
Krabbe Disease Treatment Market Report 2026
Global Outlook – By Treatment Type (Medication, Enzyme Replacement Therapy, Gene Therapy, Stem Cell Therapy), By Diagnostic Type (Newborn Screening, Genetic Testing), By Distribution Channel (Retail Pharmacies, E-Commerce, Hospital Pharmacies), By End-User (Hospitals And Clinics, Research Centers, Laboratories, Other End-Users) - Market Size, Trends, And Global Forecast 2026-2035
Krabbe Disease Treatment Market Overview
• Krabbe Disease Treatment market size has reached to $2.47 billion in 2025 • Expected to grow to $3.36 billion in 2030 at a compound annual growth rate (CAGR) of 6.3% • Growth Driver: Expansion Of Newborn Screening Programs Fueling The Growth Of The Market Due To Early Diagnosis And Timely Intervention • Market Trend: Innovative Gene Therapy Programs Driving Growth In The Market Through Enhanced Clinical Outcomes And Regulatory Support • North America was the largest region in 2025 and Asia-Pacific is the fastest growing region.What Is Covered Under Krabbe Disease Treatment Market?
Krabbe disease treatment refers to medical interventions aimed at managing the symptoms and slowing the progression of Krabbe disease, a rare and often fatal genetic disorder affecting the nervous system. Treatment may include hematopoietic stem cell transplantation (HSCT), especially effective if performed early, as well as supportive care such as physical therapy, seizure management, nutritional support, and pain relief. The main types of krabbe disease treatment are medication, enzyme replacement therapy, gene therapy, and stem cell therapy. Medications for Krabbe disease, such as anticonvulsants and muscle relaxants, help manage symptoms such as seizures and muscle stiffness to improve quality of life. It is diagnosed by various diagnostics such as newborn screening and genetic testing and is distributed through several distribution channels, including retail pharmacies, e-commerce, and hospital pharmacies. It is applicable for various applications, such as infantile onset, late onset, and adult onset, and is used by several end users, including hospitals and clinics, research centers, laboratories, and others.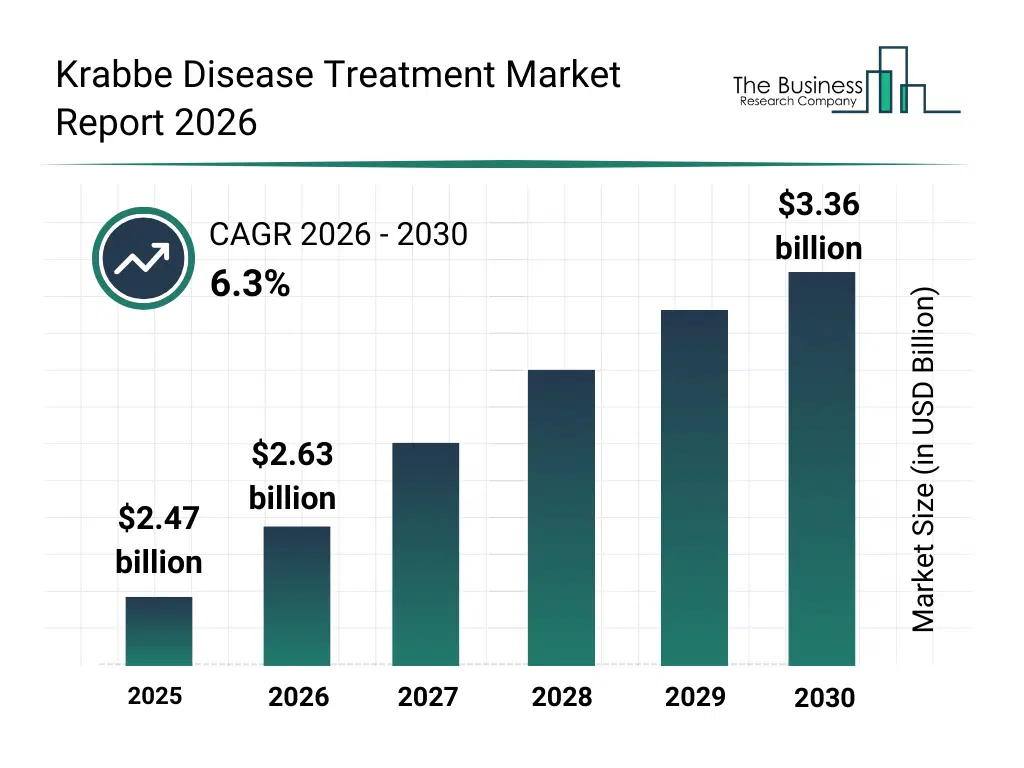
What Is The Krabbe Disease Treatment Market Size and Share 2026?
The krabbe disease treatment market size has grown strongly in recent years. It will grow from $2.47 billion in 2025 to $2.63 billion in 2026 at a compound annual growth rate (CAGR) of 6.6%. The growth in the historic period can be attributed to improved awareness of rare neurodegenerative disorders, expansion of newborn screening initiatives, early clinical use of hsct therapies, growth in rare disease research funding, availability of supportive care treatments.What Is The Krabbe Disease Treatment Market Growth Forecast?
The krabbe disease treatment market size is expected to see strong growth in the next few years. It will grow to $3.36 billion in 2030 at a compound annual growth rate (CAGR) of 6.3%. The growth in the forecast period can be attributed to increasing gene therapy clinical trials, rising investments in precision medicine for rare diseases, expansion of early diagnostic technologies, growing focus on disease-modifying treatments, increased regulatory support for orphan drugs. Major trends in the forecast period include increasing adoption of gene therapy approaches, rising use of early newborn screening programs, growing focus on early hsct intervention, expansion of supportive and symptomatic care protocols, increased research into enzyme replacement solutions.Global Krabbe Disease Treatment Market Segmentation
1) By Treatment Type: Medication, Enzyme Replacement Therapy, Gene Therapy, Stem Cell Therapy 2) By Diagnostic Type: Newborn Screening, Genetic Testing 3) By Distribution Channel: Retail Pharmacies, E-Commerce, Hospital Pharmacies 4) By End-User: Hospitals And Clinics, Research Centers, Laboratories, Other End-Users Subsegments: 1) By Medication: Anticonvulsants, Muscle Relaxants, Anti-Inflammatory Drugs, Pain Management Medications, Psychotropic Agents 2) By Enzyme Replacement Therapy (ERT): Intravenous Enzyme Infusion, Intrathecal Enzyme Administration, PEGylated Enzyme Formulations, Recombinant Galactocerebrosidase (GALC) 3) By Gene Therapy: Adeno-Associated Virus (AAV)-Based Gene Therapy, Lentiviral Vector-Based Gene Therapy, In Vivo Gene Therapy, Ex Vivo Gene-Modified Stem Cell Therapy 4) By Stem Cell Therapy: Hematopoietic Stem Cell Transplantation (HSCT), Umbilical Cord Blood Stem Cell Therapy, Bone Marrow Transplant, Allogeneic Stem Cell TherapyWhat Is The Driver Of The Krabbe Disease Treatment Market?
The increasing prevalence of newborn screening programs is expected to propel the growth of the krabbe disease treatment market going forward. Newborn screening programs are health programs that check babies soon after birth for potentially serious but treatable genetic, metabolic, and developmental conditions. Advancements in medical technology are driving the rise of newborn screening by enabling earlier, accurate detection and timely prevention of serious health issues. Newborn screening programs enable early detection of krabbe disease, allowing for prompt interventions before symptoms develop. This early diagnosis facilitates treatments such as stem cell transplantation, improving outcomes and slowing disease progression. For instance, in November 2023, according to the Michigan Department of Health and Human Services, a US-based state agency, as of 2022, over 7.7 million infants have been screened, with more than 8,000 diagnosed with conditions from the NBS blood spot panel. In comparison, among the 100,176 infants screened in 2022, the majority were Michigan residents, and 322 (0.3%) were diagnosed with a disease. Therefore, the increasing prevalence of newborn screening programs is driving the growth of the krabbe disease treatment industry.Key Players In The Global Krabbe Disease Treatment Market
Major companies operating in the krabbe disease treatment market are Ajinomoto Co Inc, Passage Bio Inc, Magenta Therapeutics Inc, Gain Therapeutics Inc, M6P Therapeutics, Neurogene Inc, Greenwood Genetic Center, PreventionGenetics, MediciNova Inc, Polaryx Therapeutics Inc, Orchard Therapeutics Ltd, AVROBIO Inc, Bluebird Bio Inc, Regenxbio Inc, Homology Medicines Inc, Sangamo Therapeutics Inc, Editas Medicine Inc, CRISPR Therapeutics AG, Pfizer Inc, Sanofi SA, Takeda Pharmaceutical Company Limited, Amgen Inc, Biogen Inc, JCR Pharmaceuticals Co Ltd, Ultragenyx Pharmaceutical IncGlobal Krabbe Disease Treatment Market Trends and Insights
Major companies operating in the krabbe disease treatment market are focused on developing advanced gene therapy programs to enhance treatment effectiveness and broaden global accessibility. Gene therapy programs involve modifying or replacing genes within a person's cells to treat or prevent diseases. These therapies aim to correct genetic mutations, providing potential cures for genetic disorders by introducing, removing, or altering specific genes. For instance, in March 2024, Forge Biologics, a US-based gene therapy company, introduced FBX-101, an innovative adeno-associated viral (AAV) gene therapy. The therapy received the Innovation Passport designation from the UK’s Medicines and Healthcare Products Regulatory Agency (MHRA), enabling it to enter the Innovative Licensing and Access Pathway (ILAP). This pathway is designed to accelerate the regulatory and market access processes for treatments targeting serious or life-threatening conditions. FBX-101 has demonstrated encouraging safety and efficacy results in early clinical trials and works by delivering a functional GALC gene to patients. It is intended to improve clinical outcomes for children affected by this rare and fatal neurodegenerative disease.What Are Latest Mergers And Acquisitions In The Krabbe Disease Treatment Market?
In November 2023, Ajinomoto Co., Inc., a Japan-based biotechnology and food company, acquired Forge Biologics Holdings, LLC for $554 million. Through this acquisition, Ajinomoto aims to expand its presence in the gene therapy space and strengthen its biopharma services business. It aligns with Ajinomoto’s ASV Initiatives 2030 Roadmap to make healthcare a core growth area by integrating advanced gene therapy development and manufacturing capabilities. Forge Biologics Holdings LLC is a US-based provider of gene therapy for krabbe disease.Regional Outlook
North America was the largest region in the krabbe disease treatment market in 2025. Asia-Pacific is expected to be the fastest-growing region in the forecast period. The regions covered in this market report are Asia-Pacific, South East Asia, Western Europe, Eastern Europe, North America, South America, Middle East, Africa. The countries covered in this market report are Australia, Brazil, China, France, Germany, India, Indonesia, Japan, Taiwan, Russia, South Korea, UK, USA, Canada, Italy, Spain.What Defines the Krabbe Disease Treatment Market?
The krabbe disease treatment market consists of revenues earned by entities providing services such as genetic counseling, enzyme activity testing, hematopoietic stem cell transplantation (HSCT), and supportive care management. The market value includes the value of related goods sold by the service provider or included within the service offering. The krabbe disease treatment market also includes sales of viral vectors, infusion equipment, diagnostic kits, and neurological monitoring systems. Values in this market are ‘factory gate’ values, that is the value of goods sold by the manufacturers or creators of the goods, whether to other entities (including downstream manufacturers, wholesalers, distributors and retailers) or directly to end customers. The value of goods in this market includes related services sold by the creators of the goods.How is Market Value Defined and Measured?
The market value is defined as the revenues that enterprises gain from the sale of goods and/or services within the specified market and geography through sales, grants, or donations in terms of the currency (in USD unless otherwise specified). The revenues for a specified geography are consumption values that are revenues generated by organizations in the specified geography within the market, irrespective of where they are produced. It does not include revenues from resales along the supply chain, either further along the supply chain or as part of other products.What Key Data and Analysis Are Included in the Krabbe Disease Treatment Market Report 2026?
The krabbe disease treatment market research report is one of a series of new reports from The Business Research Company that provides market statistics, including industry global market size, regional shares, competitors with the market share, detailed market segments, market trends and opportunities, and any further data you may need to thrive in the krabbe disease treatment industry. The market research report delivers a complete perspective of everything you need, with an in-depth analysis of the current and future state of the industry.Krabbe Disease Treatment Market Report Forecast Analysis
| Report Attribute | Details |
|---|---|
| Market Size Value In 2026 | $2.63 billion |
| Revenue Forecast In 2035 | $3.36 billion |
| Growth Rate | CAGR of 6.6% from 2026 to 2035 |
| Base Year For Estimation | 2025 |
| Actual Estimates/Historical Data | 2020-2025 |
| Forecast Period | 2026 - 2030 - 2035 |
| Market Representation | Revenue in USD Billion and CAGR from 2026 to 2035 |
| Segments Covered | Treatment Type, Diagnostic Type, Distribution Channel, End-User |
| Regional Scope | Asia-Pacific, Western Europe, Eastern Europe, North America, South America, Middle East, Africa |
| Country Scope | The countries covered in the report are Australia, Brazil, China, France, Germany, India, ... |
| Key Companies Profiled | Ajinomoto Co Inc, Passage Bio Inc, Magenta Therapeutics Inc, Gain Therapeutics Inc, M6P Therapeutics, Neurogene Inc, Greenwood Genetic Center, PreventionGenetics, MediciNova Inc, Polaryx Therapeutics Inc, Orchard Therapeutics Ltd, AVROBIO Inc, Bluebird Bio Inc, Regenxbio Inc, Homology Medicines Inc, Sangamo Therapeutics Inc, Editas Medicine Inc, CRISPR Therapeutics AG, Pfizer Inc, Sanofi SA, Takeda Pharmaceutical Company Limited, Amgen Inc, Biogen Inc, JCR Pharmaceuticals Co Ltd, Ultragenyx Pharmaceutical Inc |
| Customization Scope | Request for Customization |
| Pricing And Purchase Options | Explore Purchase Options |
