
Left Atrial Appendage Closure Device Market Report 2026
Global Outlook – By Product (Epicardial Left Atrial Appendage (LAA) Devices, Endocardial Left Atrial Appendage (LAA) Devices), By Procedure (Percutaneous, Surgical), By End-User (Hospital, Ambulatory Surgical Centers, Other End-Users) – Market Size, Trends, Strategies, and Forecast to 2035
Left Atrial Appendage Closure Device Market Overview
• Left Atrial Appendage Closure Device market size has reached to $1.99 billion in 2025 • Expected to grow to $4.37 billion in 2030 at a compound annual growth rate (CAGR) of 17% • Growth Driver: Elevated Prevalence Of Atrial Fibrillation Fuels Growth In The Left Atrial Appendage (LAA) Closure Device Market • Market Trend: Boston Scientific Unveils Next-Generation WATCHMAN FLX™ Pro with Enhanced Size Range and Visibility Features • North America was the largest region in 2025.What Is Covered Under Left Atrial Appendage Closure Device Market?
A left atrial appendage closure device is a medical implant used to treat certain heart conditions, specifically in atrial fibrillation (AF) patients. These devices are implanted in a minimally invasive surgery to lower the risk of stroke in AF patients by sealing off or occluding the left atrial appendage (LAA), preventing blood from entering and clotting. The main types of products in left atrial appendage closure devices are epicardial left atrial appendage (LAA) devices and endocardial left atrial appendage (LAA) devices. Epicardial left atrial appendage (LAA) devices refer to medical devices and techniques used for the closure or exclusion of the left atrial appendage from the outside of the heart. It includes percutaneous and surgical procedures, and the various end-users include hospitals, ambulatory surgical centers and others.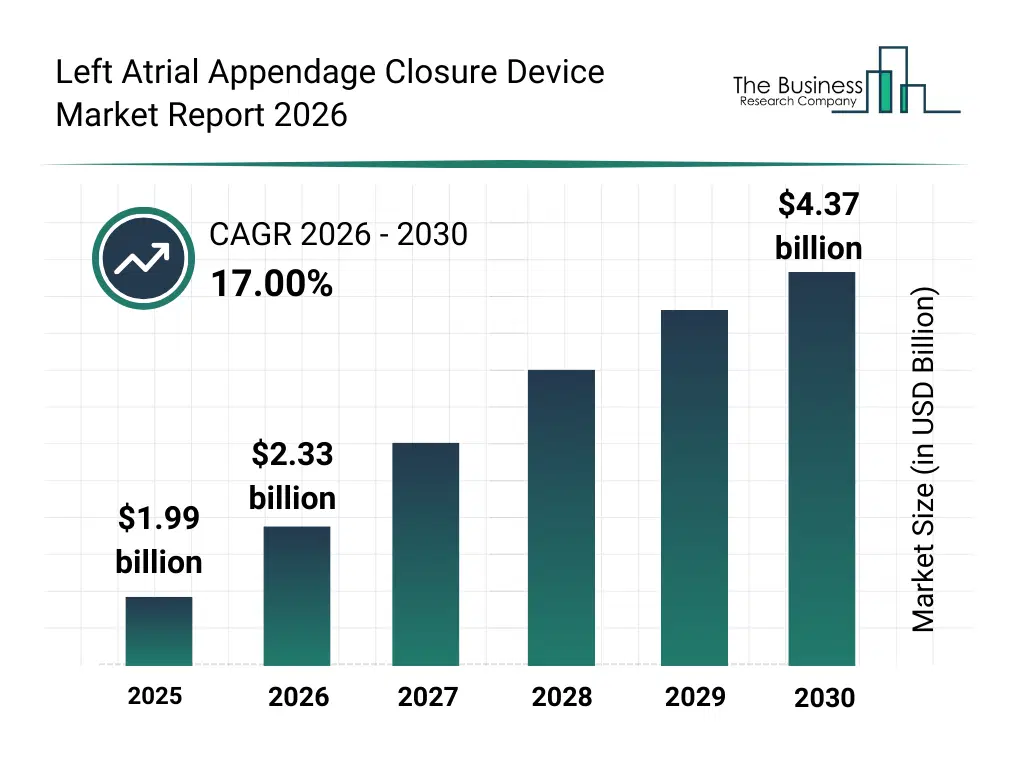
What Is The Left Atrial Appendage Closure Device Market Size and Share 2026?
The left atrial appendage closure device market size has grown rapidly in recent years. It will grow from $1.99 billion in 2025 to $2.33 billion in 2026 at a compound annual growth rate (CAGR) of 17.4%. The growth in the historic period can be attributed to increasing prevalence of atrial fibrillation, rising stroke incidence among aging populations, expansion of interventional cardiology procedures, growing availability of minimally invasive cardiac treatments, improved clinical outcomes data.What Is The Left Atrial Appendage Closure Device Market Growth Forecast?
The left atrial appendage closure device market size is expected to see rapid growth in the next few years. It will grow to $4.37 billion in 2030 at a compound annual growth rate (CAGR) of 17.0%. The growth in the forecast period can be attributed to increasing adoption of non-pharmacological stroke prevention, expansion of cardiac catheterization labs, growing demand for personalized cardiac implants, rising investments in structural heart disease treatments, technological advancements in implant materials and delivery systems. Major trends in the forecast period include increasing adoption of minimally invasive cardiac implants, rising demand for stroke prevention devices, growing use of percutaneous laa closure procedures, expansion of advanced occlusion device designs, enhanced focus on patient-specific cardiac solutions.Global Left Atrial Appendage Closure Device Market Segmentation
1) By Product: Epicardial Left Atrial Appendage (LAA) Devices, Endocardial Left Atrial Appendage (LAA) Devices 2) By Procedure: Percutaneous, Surgical 3) By End-User: Hospital, Ambulatory Surgical Centers, Other End-Users Subsegments: 1) By Epicardial Left Atrial Appendage (LAA) Devices: Suture-Based Closure Devices, Ligation-Based Closure Devices, Clip-Based Closure Devices 2) By Endocardial Left Atrial Appendage (LAA) Devices: Plug-Based Closure Devices, Disc-Based Closure Devices, Mesh-Based Closure DevicesWhat Is The Driver Of The Left Atrial Appendage Closure Device Market?
The rising prevalence of atrial fibrillation is expected to propel the growth of the left atrial appendage (LAA) closure device market going forward. Atrial fibrillation is a medical condition characterized by an irregular and often rapid heartbeat. It occurs when the heart's upper chambers, called the atria, do not beat in a coordinated and rhythmic manner. Left atrial appendage (LAA) closure devices are primarily used in treating atrial fibrillation by reducing the risk of stroke in patients with this cardiac arrhythmia and preventing blood clots from forming or escaping. For instance, in May 2024, according to the Centre’s for Disease Control and Prevention, a US-based public health agency, in 2023, the number of adults living with atrial fibrillation in the United States has risen to an estimated 12.1?million, up from approximately 5.2?million in 2022. Therefore, the rising prevalence of atrial fibrillation is driving the growth of the left atrial appendage (LAA) closure device market.Key Players In The Global Left Atrial Appendage Closure Device Market
Major companies operating in the left atrial appendage closure device market are Boston Scientific Corporation, Abbott Laboratories, Medtronic plc, AtriCure Inc, Lifetech Scientific Corporation, Occlutech GmbH, Cardia Inc, SentreHEART Inc, Coherex Medical Inc, Lepu Medical Technology Co Ltd, MicroPort Scientific Corporation, NMT Medical Inc, Transcatheter Technologies GmbH, Shanghai Shape Memory Alloy Co Ltd, Braile Biomedica, Innovative Cardiovascular Solutions LLC, Eclipse Medical, PushMed (PushMed Medical), Cardiawave, Catheter Precision Inc.Global Left Atrial Appendage Closure Device Market Trends and Insights
Major companies operating in the left atrial appendage closure device market are focusing on technological innovation, such as expanded-size, radiopaque-marked closure systems with advanced polymer coatings, which enhance procedural accuracy, visibility, and patient safety while accommodating a broader range of left atrial appendage anatomies. Expanded-size, radiopaque-marked closure systems integrate multiple device sizes, radiopaque markers, and biocompatible polymer coatings to enable precise placement under fluoroscopy, reduce the risk of stroke from atrial fibrillation, and minimize complications associated with anticoagulation therapy. For instance, in April 2024, Boston Scientific Japan Co., Ltd., a Tokyo-based cardiovascular medical device company, launched the WATCHMAN FLX Pro Left Atrial Appendage Closure System nationwide. The latest-generation system introduces a new 40mm device size for larger left atrial appendages, incorporates three radiopaque markers for improved visibility during implantation, and features HEMOCOAT polymer coating with a proven track record, enhancing procedural performance, safety, and treatment options for patients with non-valvular atrial fibrillation.What Are Latest Mergers And Acquisitions In The Left Atrial Appendage Closure Device Market?
In November 2023, Johnson & Johnson MedTech, a US-based medical technology company, acquired Laminar, Inc. for $400 million upfront, with additional potential milestone payments in 2024 and beyond. With this acquisition, Johnson & Johnson MedTech aims to strengthen its position in high-growth cardiac device segments by adding Laminar’s innovative left atrial appendage elimination technology to reduce stroke risk in patients with non-valvular atrial fibrillation. Laminar, Inc. is a US-based privately held medical device company specializing in left atrial appendage elimination solutions.Regional Outlook
North America was the largest region in the left atrial appendage closure devices market in 2025. The regions covered in this market report are Asia-Pacific, South East Asia, Western Europe, Eastern Europe, North America, South America, Middle East, Africa. The countries covered in this market report are Australia, Brazil, China, France, Germany, India, Indonesia, Japan, Taiwan, Russia, South Korea, UK, USA, Canada, Italy, SpainWhat Defines the Left Atrial Appendage Closure Device Market?
The left atrial appendage closure device market consists of sales of transcatheter devices, suture delivery devices, sealing material, delivery catheters, and imaging markers. Values in this market are ‘factory gate’ values, that is the value of goods sold by the manufacturers or creators of the goods, whether to other entities (including downstream manufacturers, wholesalers, distributors and retailers) or directly to end customers. The value of goods in this market includes related services sold by the creators of the goods.How is Market Value Defined and Measured?
The market value is defined as the revenues that enterprises gain from the sale of goods and/or services within the specified market and geography through sales, grants, or donations in terms of the currency (in USD unless otherwise specified). The revenues for a specified geography are consumption values that are revenues generated by organizations in the specified geography within the market, irrespective of where they are produced. It does not include revenues from resales along the supply chain, either further along the supply chain or as part of other products.Left Atrial Appendage Closure Device Market Report Forecast Analysis
| Report Attribute | Details |
|---|---|
| Market Size Value In 2026 | $2.33 billion |
| Revenue Forecast In 2035 | $4.37 billion |
| Growth Rate | CAGR of 17.4% from 2026 to 2035 |
| Base Year For Estimation | 2025 |
| Actual Estimates/Historical Data | 2020-2025 |
| Forecast Period | 2026 - 2030 - 2035 |
| Market Representation | Revenue in USD Billion and CAGR from 2026 to 2035 |
| Segments Covered | Product, Procedure, End-User |
| Regional Scope | Asia-Pacific, Western Europe, Eastern Europe, North America, South America, Middle East, Africa |
| Country Scope | The countries covered in the report are Australia, Brazil, China, France, Germany, India, ... |
| Key Companies Profiled | Boston Scientific Corporation, Abbott Laboratories, Medtronic plc, AtriCure Inc, Lifetech Scientific Corporation, Occlutech GmbH, Cardia Inc, SentreHEART Inc, Coherex Medical Inc, Lepu Medical Technology Co Ltd, MicroPort Scientific Corporation, NMT Medical Inc, Transcatheter Technologies GmbH, Shanghai Shape Memory Alloy Co Ltd, Braile Biomedica, Innovative Cardiovascular Solutions LLC, Eclipse Medical, PushMed (PushMed Medical), Cardiawave, Catheter Precision Inc. |
| Customization Scope | Request for Customization |
| Pricing And Purchase Options | Explore Purchase Options |
