
Medical Device Contract Research Organization Market Report 2026
Global Outlook – By Type (Finished Medical Devices, Medical Device Semi Finished Products, Medical Device Raw Materials), By Device Class (Class 1, Class 2, Class 3), By Phase (Preclinical, Clinical), By Application (Cardiology, Diagnostic Imaging, Orthopedic, In Vitro Diagnostic (IVD), Ophthalmic, General And Plastic Surgery, Drug Delivery) - Market Size, Trends, And Global Forecast 2026-2035
Medical Device Contract Research Organization Market Overview
• Medical Device Contract Research Organization market size has reached to $7.55 billion in 2025 • Expected to grow to $13.28 billion in 2030 at a compound annual growth rate (CAGR) of 11.9% • Growth Driver: Rising Clinical Trials Propel Growth Of Medical Device CRO Market • Market Trend: Innovative Solutions In Medical Device CRO Market Focus On Streamlining Clinical Trials • North America was the largest region in 2025 and Asia-Pacific is the fastest growing region.What Is Covered Under Medical Device Contract Research Organization Market?
A medical device contract research organization is a specialized service provider that conducts research and clinical trials on behalf of medical device companies. These organizations specialize in conducting clinical trials and other research-related activities required to develop, test, and validate medical devices. The main types of medical device contract research organizations are finished medical devices, semi-finished medical devices, and medical device raw materials. Finished medical devices refer to medical products that have completed the manufacturing process and are ready for use or distribution. These products can be classified into various types, including Class 1, Class 2, and Class 3, across different phases such as preclinical and clinical. They are applicable to a range of fields, including cardiology, diagnostic imaging, orthopedics, in vitro diagnostics (IVD), ophthalmology, general and plastic surgery, and drug delivery applications.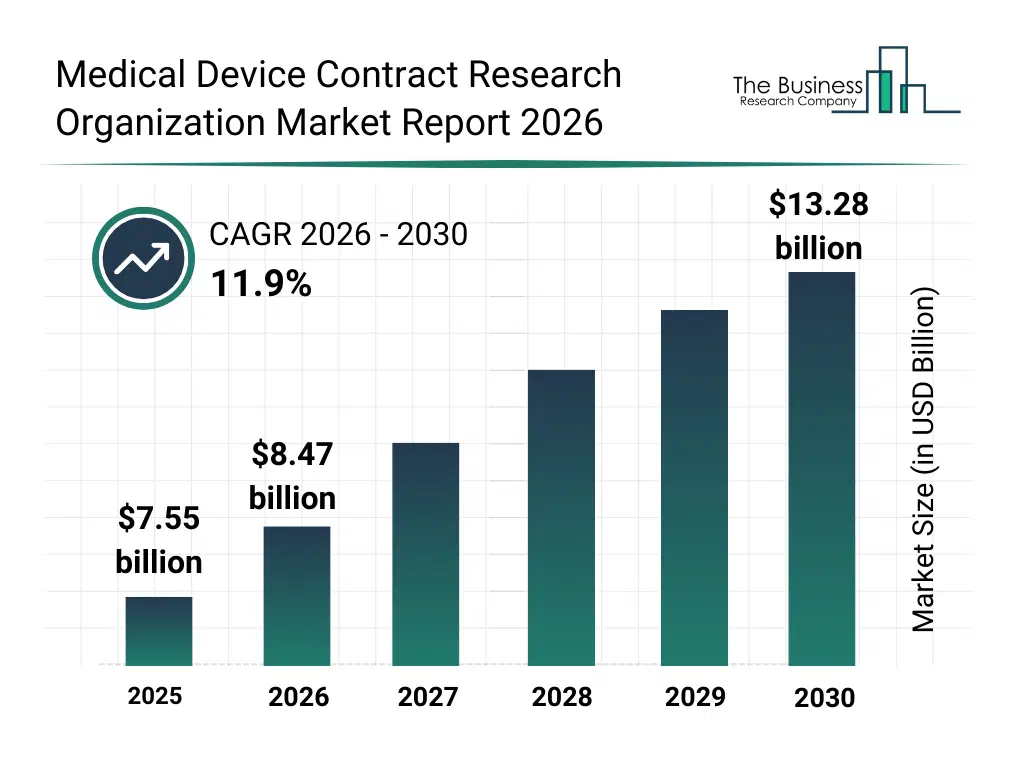
What Is The Medical Device Contract Research Organization Market Size and Share 2026?
The medical device contract research organization market size has grown rapidly in recent years. It will grow from $7.55 billion in 2025 to $8.47 billion in 2026 at a compound annual growth rate (CAGR) of 12.1%. The growth in the historic period can be attributed to growth in medical device innovation, rising clinical trial complexity, increasing regulatory requirements, early adoption of outsourced research services, expansion of global healthcare infrastructure.What Is The Medical Device Contract Research Organization Market Growth Forecast?
The medical device contract research organization market size is expected to see rapid growth in the next few years. It will grow to $13.28 billion in 2030 at a compound annual growth rate (CAGR) of 11.9%. The growth in the forecast period can be attributed to increase in class iii medical device development, growing demand for faster device approvals, rising investment in medical technology startups, expansion of clinical research in emerging markets, increasing need for post-market surveillance studies. Major trends in the forecast period include increasing outsourcing of medical device clinical trials, rising demand for regulatory and compliance expertise, growth in early-stage preclinical research services, expansion of cro support for high-risk class iii devices, increasing collaboration between device manufacturers and cros.Global Medical Device Contract Research Organization Market Segmentation
1) By Type: Finished Medical Devices, Medical Device Semi Finished Products, Medical Device Raw Materials 2) By Device Class: Class 1, Class 2, Class 3 3) By Phase: Preclinical, Clinical 4) By Application: Cardiology, Diagnostic Imaging, Orthopedic, In Vitro Diagnostic (IVD), Ophthalmic, General And Plastic Surgery, Drug Delivery Subsegments: 1) By Finished Medical Devices: Class I Medical Devices, Class II Medical Devices, Class III Medical Devices 2) By Medical Device Semi-Finished Products: Medical Device Components, Medical Device Subassemblies, Medical Device Kits 3) By Medical Device Raw Materials: Metals And Alloys, Polymers And Plastics, Ceramics And Composites, BiomaterialsWhat Is The Driver Of The Medical Device Contract Research Organization Market?
The increasing number of clinical trials is expected to propel the growth of the medical device contract research organization market going forward. Clinical trials are research studies conducted on human participants to evaluate medical treatments, aiming to assess their safety and effectiveness before widespread clinical use. The number of clinical trials is increasing due to advancements in medical technology, the rising prevalence of chronic diseases, and heightened R&D investment. Medical device contract research organizations facilitate clinical trials by providing specialized expertise, infrastructure, and regulatory knowledge to efficiently navigate research processes and accelerate trial timelines. For instance, in December 2024, according to the Association of the British Pharmaceutical Industry, a UK-based trade association, the organization reported that industry-sponsored clinical trials initiated in the UK increased from 411 in 2022 to 426 in 2023, with Phase III trials growing by 16.5%, while overall participant recruitment across commercial and non-commercial studies surged from 44,564 in 2022/23 to 150,191 in 2023/24. Therefore, the increasing number of clinical trials is driving the growth of the medical device contract research organization industry.Key Players In The Global Medical Device Contract Research Organization Market
Major companies operating in the medical device contract research organization market are Laboratory Corporation of America Holdings, ICON Public Limited Company, Eurofins Scientific Societas Europaea, Pharmaceutical Product Development LLC, WuXi AppTec Co. Ltd., Syneos Health Inc., Charles River Laboratories International Inc., Medpace Holdings Inc., Tigermed Consulting Co. Ltd., Novotech (Australia) Pty Ltd, NAMSA LLC, Promedica International, Excelya, Avania UK Ltd, OSMUNDA MEDICAL DEVICE SERVICE GROUP, CROMSOURCE, Musculoskeletal Clinical Regulatory Advisers LLC, BIO Agile Therapeutics Pvt Ltd, Vial, Qserve Group, CSSi LifeSciences, Medidee Services SA, Eclevar Medtech, Scandinavian CRO, Archer Research, Parexel International Corporation, Clinitude, Geneticist Inc.Global Medical Device Contract Research Organization Market Trends and Insights
Major companies operating in the medical device contract research organization market are focusing on developing innovative offerings, such as comprehensive contract research organization (CRO) services, to streamline regulatory compliance and enhance efficiency in clinical trials. Comprehensive CRO services encompass a wide range of support activities, including regulatory compliance and submissions, clinical trial design and management, patient recruitment and retention, data collection and analysis, monitoring and quality assurance, and post-market surveillance. For instance, in June 2024, Lindus Health Limited, a UK-based contract research organization, launched a new comprehensive all-in-one medical device CRO offering. This offering combines traditional CRO services with Lindus Health's expertise in medical device clinical trials, including regulatory pathways, participant recruitment, and virtual/hybrid trial capabilities. The all-in-one medical device CRO provides medical device companies with a wide range of features, including end-to-end CRO and study management, innovative recruitment tactics, study-specific eClinical software, medical device and regulatory expertise, and dedicated site services.What Are Latest Mergers And Acquisitions In The Medical Device Contract Research Organization Market?
In August 2024, Labquality Corporation, a Finland-based contract research organization (CRO) company, acquired Scandinavian CRO for an undisclosed amount. With this acquisition, Labquality strengthens its position as a full-service CRO, enabling further geographical expansion and growth in Scandinavia while enhancing its offerings for the health technology and pharmaceutical industries. Scandinavian CRO is a Sweden-based contract research organization company, that provides clinical research services.Regional Outlook
North America was the largest region in the medical device contract research organization market in 2025. Asia-Pacific is expected to be the fastest-growing region in the forecast period. The regions covered in this market report are Asia-Pacific, South East Asia, Western Europe, Eastern Europe, North America, South America, Middle East, Africa. The countries covered in this market report are Australia, Brazil, China, France, Germany, India, Indonesia, Japan, Taiwan, Russia, South Korea, UK, USA, Canada, Italy, Spain.What Defines the Medical Device Contract Research Organization Market?
The medical device contract research organization market includes revenues earned by entities by providing services such as clinical trial management, data collection and database management, preclinical research, medical writing, and project management. The market value includes the value of related goods sold by the service provider or included within the service offering. Only goods and services traded between entities or sold to end consumers are included.How is Market Value Defined and Measured?
The market value is defined as the revenues that enterprises gain from the sale of goods and/or services within the specified market and geography through sales, grants, or donations in terms of the currency (in USD unless otherwise specified). The revenues for a specified geography are consumption values that are revenues generated by organizations in the specified geography within the market, irrespective of where they are produced. It does not include revenues from resales along the supply chain, either further along the supply chain or as part of other products.What Key Data and Analysis Are Included in the Medical Device Contract Research Organization Market Report 2026?
The medical device contract research organization market research report is one of a series of new reports from The Business Research Company that provides market statistics, including industry global market size, regional shares, competitors with the market share, detailed market segments, market trends and opportunities, and any further data you may need to thrive in the medical device contract research organization industry. The market research report delivers a complete perspective of everything you need, with an in-depth analysis of the current and future state of the industry.Medical Device Contract Research Organization Market Report Forecast Analysis
| Report Attribute | Details |
|---|---|
| Market Size Value In 2026 | $8.47 billion |
| Revenue Forecast In 2035 | $13.28 billion |
| Growth Rate | CAGR of 12.1% from 2026 to 2035 |
| Base Year For Estimation | 2025 |
| Actual Estimates/Historical Data | 2020-2025 |
| Forecast Period | 2026 - 2030 - 2035 |
| Market Representation | Revenue in USD Billion and CAGR from 2026 to 2035 |
| Segments Covered | Type, Device Class, Phase, Application |
| Regional Scope | Asia-Pacific, Western Europe, Eastern Europe, North America, South America, Middle East, Africa |
| Country Scope | The countries covered in the report are Australia, Brazil, China, France, Germany, India, ... |
| Key Companies Profiled | Laboratory Corporation of America Holdings, ICON Public Limited Company, Eurofins Scientific Societas Europaea, Pharmaceutical Product Development LLC, WuXi AppTec Co. Ltd., Syneos Health Inc., Charles River Laboratories International Inc., Medpace Holdings Inc., Tigermed Consulting Co. Ltd., Novotech (Australia) Pty Ltd, NAMSA LLC, Promedica International, Excelya, Avania UK Ltd, OSMUNDA MEDICAL DEVICE SERVICE GROUP, CROMSOURCE, Musculoskeletal Clinical Regulatory Advisers LLC, BIO Agile Therapeutics Pvt Ltd, Vial, Qserve Group, CSSi LifeSciences, Medidee Services SA, Eclevar Medtech, Scandinavian CRO, Archer Research, Parexel International Corporation, Clinitude, Geneticist Inc. |
| Customization Scope | Request for Customization |
| Pricing And Purchase Options | Explore Purchase Options |
