
Medical Device Contract Research Organization (CRO) Market Report 2026
Global Outlook – By Service (Project Management Or Clinical Supply Management, Regulatory Or Medical Affairs, Medical Writing, Clinical Monitoring, Quality Management Or Assurance, Bio Statistics, Investigator Payments, Laboratory, Patient And Site Recruitment, Other Services), By Device Type (MedTech Devices, Diagnostic Devices, Handheld Devices, Other Device Types), By Phase (Preclinical, Clinical), By Application (Cardiology, Diagnostic Imaging, Orthopedic, In Vitro Diagnostics (IVD), Ophthalmic, General And Plastic Surgery, Cardiovascular And Implantable Devices) – Market Size, Trends, Strategies, and Forecast to 2035
Medical Device Contract Research Organization (CRO) Market Overview
• Medical Device Contract Research Organization (CRO) market size has reached to $10.64 billion in 2025 • Expected to grow to $14.75 billion in 2030 at a compound annual growth rate (CAGR) of 6.7% • Growth Driver: Surge In Clinical Trials To Drive Growth In Contract Research Organization Market • Market Trend: Innovations In Medical Device CRO Market With Rise Of Hybrid And Virtual Trials • Asia-Pacific was the largest region in 2025 and Asia-Pacific is the fastest growing region.What Is Covered Under Medical Device Contract Research Organization (CRO) Market?
A medical device contract research organization (CRO) is a specialized service provider that offers outsourced research and development support to medical device manufacturers. These organizations provide a range of services to assist companies in navigating the complex process of bringing a medical device from concept to market, ensuring compliance with regulatory requirements, and achieving successful clinical outcomes. CROs are crucial for reducing the development time, costs, and risks associated with medical device innovations. The main types of services in medical device contract research organizations (CROs) include project management or clinical supply management, regulatory or medical affairs, medical writing, clinical monitoring, quality management or assurance, biostatistics, investigator payments, laboratory, patient and site recruitment, and others. Project management or clinical supply management involves overseeing the planning, execution, and coordination of clinical trials, ensuring that all necessary supplies, such as medical devices and drugs, are available at trial sites. The devices types include medtech devices, diagnostic devices, handheld devices, and others. The phase types include preclinical and clinical for applications such as cardiology, diagnostic imaging, orthopedics, in vitro diagnostics (IVD), ophthalmology, general and plastic surgery, and drug delivery.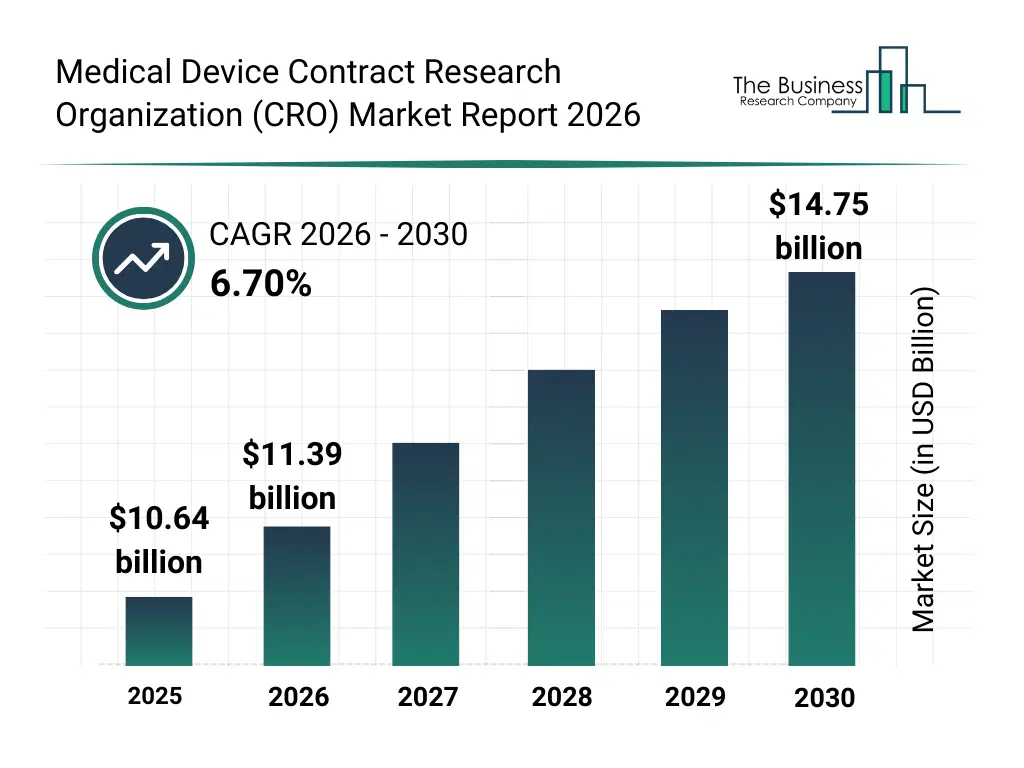
What Is The Medical Device Contract Research Organization (CRO) Market Size and Share 2026?
The medical device contract research organization (cro) market size has grown strongly in recent years. It will grow from $10.64 billion in 2025 to $11.39 billion in 2026 at a compound annual growth rate (CAGR) of 7.0%. The growth in the historic period can be attributed to increasing complexity of medical device regulations, growth in medical device innovation pipelines, rising clinical trial costs, limited in-house regulatory expertise, expansion of global clinical studies.What Is The Medical Device Contract Research Organization (CRO) Market Growth Forecast?
The medical device contract research organization (cro) market size is expected to see strong growth in the next few years. It will grow to $14.75 billion in 2030 at a compound annual growth rate (CAGR) of 6.7%. The growth in the forecast period can be attributed to increasing demand for accelerated device approvals, rising adoption of decentralized clinical trials, growing use of real-world evidence, expansion of post-market surveillance requirements, increasing integration of digital health technologies. Major trends in the forecast period include increasing outsourcing of clinical trial management, rising demand for regulatory compliance expertise, growing adoption of data-driven clinical research, expansion of remote and hybrid clinical monitoring models, enhanced focus on risk-based quality management.Global Medical Device Contract Research Organization (CRO) Market Segmentation
1) By Service: Project Management Or Clinical Supply Management, Regulatory Or Medical Affairs, Medical Writing, Clinical Monitoring, Quality Management Or Assurance, Bio Statistics, Investigator Payments, Laboratory, Patient And Site Recruitment, Other Services 2) By Device Type: MedTech Devices, Diagnostic Devices, Handheld Devices, Other Device Types 3) By Phase: Preclinical, Clinical 4) By Application: Cardiology, Diagnostic Imaging, Orthopedic, In Vitro Diagnostics (IVD), Ophthalmic, General And Plastic Surgery, Cardiovascular And Implantable Devices Subsegments: 1) By Project Management Or Clinical Supply Management: Study Planning and Coordination, Clinical Supply Chain Management, Vendor Management 2) By Regulatory or Medical Affairs: Regulatory Strategy Development, Medical Affairs Support, Regulatory Submission Preparation 3) By Medical Writing: Clinical Study Protocols, Clinical Study Reports (CSRs), Regulatory Submission Documents, Patient Information Leaflets 4) By Clinical Monitoring: Site Selection and Initiation, On-site Monitoring Visits, Remote Monitoring, Data Verification 5) By Quality Management or Assurance: Quality Assurance Audits, Standard Operating Procedure (SOP) Development, Compliance Management 6) By Biostatistics: Statistical Analysis Plan Development, Data Management And Analysis, Clinical Trial Design 7) By Investigator Payments: Payment Processing Services, Financial Disclosure Management 8) By Laboratory Services: Clinical Laboratory Testing, Bioanalytical Testing, Pathology Services 9) By Patient And Site Recruitment: Site Feasibility Assessment, Patient Recruitment Strategies, Retention And Engagement Strategies 10) By Other Services: Training And Education, Risk Management, Post-Market Surveillance, Data Management ServicesWhat Is The Driver Of The Medical Device Contract Research Organization (CRO) Market?
The increase in the number of clinical trials is expected to propel the growth of the medical device contract research organization (CRO) market going forward. Clinical trials are research studies conducted with human participants to evaluate the safety and effectiveness of new medical treatments, interventions, or diagnostic procedures. The increase in clinical trials is driven by the growing demand for innovative medical devices and regulatory requirements for rigorous testing. Medical device contract research organizations (CROs) provide specialized expertise and resources to manage, conduct, and oversee medical device clinical trials, ensuring compliance and efficient execution. For instance, in August 2024, according to the National Library of Medicine, a US-based national medical library, registered clinical trials increased from 477,227 in 2023 to 506,371 in all 50 states and across 221 countries. Therefore, the increase in clinical trials will drive the growth of the medical device contract research organization (CRO) industry.Key Players In The Global Medical Device Contract Research Organization (CRO) Market
Major companies operating in the medical device contract research organization (cro) market are Thermo Fisher Scientific Inc., IQVIA Inc., Laboratory Corporation of America Holdings, ICON plc, Eurofins Scientific SE, WuXi AppTec, Syneos Health, Charles River Laboratories, PAREXEL International, Medpace Holdings Inc., Hangzhou Tigermed Consulting Co. Ltd., Caidya, QPS Holdings LLC, Avania B.V., Veristat, Cromsource S.R.L., Osmunda Medical Technology Service Group, MedTrials Inc., Boston Biomedical Associates, Hangzhou CIRS Technology Group Co. Ltd., Clinlogix, Shanghai Haofeng Medical Technology Co. Ltd., Promedica International, Medidee ServicesGlobal Medical Device Contract Research Organization (CRO) Market Trends and Insights
Major companies operating in the medical device contract research organization (CRO) market are focusing on combining traditional services with advanced technology and recruitment strategies, such as hybrid and virtual trial models, to enhance trial efficiency, accelerate participant enrollment, and streamline regulatory compliance. Virtual and hybrid site capabilities accommodate diverse study designs and participant needs, especially in response to the growing demand for decentralized trials. For instance, in June 2024, Lindus Health, a UK-based clinical trials company, launched All-in-One Medical Device CRO, a comprehensive contract research organization (CRO) solution designed specifically for medical device trials. This innovative offering integrates traditional CRO services with advanced features such as their proprietary Citrus eClinical technology. It consolidates all essential trial tools into one platform, including clinical trial management systems (CTMS), electronic data capture (EDC), and eConsent. It enhances recruitment through its extensive database of over 30 million electronic health records (EHRs) and strategic digital marketing, ensuring efficient and timely enrollment. Additionally, Lindus Health provides robust site services, managing trials across single-site, multi-site, and virtual/hybrid models through solid relationships with principal investigators and research staff.What Are Latest Mergers And Acquisitions In The Medical Device Contract Research Organization (CRO) Market?
In October 2023, Avania, a US-based clinical research organization specializing in medical technology development acquiredHULL ASSOCIATES LLC and Ironstone Product Development (IPD) for an undisclosed amount. Through this acquisition, Avania aimed to expand its global capabilities in regulatory affairs, clinical trial management, and product development services. HULL ASSOCIATES LLC is a US-based consultancy specializing in regulatory strategies. Ironstone Product Development (IPD) is a Canada-based company focused on medical device development and commercialization strategies.Regional Outlook
Asia-Pacific was the largest region in the medical device contract research organization (CRO) market in 2025. Asia-Pacific is expected to be the fastest-growing region in the forecast period. The regions covered in this market report are Asia-Pacific, South East Asia, Western Europe, Eastern Europe, North America, South America, Middle East, Africa. The countries covered in this market report are Australia, Brazil, China, France, Germany, India, Indonesia, Japan, Taiwan, Russia, South Korea, UK, USA, Canada, Italy, SpainWhat Defines the Medical Device Contract Research Organization (CRO) Market?
The medical device contract research organization (CRO) market includes revenues earned by companies providing outsourced research and development services, such as clinical trials, regulatory affairs, and product development support, to medical device manufacturers. The market value includes the value of related goods sold by the service provider or included within the service offering. Only goods and services traded between entities or sold to end consumers are included.How is Market Value Defined and Measured?
The market value is defined as the revenues that enterprises gain from the sale of goods and/or services within the specified market and geography through sales, grants, or donations in terms of the currency (in USD unless otherwise specified). The revenues for a specified geography are consumption values that are revenues generated by organizations in the specified geography within the market, irrespective of where they are produced. It does not include revenues from resales along the supply chain, either further along the supply chain or as part of other products.Medical Device Contract Research Organization (CRO) Market Report Forecast Analysis
| Report Attribute | Details |
|---|---|
| Market Size Value In 2026 | $11.39 billion |
| Revenue Forecast In 2035 | $14.75 billion |
| Growth Rate | CAGR of 7.0% from 2026 to 2035 |
| Base Year For Estimation | 2025 |
| Actual Estimates/Historical Data | 2020-2025 |
| Forecast Period | 2026 - 2030 - 2035 |
| Market Representation | Revenue in USD Billion and CAGR from 2026 to 2035 |
| Segments Covered | Service, Device Type, Phase, Application |
| Regional Scope | Asia-Pacific, Western Europe, Eastern Europe, North America, South America, Middle East, Africa |
| Country Scope | The countries covered in the report are Australia, Brazil, China, France, Germany, India, ... |
| Key Companies Profiled | Thermo Fisher Scientific Inc., IQVIA Inc., Laboratory Corporation of America Holdings, ICON plc, Eurofins Scientific SE, WuXi AppTec, Syneos Health, Charles River Laboratories, PAREXEL International, Medpace Holdings Inc., Hangzhou Tigermed Consulting Co. Ltd., Caidya, QPS Holdings LLC, Avania B.V., Veristat, Cromsource S.R.L., Osmunda Medical Technology Service Group, MedTrials Inc., Boston Biomedical Associates, Hangzhou CIRS Technology Group Co. Ltd., Clinlogix, Shanghai Haofeng Medical Technology Co. Ltd., Promedica International, Medidee Services |
| Customization Scope | Request for Customization |
| Pricing And Purchase Options | Explore Purchase Options |
