
Lateral Flow Immunoassay (LFIA) Based Rapid Test Market Report 2026
Global Outlook – By Technique (Competitive Assay, Sandwich Assay, Multiplex Detection Assay), By Application (Infectious Disease, Pregnancy And Fertility, Toxicology, Cardiac Marker And Cholesterol Testing, Other Applications), By End-Users (Hospitals And Clinics, Diagnostic Laboratories, Home Care, Other End Users) – Market Size, Trends, Strategies, and Forecast to 2035
Lateral Flow Immunoassay (LFIA) Based Rapid Test Market Overview
• Lateral Flow Immunoassay (LFIA) Based Rapid Test market size has reached to $7.17 billion in 2025 • Expected to grow to $8.67 billion in 2030 at a compound annual growth rate (CAGR) of 4.3% • Growth Driver: Elevated Prevalence Of Chronic Diseases Fueling LFIA-Based Rapid Test Market Growth • Market Trend: Roche Launches Advanced Cobas Pro Integrated Solutions For Streamlined Patient Care • North America was the largest region in 2025 and Asia-Pacific is the fastest growing region.What Is Covered Under Lateral Flow Immunoassay (LFIA) Based Rapid Test Market?
Lateral Flow Immunoassay (LFIA) based rapid test is a simple technology-based diagnostic device that is used to determine the presence of a target such as pathogens or biomarkers in samples collected from the human body. These samples may include urine, saliva, blood, sweat and other fluids. The main techniques in lateral flow immunoassay-based rapid testing are competitive assay, sandwich assay, and multiplex detection assay. The competitive assay is used to assess the existence of a target, such as pathogens or biomarkers, in the samples. The applications are infectious disease, pregnancy and fertility, toxicology, and others that are used in hospitals and clinics, diagnostic laboratory, home care, and others.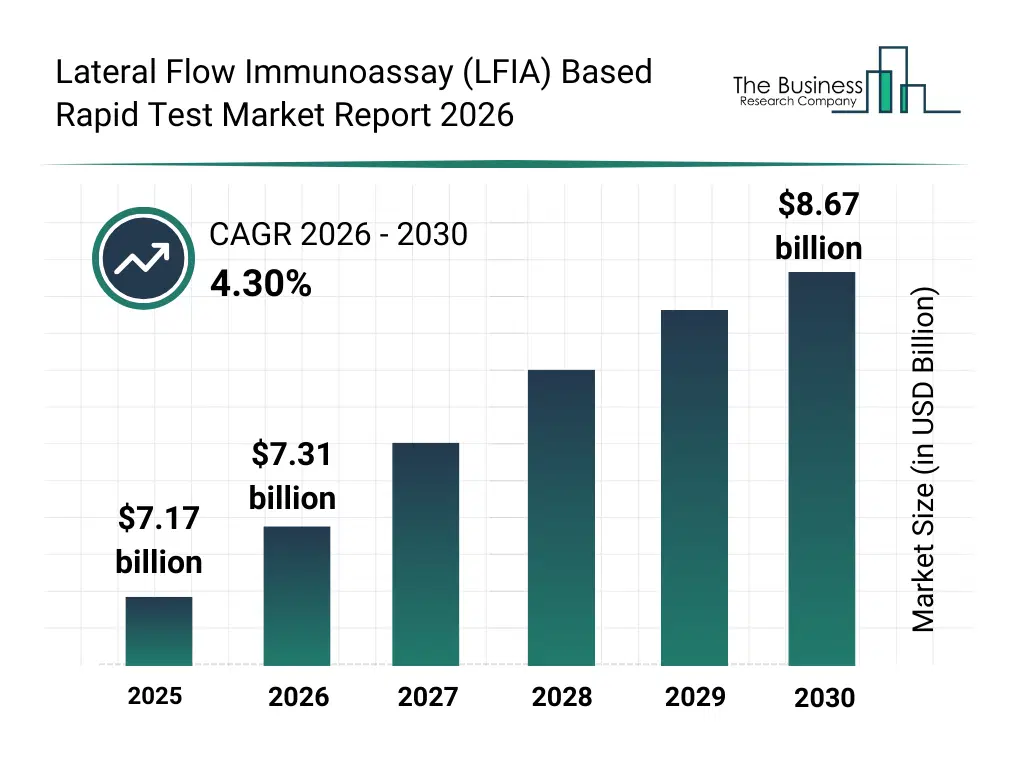
What Is The Lateral Flow Immunoassay (LFIA) Based Rapid Test Market Size and Share 2026?
The lateral flow immunoassay (lfia) based rapid test market size has grown steadily in recent years. It will grow from $7.17 billion in 2025 to $7.31 billion in 2026 at a compound annual growth rate (CAGR) of 2.0%. The growth in the historic period can be attributed to expansion of rapid infectious disease screening, increasing adoption of pregnancy and fertility tests, growth of point-of-care diagnostics, availability of low-cost rapid tests, rising public health screening initiatives.What Is The Lateral Flow Immunoassay (LFIA) Based Rapid Test Market Growth Forecast?
The lateral flow immunoassay (lfia) based rapid test market size is expected to see steady growth in the next few years. It will grow to $8.67 billion in 2030 at a compound annual growth rate (CAGR) of 4.3%. The growth in the forecast period can be attributed to increasing investments in decentralized diagnostics, expansion of self-testing applications, growing demand for rapid cardiac and toxicology tests, rising integration with digital health platforms, increasing focus on early disease detection. Major trends in the forecast period include increasing demand for rapid immunoassay-based testing, rising adoption of home-use lfia kits, growing use of multiplex detection assays, expansion of infectious disease testing applications, enhanced focus on user-friendly test design.Global Lateral Flow Immunoassay (LFIA) Based Rapid Test Market Segmentation
1) By Technique: Competitive Assay, Sandwich Assay, Multiplex Detection Assay 2) By Application: Infectious Disease, Pregnancy And Fertility, Toxicology, Cardiac Marker And Cholesterol Testing, Other Applications 3) By End-Users: Hospitals And Clinics, Diagnostic Laboratories, Home Care, Other End Users Subsegments: 1) By Competitive Assay: Direct Competitive Assays, Indirect Competitive Assays 2) By Sandwich Assay: Single-step Sandwich Assays, Two-step Sandwich Assays 3) By Multiplex Detection Assay: Multi-analyte Assays, Simultaneous Detection Of Various TargetsWhat Is The Driver Of The Lateral Flow Immunoassay (LFIA) Based Rapid Test Market?
The increasing prevalence of chronic diseases is expected to propel the growth of the lateral flow immunoassay (LFIA) based rapid test market going forward. Chronic diseases, also known as non-communicable diseases (NCDs), are long-term health conditions that persist over an extended period, often for a person's lifetime. Lateral flow immunoassay (LFIA) based rapid tests offers early detection, point-of-care testing, rapid results, regular monitoring, and reduces healthcare costs involved in chronic diseases care. LFIA-based rapid tests are appreciated in the management of chronic diseases because they offer prompt and obtainable results, enabling patients and healthcare professionals to make prompt decisions about treatment and dietary changes. For instance, in June 2024, according to the National Health Service, a UK-based government department, 3,615,330 individuals registered with a general practitioner (GP) were diagnosed with non-diabetic hyperglycemia or pre-diabetes (a condition with elevated blood sugar levels, not high enough to be classified as diabetes) in 2023, marking an 18% increase from 3,065,825 cases in 2022. Therefore, the increasing prevalence of chronic diseases is driving the growth of the lateral flow immunoassay (LFIA) based rapid test industry.Key Players In The Global Lateral Flow Immunoassay (LFIA) Based Rapid Test Market
Major companies operating in the lateral flow immunoassay (lfia) based rapid test market are Abbott Laboratories, F. Hoffmann-La Roche, Quidel Corporation, Becton Dickinson and Company, Danaher Corporation, Thermo Fisher Scientific Inc., Bio-Rad Laboratories Inc., PerkinElmer Inc., Hologic Inc., LTEK Co. Ltd., JOYSBIO Biotechnology, Jiangsu Medomics Medical Technologies, Diagnocure, Ubio Biotechnology Systems, Elabscience® Immunoassay Kits, FyoniBio / Glycotope GmbH, Atomo Diagnostics Limited, INDICAL Bioscience, Lionex Gmbh, BIOMED Labordiagnostik GmbH, EKF Diagnostics, Launch Diagnostics, Diagnostic Instruments Ltd, Qiagen N.V., Merck KGaA, 3a Diagnostics, Roche Holding AG, Biomaxima, SensDx, Vitrosens Biotechnology, Bioeasy Biotechnology, Fujirebio, Immunodiagnostic Systems Holdings PLC, Mesa Biotech Inc, CTK BiotechGlobal Lateral Flow Immunoassay (LFIA) Based Rapid Test Market Trends and Insights
Major companies operating in the lateral flow immunoassay (LFIA)-based rapid test market are focused on technologies such as Integrated Health Solutions to enhance diagnostic efficiency, streamline workflows, and provide comprehensive patient care through better data sharing and collaboration across healthcare platforms. Integrated Health Solutions refer to comprehensive healthcare systems that connect various services, technologies, and data to improve patient care and streamline operations. For instance, in June 2024, F. Hoffmann-La Roche AG, a Switzerland-based pharmaceutical company, launched the analytical units for its Cobas Pro integrated solutions. The Cobas Pro system can process up to 1,000 samples per hour with the sample supply unit and offers high-speed analytical units capable of performing up to 900 tests per hour for the ISE (ion-selective electrode) and 1,000 tests per hour for the Cobas C 503 unit, which focuses on clinical chemistry.What Are Latest Mergers And Acquisitions In The Lateral Flow Immunoassay (LFIA) Based Rapid Test Market?
In August 2023, Danaher Corporation, a US-based medical products company acquired Abcam plc for $5.7 billion. The acquisition of Abcam is consistent with Danaher's aim to grow its life sciences business and assist its clients in resolving some of the biggest healthcare problems in the world. The transaction is anticipated to increase value for shareholders while providing outstanding results for the workforce and clients. Abcam plc is a UK-based biotechnology company that manufactures lateral flow immunoassay (LFIA) based rapid tests.Regional Outlook
North America was the largest region in the lateral flow immunoassay (LFIA) based rapid test market in 2025. Asia-Pacific is expected to be the fastest-growing region in the forecast period. The regions covered in this market report are Asia-Pacific, South East Asia, Western Europe, Eastern Europe, North America, South America, Middle East, Africa. The countries covered in this market report are Australia, Brazil, China, France, Germany, India, Indonesia, Japan, Taiwan, Russia, South Korea, UK, USA, Canada, Italy, SpainWhat Defines the Lateral Flow Immunoassay (LFIA) Based Rapid Test Market?
Lateral Flow Immunoassay (LFIA) based rapid test market consists of sales of conjugate pads, nitrocellulose membrane and absorbent pad. Values in this market are factory gate values, that is the value of goods sold by the manufacturers or creators of the goods, whether to other entities (including downstream manufacturers, wholesalers, distributors, and retailers) or directly to end customers.How is Market Value Defined and Measured?
The market value is defined as the revenues that enterprises gain from the sale of goods and/or services within the specified market and geography through sales, grants, or donations in terms of the currency (in USD unless otherwise specified). The revenues for a specified geography are consumption values that are revenues generated by organizations in the specified geography within the market, irrespective of where they are produced. It does not include revenues from resales along the supply chain, either further along the supply chain or as part of other products.Lateral Flow Immunoassay (LFIA) Based Rapid Test Market Report Forecast Analysis
| Report Attribute | Details |
|---|---|
| Market Size Value In 2026 | $7.31 billion |
| Revenue Forecast In 2035 | $8.67 billion |
| Growth Rate | CAGR of 2.0% from 2026 to 2035 |
| Base Year For Estimation | 2025 |
| Actual Estimates/Historical Data | 2020-2025 |
| Forecast Period | 2026 - 2030 - 2035 |
| Market Representation | Revenue in USD Billion and CAGR from 2026 to 2035 |
| Segments Covered | Technique, Application, End-Users |
| Regional Scope | Asia-Pacific, Western Europe, Eastern Europe, North America, South America, Middle East, Africa |
| Country Scope | The countries covered in the report are Australia, Brazil, China, France, Germany, India, ... |
| Key Companies Profiled | Abbott Laboratories, F. Hoffmann-La Roche, Quidel Corporation, Becton Dickinson and Company, Danaher Corporation, Thermo Fisher Scientific Inc., Bio-Rad Laboratories Inc., PerkinElmer Inc., Hologic Inc., LTEK Co. Ltd., JOYSBIO Biotechnology, Jiangsu Medomics Medical Technologies, Diagnocure, Ubio Biotechnology Systems, Elabscience® Immunoassay Kits, FyoniBio / Glycotope GmbH, Atomo Diagnostics Limited, INDICAL Bioscience, Lionex Gmbh, BIOMED Labordiagnostik GmbH, EKF Diagnostics, Launch Diagnostics, Diagnostic Instruments Ltd, Qiagen N.V., Merck KGaA, 3a Diagnostics, Roche Holding AG, Biomaxima, SensDx, Vitrosens Biotechnology, Bioeasy Biotechnology, Fujirebio, Immunodiagnostic Systems Holdings PLC, Mesa Biotech Inc, CTK Biotech |
| Customization Scope | Request for Customization |
| Pricing And Purchase Options | Explore Purchase Options |
