
Mechanical Thrombectomy Devices Market Report 2026
Global Outlook – By Product Type (Stent Retrievers, Catheter-Based Thrombectomy Devices, Mechanical Aspiration Devices, Combined Devices), By Technology (Manual Mechanical Thrombectomy Devices, Automated Mechanical Thrombectomy Devices, Electromechanical Thrombectomy Devices, Hybrid Devices), By Application (Ischemic Stroke, Peripheral Artery Disease (PAD), Coronary Thrombosis, Post-Surgical Thrombosis), By Distribution Channel (Direct Sales, Distributors And Wholesalers, Online Sales, Retail Pharmacies), By End User (Hospitals, Ambulatory Surgical Centers, Specialized Invasive Care Centers, Research And Academic Institutions) – Market Size, Trends, Strategies, and Forecast to 2035
Mechanical Thrombectomy Devices Market Overview
• Mechanical Thrombectomy Devices market size has reached to $1.52 billion in 2025 • Expected to grow to $2.36 billion in 2030 at a compound annual growth rate (CAGR) of 9% • Growth Driver: Increasing Prevalence Of Stroke Is Fueling The Growth Of The Market Due To Aging Populations And Rising Stroke Mortality Rates • Market Trend: Advancements In Technology Improving Efficiency And Safety Of Mechanical Thrombectomy Procedures • North America was the largest region in 2025 and Asia-Pacific is the fastest growing region.What Is Covered Under Mechanical Thrombectomy Devices Market?
Mechanical thrombectomy devices are medical tools used to physically extract blockages from blood vessels through minimally invasive methods. They are built to move precisely within the vascular network, allowing for the safe capture and removal of obstructions while reducing potential damage to surrounding tissues. Their design incorporates features that enable controlled operation, effective grasping, and reliable retrieval of the material causing the blockage, helping to reestablish normal blood circulation in the vessel. The main product types of mechanical thrombectomy devices include stent retrievers, catheter-based thrombectomy devices, mechanical aspiration devices, and combined devices. Stent retrievers are medical devices used to remove blood clots from blocked arteries in the brain during stroke treatment by temporarily trapping and extracting the clot. It uses different technologies such as manual mechanical thrombectomy devices, automated mechanical thrombectomy devices, electromechanical thrombectomy devices, and hybrid devices. It is applied for ischemic stroke, peripheral artery disease (PAD), coronary thrombosis, and post-surgical thrombosis and is distributed through direct sales, distributors and wholesalers, online sales, and retail pharmacies. They are used by several end users, such as hospitals, ambulatory surgical centers, specialized invasive care centers, and research and academic institutions.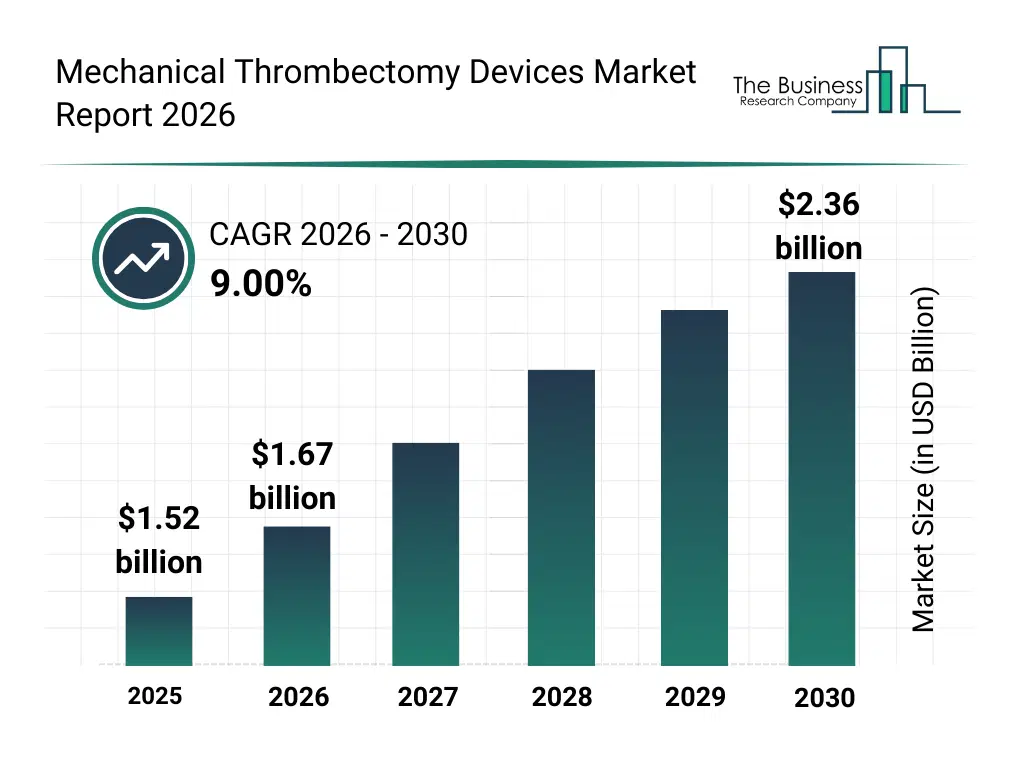
What Is The Mechanical Thrombectomy Devices Market Size and Share 2026?
The mechanical thrombectomy devices market size has grown strongly in recent years. It will grow from $1.52 billion in 2025 to $1.67 billion in 2026 at a compound annual growth rate (CAGR) of 9.9%. The growth in the historic period can be attributed to limited availability of mechanical thrombectomy devices, reliance on manual procedures for stroke treatment, growing awareness of stroke and cardiovascular risks, increasing hospital infrastructure for interventional procedures, adoption of catheter-based interventions.What Is The Mechanical Thrombectomy Devices Market Growth Forecast?
The mechanical thrombectomy devices market size is expected to see strong growth in the next few years. It will grow to $2.36 billion in 2030 at a compound annual growth rate (CAGR) of 9.0%. The growth in the forecast period can be attributed to development of automated and electromechanical thrombectomy devices, rising prevalence of ischemic stroke and coronary thrombosis, integration of ai-assisted device navigation, expansion of ambulatory surgical centers and specialized invasive care centers, increasing r&d investments in thrombectomy technologies. Major trends in the forecast period include rising adoption of minimally invasive procedures, increasing use of combined thrombectomy devices, growth in electromechanical and hybrid device segment, expansion of ischemic stroke treatment procedures, enhanced focus on device safety and regulatory compliance.Global Mechanical Thrombectomy Devices Market Segmentation
1) By Product Type: Stent Retrievers, Catheter-Based Thrombectomy Devices, Mechanical Aspiration Devices, Combined Devices 2) By Technology: Manual Mechanical Thrombectomy Devices, Automated Mechanical Thrombectomy Devices, Electromechanical Thrombectomy Devices, Hybrid Devices 3) By Application: Ischemic Stroke, Peripheral Artery Disease (PAD), Coronary Thrombosis, Post-Surgical Thrombosis 4) By Distribution Channel: Direct Sales, Distributors And Wholesalers, Online Sales, Retail Pharmacies 5) By End User: Hospitals, Ambulatory Surgical Centers, Specialized Invasive Care Centers, Research And Academic Institutions Subsegments: 1) By Stent Retrievers: Closed-Cell Stent Retrievers, Open-Cell Stent Retrievers, Hybrid Stent Retrievers 2) By Catheter-Based Thrombectomy Devices: Aspiration Catheters, Guide Catheters, Micro Catheters 3) By Mechanical Aspiration Devices: Manual Aspiration Devices, Powered Aspiration Devices 4) By Combined Devices: Aspiration Plus Stent Retrievers, Aspiration Plus Mechanical DevicesWhat Is The Driver Of The Mechanical Thrombectomy Devices Market?
The increasing prevalence of stroke is expected to propel the growth of the mechanical thrombectomy devices market going forward. Stroke is a medical condition that occurs when blood flow to a part of the brain is interrupted or reduced, causing brain cells to die due to lack of oxygen and nutrients. The increasing prevalence of stroke is due to aging populations, as older age significantly raises the risk of stroke due to natural changes in blood vessels and overall health. Mechanical thrombectomy devices are useful for stroke treatment by quickly removing blood clots from blocked arteries in the brain, thereby restoring blood flow, minimizing brain damage, and significantly improving patient recovery outcomes. For instance, in October 2023, according to the World Economic Forum, a Switzerland-based non-profit organization, by 2050, the number of stroke deaths could rise by 50%. Therefore, the increasing prevalence of stroke is driving growth in the mechanical thrombectomy devices industry.Key Players In The Global Mechanical Thrombectomy Devices Market
Major companies operating in the mechanical thrombectomy devices market are Stryker Corporation, Medtronic plc, Penumbra Inc., Terumo Corporation, Boston Scientific Corporation, Johnson & Johnson, Edwards Lifesciences Corporation, AngioDynamics Inc., Acandis GmbH & Co. KG, Rapid Medical, Vesalio, Control Medical Technology LLC, Perflow Medical, Sino Medical Sciences Technology Inc., Natec Medical Ltd., Straub Medical AG, Vascular Medcure Inc., Imperative Care Inc., NeuroVasc Technologies Inc., Inquis Medical Inc., Balt Extrusion S.A.S., Anaconda Biomed SL, SILK Road Medical Inc., Argon Medical Devices Inc., Minnetronix Inc.Global Mechanical Thrombectomy Devices Market Trends and Insights
Major companies operating in the mechanical thrombectomy devices market are focusing on developing innovative approaches, such as dual clot detection algorithms, to develop advanced systems that enhance clot removal efficiency and improve patient safety. Dual clot detection algorithms are advanced computer-aided methods that use both pressure and flow data to accurately identify and differentiate blood clots from normal blood flow during thrombectomy procedures. For instance, in January 2023, Penumbra Inc., a US-based healthcare company, launched the Lightning Flash, an advanced mechanical thrombectomy system designed to transform the way blood clots are removed from the body. This innovative device combines powerful aspiration technology with a highly engineered catheter design to efficiently and safely remove large blood clots, including venous thrombi and pulmonary emboli, helping physicians perform quicker procedures while minimizing potential complications and improving overall patient outcomes.What Are Latest Mergers And Acquisitions In The Mechanical Thrombectomy Devices Market?
In February 2025, Stryker Corporation, a US-based medical technology company, acquired Inari Medical Inc. for an undisclosed amount. With this acquisition, Stryker Corporation aims to significantly expand its interventional endovascular portfolio, especially in the high-growth venous thromboembolism (VTE) and thrombectomy segment. Inari Medical Inc. is a US-based medical device company that specializes in mechanical thrombectomy solutions, including the flowtriever system (for pulmonary embolism) and the clottriever system (for peripheral vessel clot removal).Regional Outlook
North America was the largest region in the mechanical thrombectomy devices market in 2025. Asia-Pacific is expected to be the fastest-growing region in the forecast period. The regions covered in this market report are Asia-Pacific, South East Asia, Western Europe, Eastern Europe, North America, South America, Middle East, Africa. The countries covered in this market report are Australia, Brazil, China, France, Germany, India, Indonesia, Japan, Taiwan, Russia, South Korea, UK, USA, Canada, Italy, SpainWhat Defines the Mechanical Thrombectomy Devices Market?
The mechanical thrombectomy devices market consists of sales of guidewires, microcatheters, balloon catheters, aspiration pumps, embolic protection devices, angioplasty balloons, contrast media, and hemostatic devices. Values in this market are ‘factory gate’ values, that is the value of goods sold by the manufacturers or creators of the goods, whether to other entities (including downstream manufacturers, wholesalers, distributors and retailers) or directly to end customers. The value of goods in this market includes related services sold by the creators of the goods.How is Market Value Defined and Measured?
The market value is defined as the revenues that enterprises gain from the sale of goods and/or services within the specified market and geography through sales, grants, or donations in terms of the currency (in USD unless otherwise specified). The revenues for a specified geography are consumption values that are revenues generated by organizations in the specified geography within the market, irrespective of where they are produced. It does not include revenues from resales along the supply chain, either further along the supply chain or as part of other products.Mechanical Thrombectomy Devices Market Report Forecast Analysis
| Report Attribute | Details |
|---|---|
| Market Size Value In 2026 | $1.67 billion |
| Revenue Forecast In 2035 | $2.36 billion |
| Growth Rate | CAGR of 9.9% from 2026 to 2035 |
| Base Year For Estimation | 2025 |
| Actual Estimates/Historical Data | 2020-2025 |
| Forecast Period | 2026 - 2030 - 2035 |
| Market Representation | Revenue in USD Billion and CAGR from 2026 to 2035 |
| Segments Covered | Product Type, Technology, Application, Distribution Channel, End User |
| Regional Scope | Asia-Pacific, Western Europe, Eastern Europe, North America, South America, Middle East, Africa |
| Country Scope | The countries covered in the report are Australia, Brazil, China, France, Germany, India, ... |
| Key Companies Profiled | Stryker Corporation, Medtronic plc, Penumbra Inc., Terumo Corporation, Boston Scientific Corporation, Johnson & Johnson, Edwards Lifesciences Corporation, AngioDynamics Inc., Acandis GmbH & Co. KG, Rapid Medical, Vesalio, Control Medical Technology LLC, Perflow Medical, Sino Medical Sciences Technology Inc., Natec Medical Ltd., Straub Medical AG, Vascular Medcure Inc., Imperative Care Inc., NeuroVasc Technologies Inc., Inquis Medical Inc., Balt Extrusion S.A.S., Anaconda Biomed SL, SILK Road Medical Inc., Argon Medical Devices Inc., Minnetronix Inc. |
| Customization Scope | Request for Customization |
| Pricing And Purchase Options | Explore Purchase Options |
