
Intravenous Fluid Transfer Drug Devices Market Report 2026
Global Outlook – By Type (Infusion Bags, Infusion Devices, Other Types), By Application (Autoimmune Diseases, Blood Disorders, Cardiovascular Disorders, Neurology, Oncology, Other Applications), By End-Use Sector (Hospitals, Specialized Clinics, Other End-Use Sectors) – Market Size, Trends, Strategies, and Forecast to 2035
Intravenous Fluid Transfer Drug Devices Market Overview
• Intravenous Fluid Transfer Drug Devices market size has reached to $7.85 billion in 2025 • Expected to grow to $10.55 billion in 2030 at a compound annual growth rate (CAGR) of 6% • Growth Driver: Rising Hospitalization Rates Driving Growth In Intravenous Fluid Transfer Drug Device Market • Market Trend: Advancements In Intravenous Fluid Transfer Devices By Enhancing Safety And Efficiency • North America was the largest region in 2025 and Asia-Pacific is the fastest growing region.What Is Covered Under Intravenous Fluid Transfer Drug Devices Market?
Intravenous fluid transfer drug devices are tools used to deliver fluids and medications directly into a patient’s bloodstream via an intravenous line. The device ensures efficient, controlled, and immediate administration of treatments, which is crucial for hydration, medication delivery, and critical care. These devices are designed to prevent contamination of both the medication and the IV fluid, maintaining sterility and reducing the risk of infection. The main types in the intravenous fluid transfer drug devices market are infusion bags, infusion devices, and others. Infusion bags refer to flexible, sterile containers used to hold and administer fluids, medications, or nutrients directly into a patient's bloodstream through an intravenous (IV) line. The various applications include autoimmune diseases, blood disorders, cardiovascular disorders, neurology, oncology, and others, and several end-use sectors includes hospitals, specialized clinics, and others.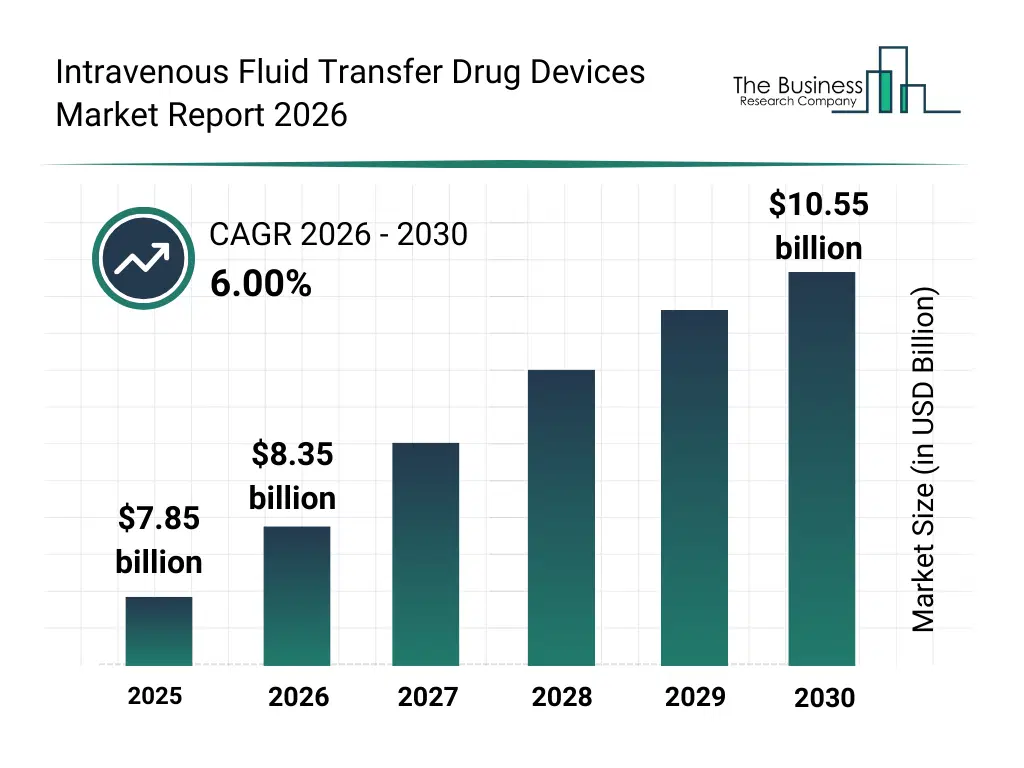
What Is The Intravenous Fluid Transfer Drug Devices Market Size and Share 2026?
The intravenous fluid transfer drug devices market size has grown strongly in recent years. It will grow from $7.85 billion in 2025 to $8.35 billion in 2026 at a compound annual growth rate (CAGR) of 6.3%. The growth in the historic period can be attributed to reliance on conventional iv sets, increasing hospital and clinical procedures, limited availability of smart infusion devices, growing awareness of iv therapy benefits, adoption of pvc infusion bags.What Is The Intravenous Fluid Transfer Drug Devices Market Growth Forecast?
The intravenous fluid transfer drug devices market size is expected to see strong growth in the next few years. It will grow to $10.55 billion in 2030 at a compound annual growth rate (CAGR) of 6.0%. The growth in the forecast period can be attributed to technological advancements in smart infusion pumps, rising demand for pre-filled and multi-chamber infusion bags, increasing applications in oncology and neurology, growth in home healthcare and ambulatory infusion, stricter regulatory and quality standards. Major trends in the forecast period include rising adoption of smart infusion devices and pumps, increasing use of pre-filled and multi-chamber infusion bags, growing demand for needleless connectors and safety iv sets, expansion of iv therapy across chronic and critical care treatments, enhanced focus on sterility, accuracy, and flow control.Global Intravenous Fluid Transfer Drug Devices Market Segmentation
1) By Type: Infusion Bags, Infusion Devices, Other Types 2) By Application: Autoimmune Diseases, Blood Disorders, Cardiovascular Disorders, Neurology, Oncology, Other Applications 3) By End-Use Sector: Hospitals, Specialized Clinics, Other End-Use Sectors Subsegments: 1) By Infusion Bags: PVC Infusion Bags, Non-PVC Infusion Bags, Pre-Filled Infusion Bags, Multi-Chamber Infusion Bags, IV Fluid Administration Bags 2) By Infusion Devices: Infusion Pumps, Syringe Pumps, Ambulatory Infusion Pumps, Gravity Infusion Devices, Smart Infusion Devices 3) By Other Types: IV Sets And Accessories, IV Catheters, Needleless Connectors, Flow Regulators, Pressure Infusion DevicesWhat Is The Driver Of The Intravenous Fluid Transfer Drug Devices Market?
The rising hospitalization rate is expected to propel the growth of the intravenous fluid transfer drug device market going forward. The rising hospitalization rate is driven by an aging population, chronic illnesses, advanced diagnostics, and increased healthcare access, alongside lifestyle-related diseases and medical technology advancements. Fluid transfer systems ensure precise and controlled administration of fluids and medications directly into the bloodstream in hospitalized patients. For instance, in March 2023, according to GOV.UK, a US-based government agency, in 2023, COVID-19 hospital admissions in England rose slightly to 10.62 per 100,000, while ICU/HDU admissions stayed low at 0.28 per 100,000. Most regions saw small increases, with rates largely stable across age groups. Therefore, a rising hospitalization rate will drive intravenous fluid transfer drug device industry growth.Key Players In The Global Intravenous Fluid Transfer Drug Devices Market
Major companies operating in the intravenous fluid transfer drug devices market are Cardinal Health Inc., Medtronic plc, Becton Dickinson and Company, Baxter International Inc., B Braun Holding GmbH Co KG, Fresenius Kabi AG, Nipro Corporation, West Pharmaceutical Services Inc., ICU Medical Inc., Merit Medical Systems Inc., Vygon SAS, AngioDynamics Inc., Elcam Medical Inc., InfuSystem Holdings Inc., CODAN US Corporation, Arcomed AG, Terumo Corporation, Smiths Medical, IRadimed Corporation, Micrel Medical Devices SA, Boston Scientific Corporation, AdvaCare Pharma, Eitan Medical, Ypsomed Holding AGGlobal Intravenous Fluid Transfer Drug Devices Market Trends and Insights
Major companies in the intravenous fluid transfer drug device market are developing modular and comprehensive infusion systems with features such as auto-identification to enhance accuracy, improve patient safety, and streamline the administration of fluids and medications. Auto-identification in infusion systems automatically verifies and matches medications to the correct patient using barcode scanning or RFID technology. For instance, in July 2023, Becton, Dickinson, and Company, a US-based medical device company, received FDA 510(k) clearance for its updated BD Alaris Infusion System. The BD Alaris Infusion System has updated hardware for its Point-of-Care Unit (PCU), including large volume pumps, syringe pumps, patient-controlled analgesia (PCA) pumps, respiratory monitoring, and auto-identification modules. It also introduces an advanced software version with improved cybersecurity and interoperability features that allow for seamless integration with widely used electronic medical record (EMR) systems, supporting smart and connected healthcare.What Are Latest Mergers And Acquisitions In The Intravenous Fluid Transfer Drug Devices Market?
In May 2024, Nordson Corporation, a US-based precision technology company, acquired Atrion Corporation for an undisclosed amount. With this acquisition, Nordson Corporation’s aim is to expand and strengthen its medical portfolio by integrating Atrion's advanced medical infusion and cardiovascular technologies. Atrion Corporation is a US-based technology company that provides components and devices for intravenous fluid transfer and drug delivery.Regional Outlook
North America was the largest region in the intravenous fluid transfer drug devices market in 2025. Asia-Pacific is expected to be the fastest-growing region in the forecast period. The regions covered in this market report are Asia-Pacific, South East Asia, Western Europe, Eastern Europe, North America, South America, Middle East, Africa. The countries covered in this market report are Australia, Brazil, China, France, Germany, India, Indonesia, Japan, Taiwan, Russia, South Korea, UK, USA, Canada, Italy, SpainWhat Defines the Intravenous Fluid Transfer Drug Devices Market?
The intravenous fluid transfer drug devices market consists of sales of infusion pumps, IV bags, tubing sets, connectors, catheters, needles, flow regulators, and syringes. Values in this market are ‘factory gate’ values, that is the value of goods sold by the manufacturers or creators of the goods, whether to other entities (including downstream manufacturers, wholesalers, distributors and retailers) or directly to end customers. The value of goods in this market includes related services sold by the creators of the goods.How is Market Value Defined and Measured?
The market value is defined as the revenues that enterprises gain from the sale of goods and/or services within the specified market and geography through sales, grants, or donations in terms of the currency (in USD unless otherwise specified). The revenues for a specified geography are consumption values that are revenues generated by organizations in the specified geography within the market, irrespective of where they are produced. It does not include revenues from resales along the supply chain, either further along the supply chain or as part of other products.Intravenous Fluid Transfer Drug Devices Market Report Forecast Analysis
| Report Attribute | Details |
|---|---|
| Market Size Value In 2026 | $8.35 billion |
| Revenue Forecast In 2035 | $10.55 billion |
| Growth Rate | CAGR of 6.3% from 2026 to 2035 |
| Base Year For Estimation | 2025 |
| Actual Estimates/Historical Data | 2020-2025 |
| Forecast Period | 2026 - 2030 - 2035 |
| Market Representation | Revenue in USD Billion and CAGR from 2026 to 2035 |
| Segments Covered | Type, Application, End-Use Sector |
| Regional Scope | Asia-Pacific, Western Europe, Eastern Europe, North America, South America, Middle East, Africa |
| Country Scope | The countries covered in the report are Australia, Brazil, China, France, Germany, India, ... |
| Key Companies Profiled | Cardinal Health Inc., Medtronic plc, Becton Dickinson and Company, Baxter International Inc., B Braun Holding GmbH Co KG, Fresenius Kabi AG, Nipro Corporation, West Pharmaceutical Services Inc., ICU Medical Inc., Merit Medical Systems Inc., Vygon SAS, AngioDynamics Inc., Elcam Medical Inc., InfuSystem Holdings Inc., CODAN US Corporation, Arcomed AG, Terumo Corporation, Smiths Medical, IRadimed Corporation, Micrel Medical Devices SA, Boston Scientific Corporation, AdvaCare Pharma, Eitan Medical, Ypsomed Holding AG |
| Customization Scope | Request for Customization |
| Pricing And Purchase Options | Explore Purchase Options |
