Intravenous Therapy And Vein Access Devices Market Report 2026
Intravenous Therapy And Vein Access Devices Market Report 2026
Global Outlook – By Type (Intravenous (IV) Therapy And Vein Access Implantable Ports, Intravenous (IV) Therapy And Vein Access Intravenous Catheters, Hypodermic Intravenous (IV) Therapy And Vein Access Needles, Intravenous (IV) Therapy And Vein Access Infusion Pumps, Other Types), By Application (Volume Expanders, Medication Administration, Blood-Based Products, Nutrition And Buffer Solutions), By End Use (Hospitals, Clinics, Ambulatory Surgical Centers) – Market Size, Trends, Strategies, and Forecast to 2035
Intravenous Therapy And Vein Access Devices Market Overview
• Intravenous Therapy And Vein Access Devices market size has reached to $33.15 billion in 2025 • Expected to grow to $48.1 billion in 2030 at a compound annual growth rate (CAGR) of 7.7% • Growth Driver: Rising Surgical Procedures Driving Growth in Intravenous Therapy and Vein Access Devices Market • Market Trend: Advancements in Short Peripheral Intravenous Catheters • North America was the largest region in 2025.What Is Covered Under Intravenous Therapy And Vein Access Devices Market?
Intravenous therapy and vein access devices refer to the administration of fluids, medications, or nutrients directly into the bloodstream using tools such as catheters and needles for accessing veins. These devices are essential in medical treatments for hydration, medication delivery, blood transfusions, and nutrition, enabling precise and continuous care in various healthcare settings. The main types in the intravenous therapy and vein access devices market are intravenous (IV) therapy and vein access implantable ports, intravenous (IV) therapy and vein access intravenous catheters, hypodermic intravenous (IV) therapy and vein access needles, intravenous (iv) therapy and vein access infusion pumps, and others. Intravenous (IV) therapy and vein access implantable ports are small devices implanted under the skin that provide a durable and easily accessible site for repeated IV access, facilitating long-term treatments and minimizing discomfort from frequent needle insertions. The various applications include volume expanders, medication administration, blood-based products, and nutrition and buffer solutions, several end users including hospitals, clinics, and ambulatory surgical centers.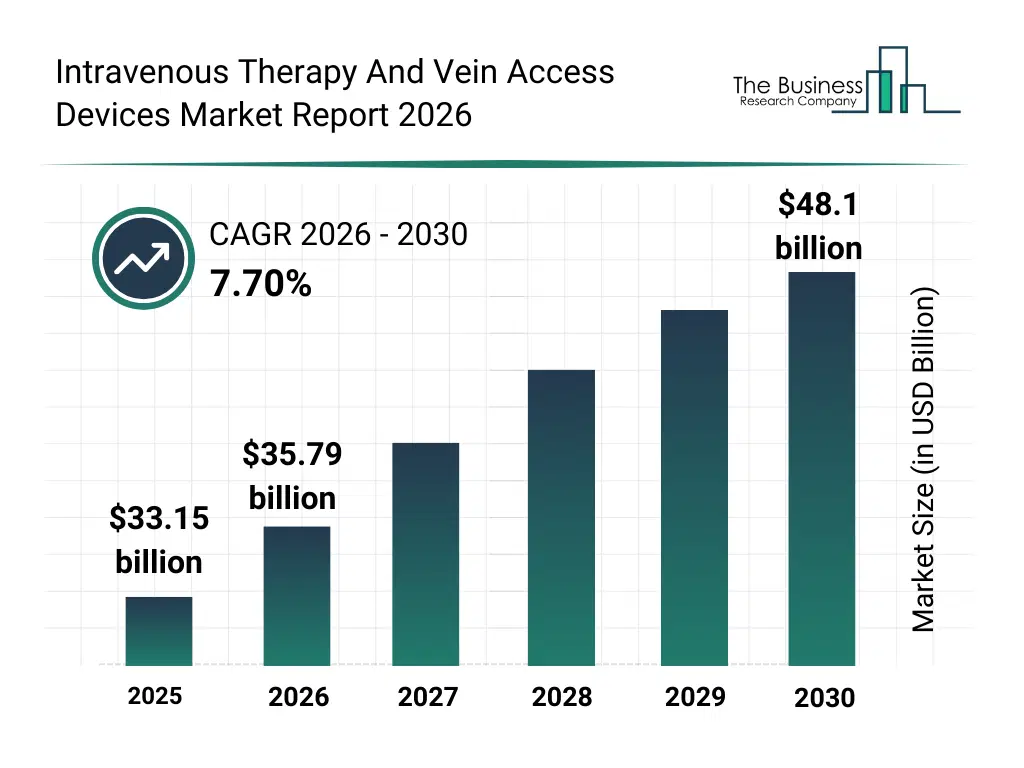
What Is The Intravenous Therapy And Vein Access Devices Market Size and Share 2026?
The intravenous therapy and vein access devices market size has grown strongly in recent years. It will grow from $33.15 billion in 2025 to $35.79 billion in 2026 at a compound annual growth rate (CAGR) of 8.0%. The growth in the historic period can be attributed to limited availability of advanced iv therapy devices, reliance on conventional catheters and needles, growing hospital and clinic infrastructure, increasing demand for blood-based products and medication administration, awareness of infection control protocols.What Is The Intravenous Therapy And Vein Access Devices Market Growth Forecast?
The intravenous therapy and vein access devices market size is expected to see strong growth in the next few years. It will grow to $48.1 billion in 2030 at a compound annual growth rate (CAGR) of 7.7%. The growth in the forecast period can be attributed to development of implantable ports and advanced infusion pumps, rising adoption of needleless connectors and iv administration sets, expansion of ambulatory surgical centers, integration of smart monitoring and digital solutions, increasing investment in patient safety and regulatory compliance. Major trends in the forecast period include rising demand for peripheral and central venous access devices, increasing adoption of safety-engineered needles and catheters, growth in infusion pump technologies and patient-controlled analgesia systems, expansion of nutrition and medication administration applications, enhanced focus on sterility, accuracy, and regulatory compliance.Global Intravenous Therapy And Vein Access Devices Market Segmentation
1) By Type: Intravenous (IV) Therapy And Vein Access Implantable Ports, Intravenous (IV) Therapy And Vein Access Intravenous Catheters, Hypodermic Intravenous (IV) Therapy And Vein Access Needles, Intravenous (IV) Therapy And Vein Access Infusion Pumps, Other Types 2) By Application: Volume Expanders, Medication Administration, Blood-Based Products, Nutrition And Buffer Solutions 3) By End Use: Hospitals, Clinics, Ambulatory Surgical Centers Subsegments: 1) By Intravenous (IV) Therapy And Vein Access Implantable Ports: Single-Lumen Implantable Ports, Double-Lumen Implantable Ports, Triple-Lumen Implantable Ports 2) By Intravenous (IV) Therapy And Vein Access Intravenous Catheters: Peripheral IV Catheters, Central Venous Catheters (CVC), Peripherally Inserted Central Catheters (PICC), Tunneled Catheters 3) By Hypodermic Intravenous (IV) Therapy And Vein Access Needles: Standard Hypodermic Needles, Butterfly Needles, Safety Hypodermic Needles 4) By Intravenous (IV) Therapy And Vein Access Infusion Pumps: Volumetric Infusion Pumps, Syringe Infusion Pumps, Patient-Controlled Analgesia (PCA) Pumps, Ambulatory Infusion Pumps 5) By Other Types: IV Administration Sets, IV Fluid Bags And Tubing, Needleless Connectors, IV Drip ChambersWhat Is The Driver Of The Intravenous Therapy And Vein Access Devices Market?
The increasing number of surgical procedures is expected to propel the growth of the intravenous therapy and vein access devices market going forward. The growing demand for elective surgeries, such as cosmetic procedures, plastic surgery, rhinoplasty and other non-essential improving the surgeries market. Intravenous therapy and vein access devices are essential in surgical procedures for administering anesthesia, medications, fluids, and nutrients, ensuring patient stability, and allowing rapid intervention when necessary. For instance, in June 2024, according to the International Society of Aesthetic Plastic Surgery (ISAPS), a US-based nonprofit professional organization, total surgical and non-surgical procedures increased by 3.4% in 2023, reaching 34.9 million. This growth was driven by a 5.5% rise in surgical procedures. Plastic surgeons performed over 15.8 million surgical procedures and 19.1 million non-surgical procedures, marking a 40% overall increase in procedures over the past four years. Therefore, the increasing number of surgical procedures drives the growth of the intravenous therapy and vein access devices industry.Key Players In The Global Intravenous Therapy And Vein Access Devices Market
Major companies operating in the intravenous therapy and vein access devices market are Cardinal Health Inc., Johnson & Johnson Services Inc., 3M Company, Medtronic plc, Becton Dickinson and Company, Baxter International Inc., B Braun Holding GmbH Co KG, Fresenius Kabi AG, Terumo Corporation, West Pharmaceutical Services Inc., ICU Medical Inc., Merit Medical Systems Inc., Avanos Medical Inc., Vygon SAS, AngioDynamics Inc., Elcam Medical Inc., Teleflex Incorporated, Smiths Medical, Nipro Corporation, Argon Medical Devices Inc., Angiplast Private Limited, Poly Medicure Limited, Iradimed CorporationGlobal Intravenous Therapy And Vein Access Devices Market Trends and Insights
Major companies operating in the intravenous therapy and vein access devices market are developing short peripheral intravenous catheters to enhance patient comfort, reduce insertion-related complications, and improve the efficiency of fluid and medication delivery. Short peripheral intravenous catheters provide a quick and efficient means of delivering fluids, medications, and nutrients directly into the bloodstream, minimizing patient discomfort, reducing the risk of infection, and allowing for easy and reliable venous access to various medical treatments. For instance, in October 2023, B. Braun, a Germany-based medical device company, launched the Introcan Safety 2 IV Catheter, a short peripheral intravenous catheter with passive needlestick prevention and a Multi-Access blood control septum. The advanced catheter is designed to enhance clinician safety by providing automatic needlestick protection and a blood control feature that minimizes exposure to bloodborne pathogens every time the hub is accessed.What Are Latest Mergers And Acquisitions In The Intravenous Therapy And Vein Access Devices Market?
In June 2023, Merit Medical Systems, Inc., a US-based healthcare technology company, acquired the dialysis catheter product portfolio and the BioSentry Biopsy Tract Sealant System from AngioDynamics, Inc. for an undisclosed amount. With this acquisition, Merit Medical’s aim was to strategically expand its product portfolio in key markets—specifically dialysis and biopsy. AngioDynamics, Inc., is a US-based medical device company that provides devices for intravenous (IV) therapy and vein access, among other medical product lines.Regional Outlook
North America was the largest region in the intravenous therapy and vein access devices market in 2025. The regions covered in this market report are Asia-Pacific, South East Asia, Western Europe, Eastern Europe, North America, South America, Middle East, Africa. The countries covered in this market report are Australia, Brazil, China, France, Germany, India, Indonesia, Japan, Taiwan, Russia, South Korea, UK, USA, Canada, Italy, SpainWhat Defines the Intravenous Therapy And Vein Access Devices Market?
The intravenous therapy and vein access devices market consists of revenues earned by entities by providing services such as catheter insertion, intravenous administration, device maintenance, sterilization, training for healthcare professionals. The market value includes the value of related goods sold by the service provider or included within the service offering. The intravenous therapy and vein access devices market also includes sales of catheters, infusion pumps, and needleless connectors. Values in this market are ‘factory gate’ values, that is the value of goods sold by the manufacturers or creators of the goods, whether to other entities (including downstream manufacturers, wholesalers, distributors and retailers) or directly to end customers. The value of goods in this market includes related services sold by the creators of the goods.How is Market Value Defined and Measured?
The market value is defined as the revenues that enterprises gain from the sale of goods and/or services within the specified market and geography through sales, grants, or donations in terms of the currency (in USD unless otherwise specified). The revenues for a specified geography are consumption values that are revenues generated by organizations in the specified geography within the market, irrespective of where they are produced. It does not include revenues from resales along the supply chain, either further along the supply chain or as part of other products.Intravenous Therapy And Vein Access Devices Market Report Forecast Analysis
| Report Attribute | Details |
|---|---|
| Market Size Value In 2026 | $35.79 billion |
| Revenue Forecast In 2035 | $48.1 billion |
| Growth Rate | CAGR of 8.0% from 2026 to 2035 |
| Base Year For Estimation | 2025 |
| Actual Estimates/Historical Data | 2020-2025 |
| Forecast Period | 2026 - 2030 - 2035 |
| Market Representation | Revenue in USD Billion and CAGR from 2026 to 2035 |
| Segments Covered | Type, Application, End Use |
| Regional Scope | Asia-Pacific, Western Europe, Eastern Europe, North America, South America, Middle East, Africa |
| Country Scope | The countries covered in the report are Australia, Brazil, China, France, Germany, India, ... |
| Key Companies Profiled | Cardinal Health Inc., Johnson & Johnson Services Inc., 3M Company, Medtronic plc, Becton Dickinson and Company, Baxter International Inc., B Braun Holding GmbH Co KG, Fresenius Kabi AG, Terumo Corporation, West Pharmaceutical Services Inc., ICU Medical Inc., Merit Medical Systems Inc., Avanos Medical Inc., Vygon SAS, AngioDynamics Inc., Elcam Medical Inc., Teleflex Incorporated, Smiths Medical, Nipro Corporation, Argon Medical Devices Inc., Angiplast Private Limited, Poly Medicure Limited, Iradimed Corporation |
| Customization Scope | Request for Customization |
| Pricing And Purchase Options | Explore Purchase Options |
Frequently Asked Questions
The Intravenous Therapy And Vein Access Devices Market Report 2026 market was valued at $33.15 billion in 2025, increased to $35.79 billion in 2026, and is projected to reach $48.1 billion by 2030.
The expected CAGR for the Intravenous Therapy And Vein Access Devices Market Report 2026 market during the forecast period 2025–2030 is 7.7%.
Major growth driver of the market includes: Rising Surgical Procedures Driving Growth in Intravenous Therapy and Vein Access Devices Market in the Intravenous Therapy And Vein Access Devices Market Report 2026 market. For further insights on this market,
The intravenous therapy and vein access devices market covered in this report is segmented –
1) By Type: Intravenous (IV) Therapy And Vein Access Implantable Ports, Intravenous (IV) Therapy And Vein Access Intravenous Catheters, Hypodermic Intravenous (IV) Therapy And Vein Access Needles, Intravenous (IV) Therapy And Vein Access Infusion Pumps, Other Types
2) By Application: Volume Expanders, Medication Administration, Blood-Based Products, Nutrition And Buffer Solutions
3) By End Use: Hospitals, Clinics, Ambulatory Surgical Centers Subsegments:
1) By Intravenous (IV) Therapy And Vein Access Implantable Ports: Single-Lumen Implantable Ports, Double-Lumen Implantable Ports, Triple-Lumen Implantable Ports
2) By Intravenous (IV) Therapy And Vein Access Intravenous Catheters: Peripheral IV Catheters, Central Venous Catheters (CVC), Peripherally Inserted Central Catheters (PICC), Tunneled Catheters
3) By Hypodermic Intravenous (IV) Therapy And Vein Access Needles: Standard Hypodermic Needles, Butterfly Needles, Safety Hypodermic Needles
4) By Intravenous (IV) Therapy And Vein Access Infusion Pumps: Volumetric Infusion Pumps, Syringe Infusion Pumps, Patient-Controlled Analgesia (PCA) Pumps, Ambulatory Infusion Pumps
5) By Other Types: IV Administration Sets, IV Fluid Bags And Tubing, Needleless Connectors, IV Drip Chambers
1) By Type: Intravenous (IV) Therapy And Vein Access Implantable Ports, Intravenous (IV) Therapy And Vein Access Intravenous Catheters, Hypodermic Intravenous (IV) Therapy And Vein Access Needles, Intravenous (IV) Therapy And Vein Access Infusion Pumps, Other Types
2) By Application: Volume Expanders, Medication Administration, Blood-Based Products, Nutrition And Buffer Solutions
3) By End Use: Hospitals, Clinics, Ambulatory Surgical Centers Subsegments:
1) By Intravenous (IV) Therapy And Vein Access Implantable Ports: Single-Lumen Implantable Ports, Double-Lumen Implantable Ports, Triple-Lumen Implantable Ports
2) By Intravenous (IV) Therapy And Vein Access Intravenous Catheters: Peripheral IV Catheters, Central Venous Catheters (CVC), Peripherally Inserted Central Catheters (PICC), Tunneled Catheters
3) By Hypodermic Intravenous (IV) Therapy And Vein Access Needles: Standard Hypodermic Needles, Butterfly Needles, Safety Hypodermic Needles
4) By Intravenous (IV) Therapy And Vein Access Infusion Pumps: Volumetric Infusion Pumps, Syringe Infusion Pumps, Patient-Controlled Analgesia (PCA) Pumps, Ambulatory Infusion Pumps
5) By Other Types: IV Administration Sets, IV Fluid Bags And Tubing, Needleless Connectors, IV Drip Chambers
Major trend in this market includes: Advancements in Short Peripheral Intravenous Catheters For further insights on this market,
Request for SampleMajor companies operating in the Intravenous Therapy And Vein Access Devices Market Report 2026 market are Major companies operating in the intravenous therapy and vein access devices market are Cardinal Health Inc., Johnson & Johnson Services Inc., 3M Company, Medtronic plc, Becton Dickinson and Company, Baxter International Inc., B Braun Holding GmbH Co KG, Fresenius Kabi AG, Terumo Corporation, West Pharmaceutical Services Inc., ICU Medical Inc., Merit Medical Systems Inc., Avanos Medical Inc., Vygon SAS, AngioDynamics Inc., Elcam Medical Inc., Teleflex Incorporated, Smiths Medical, Nipro Corporation, Argon Medical Devices Inc., Angiplast Private Limited, Poly Medicure Limited, Iradimed Corporation
North America was the largest region in the intravenous therapy and vein access devices market in 2025. The regions covered in the intravenous therapy and vein access devices market report are Asia-Pacific, South East Asia, Western Europe, Eastern Europe, North America, South America, Middle East, Africa.
