
Medical Device Engineering Services Market Report 2026
Global Outlook – By Services (Designing & Engineering, Machining, Molding, Packaging), By Device Type (Imaging Devices, Therapeutic Devices, In-Vitro Diagnostic Devices, Diagnostic And Patient Monitoring Devices, Surgical Devices, Other Devices), By End-use (Medical Device Companies, Biotechnology Companies) – Market Size, Trends, Strategies, and Forecast to 2035
Medical Device Engineering Services Market Overview
• Medical Device Engineering Services market size has reached to $5.6 billion in 2025 • Expected to grow to $9.39 billion in 2030 at a compound annual growth rate (CAGR) of 10.7% • Growth Driver: Impact Of Diabetes Prevalence On Medical Device Engineering Services Market • Market Trend: Innovative Integrated Medical Device Services Enhance MedTech Product Lifecycle Efficiency • North America was the largest region in 2025 and Asia-Pacific is the fastest growing region.What Is Covered Under Medical Device Engineering Services Market?
Medical device engineering services refers to a specialized sector within the medical device industry that focuses on providing engineering and technical support throughout the lifecycle of medical devices. These services encompass a broad range of activities essential for the design, development, manufacturing, testing, regulatory compliance, and maintenance of medical devices. The main type of services in medical device engineering services are designing and engineering, machining, molding, packaging. Designing and engineering refer to the processes of conceptualizing, planning, creating, and optimizing products, systems, or solutions to meet specific requirements or objectives. The device type includes imaging devices, therapeutic devices, in-vitro diagnostic devices, diagnostic and patient monitoring devices, surgical devices, and others. The end users are medical device companies, and biotechnology companies.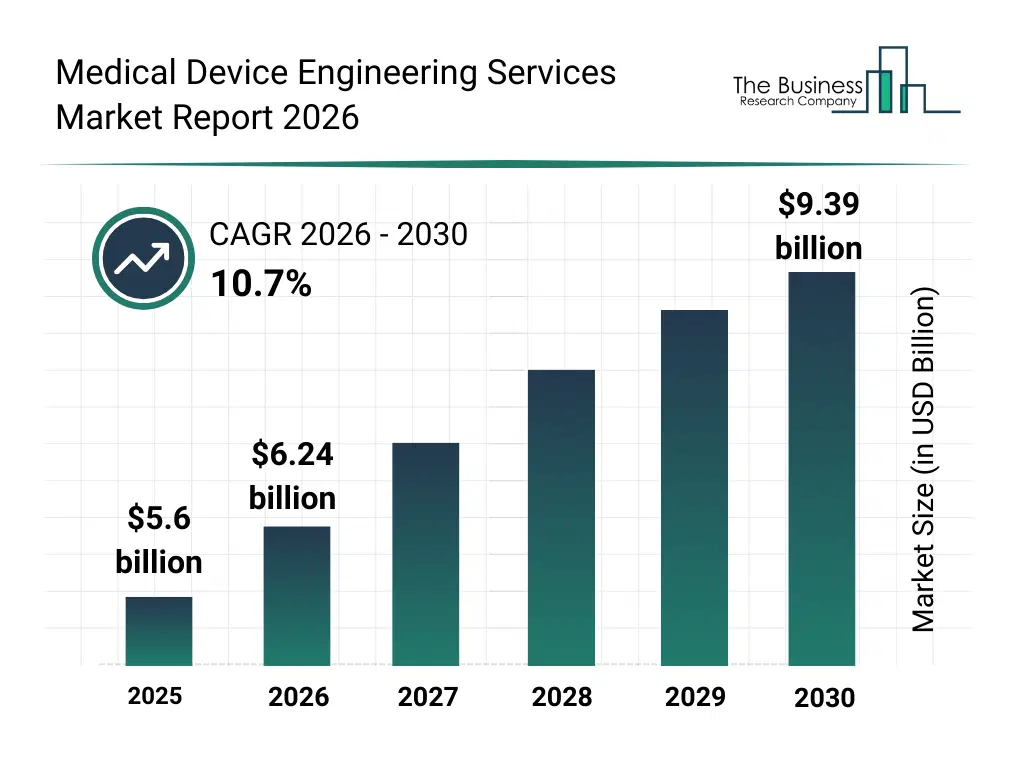
What Is The Medical Device Engineering Services Market Size and Share 2026?
The medical device engineering services market size has grown rapidly in recent years. It will grow from $5.6 billion in 2025 to $6.24 billion in 2026 at a compound annual growth rate (CAGR) of 11.5%. The growth in the historic period can be attributed to increasing medical device innovation cycles, growing complexity of regulatory frameworks, expansion of contract manufacturing adoption, rising demand for precision-engineered components, availability of specialized engineering expertise.What Is The Medical Device Engineering Services Market Growth Forecast?
The medical device engineering services market size is expected to see rapid growth in the next few years. It will grow to $9.39 billion in 2030 at a compound annual growth rate (CAGR) of 10.7%. The growth in the forecast period can be attributed to increasing demand for connected and smart medical devices, rising investments in digital health technologies, expansion of personalized and minimally invasive devices, growing outsourcing of regulatory and validation services, increasing focus on sustainable device manufacturing. Major trends in the forecast period include increasing demand for end-to-end device development services, rising adoption of digital engineering and simulation tools, growing focus on regulatory-compliant design processes, expansion of outsourced prototyping and testing services, enhanced integration of smart manufacturing techniques.Global Medical Device Engineering Services Market Segmentation
1) By Services: Designing & Engineering, Machining, Molding, Packaging 2) By Device Type: Imaging Devices, Therapeutic Devices, In-Vitro Diagnostic Devices, Diagnostic And Patient Monitoring Devices, Surgical Devices, Other Devices 3) By End-use: Medical Device Companies, Biotechnology Companies Subsegments: 1) By Designing & Engineering: Product Design, Prototyping, Testing And Validation, Regulatory Support 2) By Machining: CNC Machining, Laser Cutting, EDM (Electrical Discharge Machining) 3) By Injection Molding: Plastic Injection Molding, Rubber Molding, Metal Injection Molding, Compression Molding 4) By Packaging: Sterile Packaging, Custom Packaging Solutions, Labeling And Documentation, Shipping And Storage SolutionsWhat Is The Driver Of The Medical Device Engineering Services Market?
The growing patient population suffering from diabetes is expected to propel the growth of the medical device engineering services market going forward. Diabetes is a medical condition characterized by high blood glucose (sugar) levels resulting from defects in insulin production, insulin action, or both. The rising prevalence of diabetes is attributed to factors like sedentary lifestyles, aging populations, genetic predisposition, and dietary changes, driving an increase in the patient population. Medical device engineering services drive diabetes management by innovating glucose monitors, insulin delivery systems, artificial pancreas technology, wearables, and user-friendly designs. For instance, in February 2023, according to the Cleveland Clinic, a US-based non-profit academic medical center, globally, an estimated 537 million adults were diagnosed with diabetes, with projections suggesting an increase to 643 million by 2030 and 783 million by 2045. Therefore, the growing patient population suffering from diabetes is driving the growth of the medical device engineering services industry.Key Players In The Global Medical Device Engineering Services Market
Major companies operating in the medical device engineering services market are Johnson & Johnson, Accenture Plc, Abbott Laboratories, 3M Healthcare, Medtronic Plc, Tata Consultancy Services, Siemens Healthineers, Philips Healthcare, Becton Dickinson and Company, Stryker Corporation, GE Healthcare, Infosys Limited, Baxter International, Boston Scientific, HCL Technologies Limited, Wipro Limited, Zimmer Biomet, Tech Mahindra, Terumo Corporation, Intuitive Surgical, Edwards Lifesciences, Smith & Nephew, Hologic Inc., Varian Medical Systems, Nordson Corporation, L&T Technology Services Limited., Althea Group, Roche Diagnostics, Altran Technologies SA, ClariMed Inc.Global Medical Device Engineering Services Market Trends and Insights
Major companies operating in the medical device engineering services market focus on developing innovative technological solutions, such as integrated medical device services partners, to enhance their functionality, connectivity, and patient monitoring capabilities. Integrated medical device services are a comprehensive suite of services provided by specialized firms that support the entire lifecycle of a medical device, from initial concept through design, development, and post-market support, ensuring seamless coordination and efficiency across all stages. For instance, in January 2023, ClariMed, Inc., a US-based medical device development and regulatory services company, launched an integrated medical device services partner to address the growing need in the MedTech industry for an integrated approach to medical product development. This initiative aims to provide a human-centric approach to the MedTech product lifecycle by integrating best-of-breed service partners across product design, engineering, and development services. This model involves collaborating with top-tier service providers across various domains, such as product design, engineering, and development services, to streamline the product lifecycle and enhance the efficiency of bringing medical devices to market.What Are Latest Mergers And Acquisitions In The Medical Device Engineering Services Market?
In January 2024, Sanner Group, a Germany-based provider of scientific and device solutions for drug delivery, acquired Springboard Pro Ltd. for an undisclosed amount. Through this acquisition, Sanner aims to bolster its medical device design and development capabilities, aligning with its strategic goal to enhance services in drug delivery, diagnostics, and MedTech sectors and provide comprehensive support from concept design to commercial manufacturing in the healthcare industry. Springboard Pro Ltd. is a UK-based manufacturer of medical devices that offers medical device services.Regional Insights
North America was the largest region in the medical device engineering services market in 2025. Asia-Pacific is expected to be the fastest-growing region in the forecast period. The regions covered in this market report are Asia-Pacific, South East Asia, Western Europe, Eastern Europe, North America, South America, Middle East, Africa. The countries covered in this market report are Australia, Brazil, China, France, Germany, India, Indonesia, Japan, Taiwan, Russia, South Korea, UK, USA, Canada, Italy, Spain.What Defines the Medical Device Engineering Services Market?
The medical device engineering services market includes revenues earned by entities by providing services such as product design and development, prototyping and testing, verification and validation, human factors and usability engineering, and manufacturing support and process engineering. The market value includes the value of related goods sold by the service provider or included within the service offering. Only goods and services traded between entities or sold to end consumers are included.How is Market Value Defined and Measured?
The market value is defined as the revenues that enterprises gain from the sale of goods and/or services within the specified market and geography through sales, grants, or donations in terms of the currency (in USD unless otherwise specified). The revenues for a specified geography are consumption values that are revenues generated by organizations in the specified geography within the market, irrespective of where they are produced. It does not include revenues from resales along the supply chain, either further along the supply chain or as part of other products.What Key Data and Analysis Are Included in the Medical Device Engineering Services Market Report 2026?
The medical device engineering services market research report is one of a series of new reports from The Business Research Company that provides market statistics, including industry global market size, regional shares, competitors with the market share, detailed market segments, market trends and opportunities, and any further data you may need to thrive in the medical device engineering services industry. The market research report delivers a complete perspective of everything you need, with an in-depth analysis of the current and future state of the industry.Medical Device Engineering Services Market Report Forecast Analysis
| Report Attribute | Details |
|---|---|
| Market Size Value In 2026 | $6.24 billion |
| Revenue Forecast In 2035 | $9.39 billion |
| Growth Rate | CAGR of 11.5% from 2026 to 2035 |
| Base Year For Estimation | 2025 |
| Actual Estimates/Historical Data | 2020-2025 |
| Forecast Period | 2026 - 2030 - 2035 |
| Market Representation | Revenue in USD Billion and CAGR from 2026 to 2035 |
| Segments Covered | Services, Device Type, End-use |
| Regional Scope | Asia-Pacific, Western Europe, Eastern Europe, North America, South America, Middle East, Africa |
| Country Scope | The countries covered in the report are Australia, Brazil, China, France, Germany, India, ... |
| Key Companies Profiled | Johnson & Johnson, Accenture Plc, Abbott Laboratories, 3M Healthcare, Medtronic Plc, Tata Consultancy Services, Siemens Healthineers, Philips Healthcare, Becton Dickinson and Company, Stryker Corporation, GE Healthcare, Infosys Limited, Baxter International, Boston Scientific, HCL Technologies Limited, Wipro Limited, Zimmer Biomet, Tech Mahindra, Terumo Corporation, Intuitive Surgical, Edwards Lifesciences, Smith & Nephew, Hologic Inc., Varian Medical Systems, Nordson Corporation, L&T Technology Services Limited., Althea Group, Roche Diagnostics, Altran Technologies SA, ClariMed Inc. |
| Customization Scope | Request for Customization |
| Pricing And Purchase Options | Explore Purchase Options |
