advanced neuro access devices, BMX81 and BMX96. These devices, designed for managing ischemic and hemorrhagic strokes, incorporate innovative laser-cut stainless steel hypotube technology to improve stability and trackability, while enabling larger inner diameters. This launch highlights Penumbra's commitment to enhancing stroke care in Europe.What Are Latest Mergers And Acquisitions In The Intracranial Hemorrhage Devices Market?
In September 2024, Stryker Corporation, a US-based medication company, acquired NICO Corporation for an undisclosed amount. Through this acquisition, Stryker Corporation aims to strengthen its commitment to neurotechnology through tumor and stroke care and also expand its portfolio of solutions for tumor resection and the treatment of intracerebral hemorrhage. NICO Corporation is a US-based medical equipment manufacturing company that offers devices specifically designed for the treatment of intracranial hemorrhage (ICH).Regional Outlook
North America was the largest region in the intracranial hemorrhage devices market in 2025. The regions covered in this market report are Asia-Pacific, South East Asia, Western Europe, Eastern Europe, North America, South America, Middle East, Africa. The countries covered in this market report are Australia, Brazil, China, France, Germany, India, Indonesia, Japan, Taiwan, Russia, South Korea, UK, USA, Canada, Italy, SpainWhat Defines the Intracranial Hemorrhage Devices Market?
The intracranial hemorrhage devices market consists of sales of embolization devices, computed tomography scanners, balloon occlusion devices, and neuro-thrombectomy devices. Values in this market are ‘factory gate’ values, that is, the value of goods sold by the manufacturers or creators of the goods, whether to other entities (including downstream manufacturers, wholesalers, distributors, and retailers) or directly to end customers. The value of goods in this market includes related services sold by the creators of the goods.How is Market Value Defined and Measured?
The market value is defined as the revenues that enterprises gain from the sale of goods and/or services within the specified market and geography through sales, grants, or donations in terms of the currency (in USD unless otherwise specified). The revenues for a specified geography are consumption values that are revenues generated by organizations in the specified geography within the market, irrespective of where they are produced. It does not include revenues from resales along the supply chain, either further along the supply chain or as part of other products.Intracranial Hemorrhage Devices Market Report Forecast Analysis
| Report Attribute | Details |
|---|---|
| Market Size Value In 2026 | $1.88 billion |
| Revenue Forecast In 2035 | $2.27 billion |
| Growth Rate | CAGR of 5.1% from 2026 to 2035 |
| Base Year For Estimation | 2025 |
| Actual Estimates/Historical Data | 2020-2025 |
| Forecast Period | 2026 - 2030 - 2035 |
| Market Representation | Revenue in USD Billion and CAGR from 2026 to 2035 |
| Segments Covered | Product Type, Devices, End Users |
| Regional Scope | Asia-Pacific, Western Europe, Eastern Europe, North America, South America, Middle East, Africa |
| Country Scope | The countries covered in the report are Australia, Brazil, China, France, Germany, India, ... |
| Key Companies Profiled | Johnson & Johnson Services Inc., Medtronic Inc., Siemens Healthcare GmbH, Stryker Corporation, General Electric Company, Terumo Corporation, Canon Medical Systems Corporation, Penumbra Inc., MicroPort Scientific Corporation, Aidoc Medical Ltd., Viz.ai Inc., Phenox GmbH, RAPID AI Inc., Acandis GmbH, Kaneka Medix Corporation, Hyperfine Inc., ClearMind Biomedical Inc., InfraScan Inc., Schindler Endoskopie Technologie GmbH, Sense Neuro Diagnostics Inc., MaxQ AI Inc., Invamed Inc. |
| Customization Scope | Request for Customization |
| Pricing And Purchase Options | Explore Purchase Options |
