
Low Molecular Weight Heparin Market Report 2026
Global Outlook – By Product Type (Enoxaparin, Dalteparin, Tinzaparin, Fraxiparine, Nadroparin, Other Product Types), By Packaging (Multi-Vials, Prefilled Syringes), By Application (Deep Vein Thrombosis, Acute Coronary Syndrome, Pulmonary Embolism, Other Applications) - Market Size, Trends, And Global Forecast 2026-2035
Low Molecular Weight Heparin Market Overview
• Low Molecular Weight Heparin market size has reached to $5.29 billion in 2025 • Expected to grow to $8.06 billion in 2030 at a compound annual growth rate (CAGR) of 8.6% • Growth Driver: Impact Of Increased Blood Disorders On The Low Molecular Weight Heparin Market • Market Trend: Novel Developments In Heparin With Low Molecular Weight Preservative-Free Prefilled Syringes • North America was the largest region in 2025 and Asia-Pacific is the fastest growing region.What Is Covered Under Low Molecular Weight Heparin Market?
Low molecular weight heparin (LMWH) refers to a class of anticoagulant medications derived from standard heparin through chemical or enzymatic depolymerization, resulting in smaller fragments. LMWHs are used to prevent and treat blood clots by inhibiting factor Xa and, to a lesser extent, thrombin. They are favored over unfractionated heparin due to their more predictable pharmacokinetics, longer half-life, and lower risk of adverse effects such as heparin-induced thrombocytopenia. The main product types of low molecular weight heparin are enoxaparin, dalteparin, tinzaparin, fraxiparine, nadroparin, and others. Enoxaparin is a low molecular weight heparin (LMWH) medication used for preventing blood clots in conditions such as deep vein thrombosis and pulmonary embolism. The packaging comprises multi-vials and prefilled syringes designed for various applications, including deep vein thrombosis and acute coronary syndrome.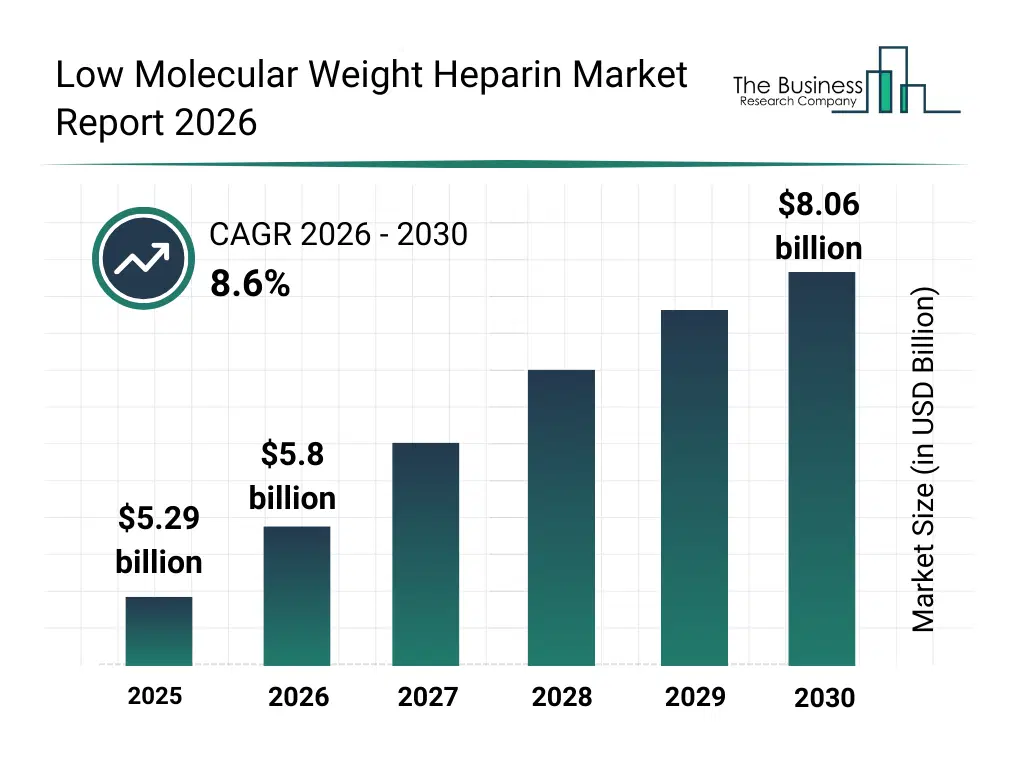
What Is The Low Molecular Weight Heparin Market Size and Share 2026?
The low molecular weight heparin market size has grown strongly in recent years. It will grow from $5.29 billion in 2025 to $5.8 billion in 2026 at a compound annual growth rate (CAGR) of 9.5%. The growth in the historic period can be attributed to increasing incidence of venous thromboembolism, limitations of unfractionated heparin therapy, widespread clinical acceptance of enoxaparin, growing use in postoperative care, improved safety profile of lmwh.What Is The Low Molecular Weight Heparin Market Growth Forecast?
The low molecular weight heparin market size is expected to see strong growth in the next few years. It will grow to $8.06 billion in 2030 at a compound annual growth rate (CAGR) of 8.6%. The growth in the forecast period can be attributed to rising aging population, increasing prevalence of cardiovascular disorders, growing demand for predictable anticoagulation therapy, expansion of home-based anticoagulant treatment, higher adoption in cancer-associated thrombosis. Major trends in the forecast period include growing preference for lmwh over unfractionated heparin, rising use in thromboprophylaxis and acute care settings, expansion of generic lmwh products, increasing adoption of prefilled syringes, growing use in outpatient and homecare treatment.Global Low Molecular Weight Heparin Market Segmentation
1) By Product Type: Enoxaparin, Dalteparin, Tinzaparin, Fraxiparine, Nadroparin, Other Product Types 2) By Packaging: Multi-Vials, Prefilled Syringes 3) By Application: Deep Vein Thrombosis, Acute Coronary Syndrome, Pulmonary Embolism, Other Applications Subsegments: 1) By Enoxaparin: Generic Enoxaparin, Branded Enoxaparin 2) By Dalteparin: Generic Dalteparin, Branded Dalteparin 3) By Tinzaparin: Generic Tinzaparin, Branded Tinzaparin 4) By Fraxiparine: Generic Fraxiparine, Branded Fraxiparine 5) By Nadroparin: Generic Nadroparin, Branded Nadroparin 6) By Other Product Types: Bemiparin, ParnaparinWhat Is The Driver Of The Low Molecular Weight Heparin Market?
The increased prevalence of blood disorders is expected to propel the growth of the low molecular weight heparin market going forward. Blood disorders refer to a range of conditions affecting the components of blood, such as red blood cells, white blood cells, platelets, and plasma proteins. These disorders, including anemia, hemophilia, and thrombosis, can impact individuals of all ages and demographics. The rise in blood disorders can be attributed to factors such as aging populations, improved diagnostic techniques, and increased exposure to environmental risk factors. Low molecular weight heparin is used in blood disorders to prevent and treat thromboembolism by inhibiting clot formation. For instance, in May 2024, according to the Leukemia Foundation, an Australia-based non-governmental organization, it is expected that 19,403 Australians will be newly diagnosed with blood cancer, such as leukemia, lymphoma, and myeloma, in 2022. This is equivalent to 53 people per day, or one person every 27 minutes. Currently, around 135,000 people in Australia are living with a blood cancer or blood disorder. By 2035, this number is projected to exceed 275,000. Therefore, the increased prevalence of blood disorders is driving the growth of the low molecular weight heparin industry.Key Players In The Global Low Molecular Weight Heparin Market
Major companies operating in the low molecular weight heparin market are Pfizer Inc., Sanofi SA, Boehringer Ingelheim International GmbH, Teva Pharmaceutical Industries Ltd., Eisai Co. Ltd., Sun Pharmaceutical Industries Ltd., Cipla Inc., Apotex Inc., Intas Pharmaceuticals Ltd, Hikma Pharmaceuticals PLC, Dr. Reddy's Laboratories Ltd., Lupin Limited, Alkem Laboratories Limited, Torrent Pharmaceuticals Ltd., Emcure Pharmaceuticals Limited, Biocon Limited, Aspen Pharmacare Holdings Limited, Ajanta Pharma Ltd., Zydus Pharmaceuticals, Gland Pharma Limited, Beximco Pharmaceuticals LtdGlobal Low Molecular Weight Heparin Market Trends and Insights
Major companies operating in the low molecular weight heparin market are focusing on innovative solutions, such as preservative-free prefilled syringes, to enhance safety and convenience for patients. Preservative-free prefilled syringes refer to those pre-filled with the anticoagulant medication enoxaparin, devoid of preservatives, ensuring safety and efficacy. It minimizes the potential for allergic reactions, providing a safer option for patients requiring anticoagulant therapy. For instance, in June 2023, Techdow USA Inc., a US-based generic injectable company, introduced 30 milligrams (mg), 40 mg, 60 mg, 80 mg, 100 mg, 120 mg, and 150 mg strengths of generic enoxaparin sodium (preservative-free) prefilled syringes for the prevention of thrombosis. Its generic version of Lovenox, a low-molecular-weight heparin used to prevent and treat blood clots, is expected to be more affordable, improve efficacy, increase accessibility, and enhance patient care.What Are Latest Mergers And Acquisitions In The Low Molecular Weight Heparin Market?
In November 2023, Vianex S.A., the Greece-based pharmaceutical manufacturer, partnered with LEO Pharma A/S to produce unfractionated heparin and low-molecular-weight heparin. With this partnership, Vianex will take over the production of standard unfractionated heparin and low-molecular-weight heparin (including raw-material management, vial filling, laboratory analyses and packaging) from LEO Pharma’s Ballerup facility to expand manufacturing capacity and secure supply across Europe, Asia, North Africa and the Middle East. LEO Pharma is a Denmark-based healthcare/pharmaceutical company that specializes in medical dermatology and thrombosis/coagulation therapies and will remain the marketing-authorization holder and distributor while outsourcing the injectable production to Vianex.Regional Outlook
North America was the largest region in the low molecular weight heparin market in 2025. Asia-Pacific is expected to be the fastest-growing region in the forecast period. The regions covered in this market report are Asia-Pacific, South East Asia, Western Europe, Eastern Europe, North America, South America, Middle East, Africa. The countries covered in this market report are Australia, Brazil, China, France, Germany, India, Indonesia, Japan, Taiwan, Russia, South Korea, UK, USA, Canada, Italy, Spain.What Defines the Low Molecular Weight Heparin Market?
The low molecular weight heparin market consists of sales of certoparin, reviparin, and bemiparin. Values in this market are ‘factory gate’ values, that is the value of goods sold by the manufacturers or creators of the goods, whether to other entities (including downstream manufacturers, wholesalers, distributors and retailers) or directly to end customers. The value of goods in this market includes related services sold by the creators of the goods.How is Market Value Defined and Measured?
The market value is defined as the revenues that enterprises gain from the sale of goods and/or services within the specified market and geography through sales, grants, or donations in terms of the currency (in USD unless otherwise specified). The revenues for a specified geography are consumption values that are revenues generated by organizations in the specified geography within the market, irrespective of where they are produced. It does not include revenues from resales along the supply chain, either further along the supply chain or as part of other products.What Key Data and Analysis Are Included in the Low Molecular Weight Heparin Market Report 2026?
The low molecular weight heparin market research report is one of a series of new reports from The Business Research Company that provides market statistics, including industry global market size, regional shares, competitors with the market share, detailed market segments, market trends and opportunities, and any further data you may need to thrive in the low molecular weight heparin industry. The market research report delivers a complete perspective of everything you need, with an in-depth analysis of the current and future state of the industry.Low Molecular Weight Heparin Market Report Forecast Analysis
| Report Attribute | Details |
|---|---|
| Market Size Value In 2026 | $5.8 billion |
| Revenue Forecast In 2035 | $8.06 billion |
| Growth Rate | CAGR of 9.5% from 2026 to 2035 |
| Base Year For Estimation | 2025 |
| Actual Estimates/Historical Data | 2020-2025 |
| Forecast Period | 2026 - 2030 - 2035 |
| Market Representation | Revenue in USD Billion and CAGR from 2026 to 2035 |
| Segments Covered | Product Type, Packaging, Application |
| Regional Scope | Asia-Pacific, Western Europe, Eastern Europe, North America, South America, Middle East, Africa |
| Country Scope | The countries covered in the report are Australia, Brazil, China, France, Germany, India, ... |
| Key Companies Profiled | Pfizer Inc., Sanofi SA, Boehringer Ingelheim International GmbH, Teva Pharmaceutical Industries Ltd., Eisai Co. Ltd., Sun Pharmaceutical Industries Ltd., Cipla Inc., Apotex Inc., Intas Pharmaceuticals Ltd, Hikma Pharmaceuticals PLC, Dr. Reddy's Laboratories Ltd., Lupin Limited, Alkem Laboratories Limited, Torrent Pharmaceuticals Ltd., Emcure Pharmaceuticals Limited, Biocon Limited, Aspen Pharmacare Holdings Limited, Ajanta Pharma Ltd., Zydus Pharmaceuticals, Gland Pharma Limited, Beximco Pharmaceuticals Ltd |
| Customization Scope | Request for Customization |
| Pricing And Purchase Options | Explore Purchase Options |
