
Investigational New Drug Contract Development And Manufacturing Organization (CDMO) Market Report 2026
Global Outlook – By Product (Small Molecules, Large Molecules), By Formulation Type (Solid Formulations, Liquid Formulations, Injectable Formulations), By Service Type (Clinical Trial Material Supply, Regulatory Support Services, Manufacturing Services), By End-User (Biotechnology Companies, Academic And Research Institutes, Other End-Users) – Market Size, Trends, Strategies, and Forecast to 2035
Investigational New Drug Contract Development And Manufacturing Organization (CDMO) Market Overview
• Investigational New Drug Contract Development And Manufacturing Organization (CDMO) market size has reached to $5.91 billion in 2025 • Expected to grow to $8.17 billion in 2030 at a compound annual growth rate (CAGR) of 6.6% • Growth Driver: Increase In Clinical Trials Fueling The Growth Of The Market Due To Rising Demand For Innovative Treatments • Market Trend: Sartorius And Repligen Form Strategic Partnership To Advance Upstream Bioprocessing • North America was the largest region in 2025 and Asia-Pacific is the fastest growing region.What Is Covered Under Investigational New Drug Contract Development And Manufacturing Organization (CDMO) Market?
An investigational new drug contract development and manufacturing organization (CDMO) refers to a contract development and manufacturing organization that supports the development and production of pharmaceutical compounds intended for use in clinical trials under an Investigational New Drug application. These organizations operate within the regulatory frameworks required for early-stage drug development, ensuring compliance with guidelines necessary for advancing experimental therapies toward market approval. The main products of investigational new drug contract development and manufacturing organization (CDMO) include small molecules and large molecules. Small molecules refer to low molecular weight compounds that can easily penetrate cell membranes and interact with intracellular targets to influence biological processes. It is used for various formulations, such as solid formulations, liquid formulations, and injectable formulations, and offers different services, including preclinical services, clinical trial material supply, regulatory support services, and manufacturing services. It is used by several end-users, including pharmaceutical companies, biotechnology companies, academic and research institutes, and others.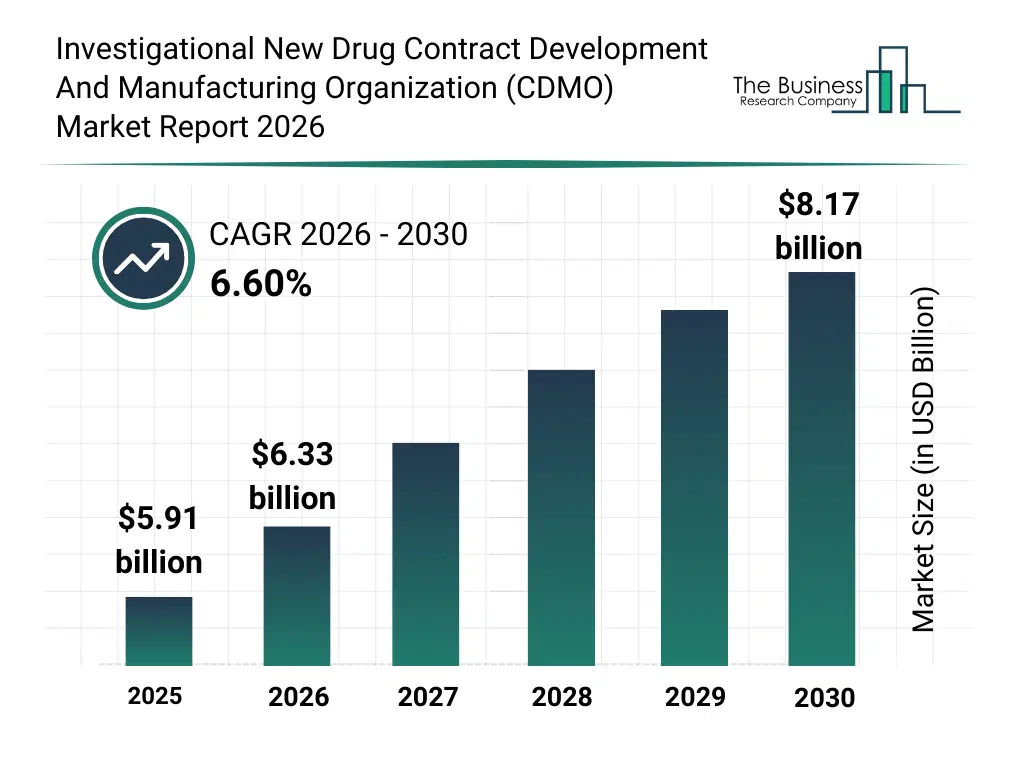
What Is The Investigational New Drug Contract Development And Manufacturing Organization (CDMO) Market Size and Share 2026?
The investigational new drug contract development and manufacturing organization (cdmo) market size has grown strongly in recent years. It will grow from $5.91 billion in 2025 to $6.33 billion in 2026 at a compound annual growth rate (CAGR) of 7.1%. The growth in the historic period can be attributed to limited availability of cdmos for investigational drugs, growing demand for small molecule synthesis, reliance on conventional manufacturing services, increasing clinical trial volumes, early adoption of regulatory support services.What Is The Investigational New Drug Contract Development And Manufacturing Organization (CDMO) Market Growth Forecast?
The investigational new drug contract development and manufacturing organization (cdmo) market size is expected to see strong growth in the next few years. It will grow to $8.17 billion in 2030 at a compound annual growth rate (CAGR) of 6.6%. The growth in the forecast period can be attributed to expansion of gene and cell therapy manufacturing capabilities, rising demand for monoclonal antibodies and recombinant proteins, investment in advanced formulation technologies, adoption of scalable manufacturing platforms, growth in global biotechnology research collaborations. Major trends in the forecast period include rising demand for early-stage drug development services, growth in clinical trial material supply services, increasing focus on regulatory compliance and quality standards, expansion of biologics and advanced therapy development, adoption of custom manufacturing solutions for small and large molecules.Global Investigational New Drug Contract Development And Manufacturing Organization (CDMO) Market Segmentation
1) By Product: Small Molecules, Large Molecules 2) By Formulation Type: Solid Formulations, Liquid Formulations, Injectable Formulations 3) By Service Type: Clinical Trial Material Supply, Regulatory Support Services, Manufacturing Services 4) By End-User: Biotechnology Companies, Academic And Research Institutes, Other End-Users Subsegments: 1) By Small Molecules: Synthetic Small Molecules, Semi-synthetic Small Molecules, Peptide-based Small Molecules, Oligonucleotides 2) By Large Molecules: Monoclonal Antibodies (mAbs), Recombinant Proteins, Vaccines, Cell Therapy Products, Gene Therapy Products, RNA-based TherapeuticsWhat Is The Driver Of The Investigational New Drug Contract Development And Manufacturing Organization (CDMO) Market?
The rising clinical trials are expected to propel the growth of the investigational new drug contract development and manufacturing organization (CDMO) market going forward. Clinical trials are carefully designed studies involving human participants to test how safe and effective a medical treatment, procedure, or intervention is under controlled conditions. The rise in clinical trials is largely driven by the growing demand for innovative treatments, as increasing rates of chronic and complex diseases push pharmaceutical and biotech companies to develop and test new therapies. Investigational new drug CDMOs support clinical trials by streamlining the development and manufacturing of trial-ready drug formulations, ensuring a timely and compliant supply of investigational products for each study phase. For instance, in December 2024, according to the Association of the British Pharmaceutical Industry, a UK-based pharmaceutical industry trade association, the total number of industry-sponsored clinical trials initiated in the United Kingdom increased from 411 in 2022 to 426 in 2023, while Phase III trial initiations grew by 16.5 percent over the same period, rising from 182 to 212. Therefore, the rising clinical trials are driving the growth of the investigational new drug contract development and manufacturing organization (CDMO) industry.Key Players In The Global Investigational New Drug Contract Development And Manufacturing Organization (CDMO) Market
Major companies operating in the investigational new drug contract development and manufacturing organization (cdmo) market are Thermo Fisher Scientific Inc., Fresenius Kabi AG, Lonza Group Ltd., Catalent Inc., Samsung Biologics Co. Ltd., Fareva SA, Recipharm AB, PCI Pharma Services, Vetter Pharma, Delpharm, Kindeva Drug Delivery LP, Ajinomoto Bio-Pharma Services, Cenexi SAS, Grand River Aseptic Manufacturing Inc., INCOG BioPharma Services, Aurigene Pharmaceutical Services, Jubilant HollisterStier LLC, Symbiosis Pharmaceutical Services Ltd., Afton Scientific Corp., Amaran Biotech Inc.Global Investigational New Drug Contract Development And Manufacturing Organization (CDMO) Market Trends and Insights
Major companies operating in the investigational new drug contract development and manufacturing organization (CDMO) market are focusing on strategic partnerships to strengthen development capabilities and speed up clinical entry. Strategic partnerships refer to formal alliances between two or more organizations that collaborate to achieve shared objectives, leveraging each other’s strengths, resources, or expertise to gain mutual benefits while remaining independent entities. For instance, in August 2023, Sartorius AG, a Germany-based life science company, partnered with Repligen Corporation, a US-based life sciences company, to launch an integrated system combining the BIOSTAT STR and XCell ATF technologies for upstream process intensification. This collaboration aims to streamline biopharmaceutical production by enhancing efficiency and scalability. The initiative marks Sartorius’ continued innovation in bioprocessing solutions, reinforcing its commitment to advancing the biomanufacturing industry.What Are Latest Mergers And Acquisitions In The Investigational New Drug Contract Development And Manufacturing Organization (CDMO) Market?
In January 2024, Alcami Corporation, a US-based pharmaceutical contract development and manufacturing organization (CDMO), acquired Pacific Pharmaceutical Services for an undisclosed amount. Through this acquisition, Alcami aims to expand its West Coast presence and enhance its analytical testing and quality control capabilities. Pacific Pharmaceutical Services is a US-based company providing pharmaceutical testing and quality assurance services.Regional Insights
North America was the largest region in the investigational new drug contract development and manufacturing organization (CDMO) market in 2025. Asia-Pacific is expected to be the fastest-growing region in the forecast period. The regions covered in this market report are Asia-Pacific, South East Asia, Western Europe, Eastern Europe, North America, South America, Middle East, Africa. The countries covered in this market report are Australia, Brazil, China, France, Germany, India, Indonesia, Japan, Taiwan, Russia, South Korea, UK, USA, Canada, Italy, Spain.What Defines the Investigational New Drug Contract Development And Manufacturing Organization (CDMO) Market?
The investigational new drug contract development and manufacturing organization (CDMO) market includes revenues earned by entities through analytical method development, clinical trial material manufacturing, process development, and quality control testing. The market value includes the value of related goods sold by the service provider or included within the service offering. Only goods and services traded between entities or sold to end consumers are included.How is Market Value Defined and Measured?
The market value is defined as the revenues that enterprises gain from the sale of goods and/or services within the specified market and geography through sales, grants, or donations in terms of the currency (in USD unless otherwise specified). The revenues for a specified geography are consumption values that are revenues generated by organizations in the specified geography within the market, irrespective of where they are produced. It does not include revenues from resales along the supply chain, either further along the supply chain or as part of other products.What Key Data and Analysis Are Included in the Investigational New Drug Contract Development And Manufacturing Organization (CDMO) Market Report 2026?
The investigational new drug contract development and manufacturing organization (cdmo) market research report is one of a series of new reports from The Business Research Company that provides market statistics, including industry global market size, regional shares, competitors with the market share, detailed market segments, market trends and opportunities, and any further data you may need to thrive in the investigational new drug contract development and manufacturing organization (cdmo) industry. The market research report delivers a complete perspective of everything you need, with an in-depth analysis of the current and future state of the industry.Investigational New Drug Contract Development And Manufacturing Organization (CDMO) Market Report Forecast Analysis
| Report Attribute | Details |
|---|---|
| Market Size Value In 2026 | $6.33 billion |
| Revenue Forecast In 2035 | $8.17 billion |
| Growth Rate | CAGR of 7.1% from 2026 to 2035 |
| Base Year For Estimation | 2025 |
| Actual Estimates/Historical Data | 2020-2025 |
| Forecast Period | 2026 - 2030 - 2035 |
| Market Representation | Revenue in USD Billion and CAGR from 2026 to 2035 |
| Segments Covered | Product, Formulation Type, Service Type, End-User |
| Regional Scope | Asia-Pacific, Western Europe, Eastern Europe, North America, South America, Middle East, Africa |
| Country Scope | The countries covered in the report are Australia, Brazil, China, France, Germany, India, ... |
| Key Companies Profiled | Thermo Fisher Scientific Inc., Fresenius Kabi AG, Lonza Group Ltd., Catalent Inc., Samsung Biologics Co. Ltd., Fareva SA, Recipharm AB, PCI Pharma Services, Vetter Pharma, Delpharm, Kindeva Drug Delivery LP, Ajinomoto Bio-Pharma Services, Cenexi SAS, Grand River Aseptic Manufacturing Inc., INCOG BioPharma Services, Aurigene Pharmaceutical Services, Jubilant HollisterStier LLC, Symbiosis Pharmaceutical Services Ltd., Afton Scientific Corp., Amaran Biotech Inc. |
| Customization Scope | Request for Customization |
| Pricing And Purchase Options | Explore Purchase Options |
