
Medical Device Testing Market Report 2026
Global Outlook – By Testing Type (Physical Testing, Chemical Or Biological Testing, Cybersecurity Testing, Microbiology And Sterility Testing, Other Testing Type), By Device Class (Class I, Class II, Class III), By Sourcing Type (In-House, Outsourced), By Service Type (Testing Services, Inspection Services, Certification Services), By End-User (Hospital, Research Center, Homecare Clinics, Other End-User) - Market Size, Trends, And Global Forecast 2026-2035
Medical Device Testing Market Overview
• Medical Device Testing market size has reached to $10.46 billion in 2025 • Expected to grow to $14.23 billion in 2030 at a compound annual growth rate (CAGR) of 6.4% • Growth Driver: The Surge In Medical Device Testing Amidst Rising Recalls • Market Trend: Innovation In Third-Party Validation Enhances Medical Device Testing Capabilities • North America was the largest region in 2025 and Asia-Pacific is the fastest growing region.What Is Covered Under Medical Device Testing Market?
Medical device testing refers to the process of evaluating and verifying the safety, effectiveness, and quality of medical devices. It is used to ensure that products are safe and effective for use in patient care and that they meet the standards and requirements set by regulatory agencies. The main types of medical device testing are physical testing, chemical or biological testing, cybersecurity testing, microbiology and sterility testing, and others. Physical testing involves subjecting medical devices to a series of physical tests to evaluate their performance, durability, and safety, which is used to help manufacturers ensure that their products are safe, effective, and reliable. The device classes are class I, class II, and class III are sourced in-house, and outsourced, and offers services such as testing services, inspection services, and certification services. It is used by various end-users such as hospital, research center, homecare clinics, and others.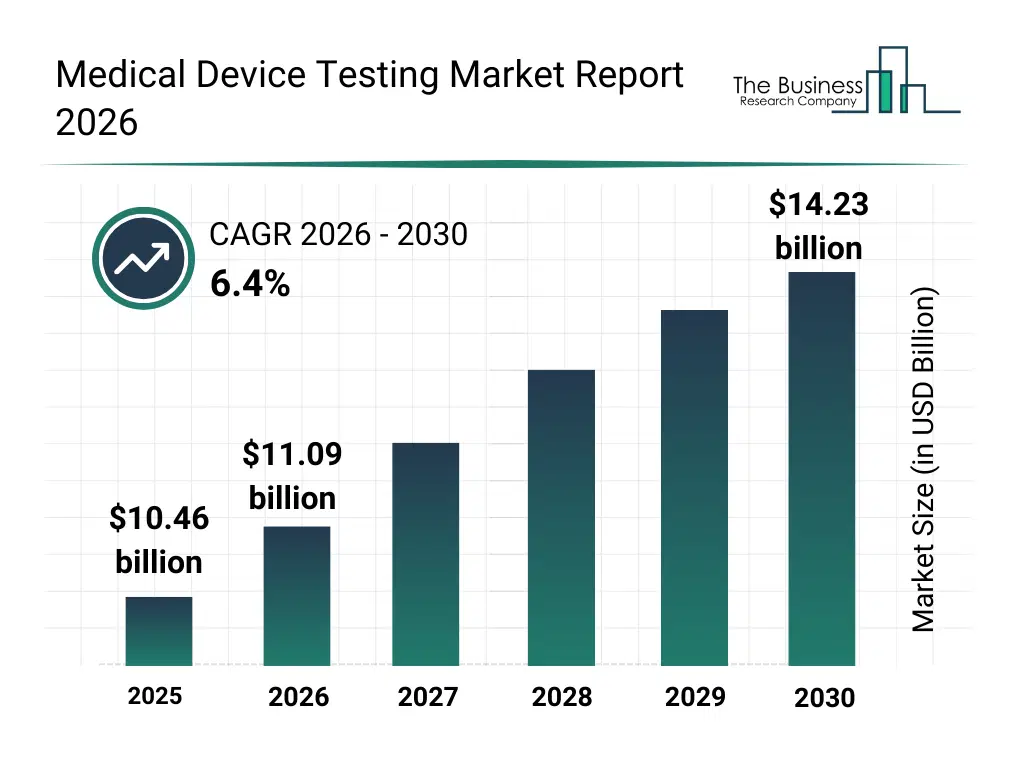
What Is The Medical Device Testing Market Size and Share 2026?
The medical device testing market size has grown strongly in recent years. It will grow from $10.46 billion in 2025 to $11.09 billion in 2026 at a compound annual growth rate (CAGR) of 6.1%. The growth in the historic period can be attributed to growth in medical device approvals, increasing regulatory scrutiny, rising adoption of implantable devices, early globalization of medical device manufacturing, demand for standardized quality assurance.What Is The Medical Device Testing Market Growth Forecast?
The medical device testing market size is expected to see strong growth in the next few years. It will grow to $14.23 billion in 2030 at a compound annual growth rate (CAGR) of 6.4%. The growth in the forecast period can be attributed to increase in connected medical devices, rising complexity of class III devices, growing focus on patient safety outcomes, expansion of outsourced testing services, evolving global regulatory frameworks. Major trends in the forecast period include rising demand for comprehensive regulatory compliance testing, growing emphasis on device safety and risk mitigation, expansion of outsourced testing and certification services, increasing focus on cybersecurity and software validation testing, higher adoption of advanced testing for high-risk medical devices.Global Medical Device Testing Market Segmentation
1) By Testing Type: Physical Testing, Chemical Or Biological Testing, Cybersecurity Testing, Microbiology And Sterility Testing, Other Testing Type 2) By Device Class: Class I, Class II, Class III 3) By Sourcing Type: In-House, Outsourced 4) By Service Type: Testing Services, Inspection Services, Certification Services 5) By End-User: Hospital, Research Center, Homecare Clinics, Other End-User Subsegments: 1) By Physical Testing: Mechanical Testing, Environmental Testing, Electrical Testing 2) By Chemical Or Biological Testing: Chemical Composition Analysis, Biocompatibility Testing, Toxicological Assessment 3) By Cybersecurity Testing: Vulnerability Assessment, Penetration Testing, Risk Assessment 4) By Microbiology And Sterility Testing: Microbial Contamination Testing, Sterility Assurance Testing, Endotoxin Testing 5) By Other Testing Types: Performance Testing, Usability Testing, Regulatory Compliance TestingWhat Is The Driver Of The Medical Device Testing Market?
A rise in medical device recalls is driving the medical device testing market forward. A device recall is a process by which the manufacturer or distributor of the device takes corrective action to remove or correct the problem in the medical device. Medical device testing is used by medical device manufacturers in identifying the potential safety issues associate with devices and can help determine whether a medical recall is necessary to ensure patient safety and the effectiveness of devices and helps in improving the quality of the product to avoid medical recalls. For instance, in March 2024, according to Sedgwick, a UK-based risk management and brand protection company, in 2023, there were 3,306 medical device recalls in Europe (EU + UK), representing a 20% increase over 2022. Therefore, the increasing number of medical device recalls is driving the medical device testing industry.Key Players In The Global Medical Device Testing Market
Major companies operating in the medical device testing market are Intertek Group plc., SGS SA, Bureau Veritas Group, TUV SUD AG, TUV Rheinland Group, Pace Analytical Services LLC., Charles River Laboratories Inc., UL Solutions, North American Science Associates LLC, Medistri SA, WuXi AppTec Inc., Cordant Health Solutions, GE Healthcare Technologies Inc., Thermo Fisher Scientific Inc., Biomedical Device Labs, NSF International, Analytical Lab Group LLC, Boston Analytical, DDL Inc., Element Materials Technology, Empirical Testing Corp., Eurofins Scientific, Gateway Analytical LLC, iuvo BioScience, Laboratory Corporation of America Holdings, Microtest Laboratories Inc., Nelson Laboratories LLC, National Technical Systems Inc., Pacific Biolabs Inc., Sterigenics International LLC, Toxikon Corporation, WCG Imaging, Westpak Inc.Global Medical Device Testing Market Trends and Insights
Major companies operating in the medical device testing market are focusing on innovation in testing and verification, such as advanced third-party validation to ensure device safety, usability, and regulatory compliance. Advanced third-party validation refers to independent testing conducted by specialized laboratories to confirm that devices meet performance and regulatory standards. For instance, in October 2023, UL Solutions, a US-based company providing medical testing, inspection, and certification services, launched expanded medical device testing capabilities at its Rochester Hills laboratory. This innovation includes verification and validation, usability testing, cybersecurity assessments, and environmental stress testing, helping manufacturers improve device safety, reliability, and regulatory compliance.What Are Latest Mergers And Acquisitions In The Medical Device Testing Market?
In January 2025, NAMSA Inc., a US-based medical research and testing company, acquired the U.S. medical-device testing operations of WuXi AppTec for an undisclosed amount. With this acquisition, NAMSA aims to strengthen its MedTech testing and clinical research capabilities by integrating WuXi AppTec’s U.S. testing operations to expand laboratory capacity, enhance service offerings, and accelerate time-to-market for medical-device clients. WuXi AppTec Co., Ltd. is a China-based life-sciences company that specializes in providing contract research, testing, and manufacturing services for pharmaceutical, biotechnology, and medical-device companies.Regional Outlook
North America was the largest region in the medical device testing market in 2025. Asia-Pacific is expected to be the fastest-growing region in the medical testing device market report during the forecast period. The regions covered in this market report are Asia-Pacific, South East Asia, Western Europe, Eastern Europe, North America, South America, Middle East, Africa. The countries covered in this market report are Australia, Brazil, China, France, Germany, India, Indonesia, Japan, Taiwan, Russia, South Korea, UK, USA, Canada, Italy, Spain.What Defines the Medical Device Testing Market?
The medical devices testing market includes revenues earned by entities by providing services such as biocompatibility testing services, bio-burden determination services, and quality assurance testing services. The market value includes the value of related goods sold by the service provider or included within the service offering. Only goods and services traded between entities or sold to end consumers are included.How is Market Value Defined and Measured?
The market value is defined as the revenues that enterprises gain from the sale of goods and/or services within the specified market and geography through sales, grants, or donations in terms of the currency (in USD unless otherwise specified). The revenues for a specified geography are consumption values that are revenues generated by organizations in the specified geography within the market, irrespective of where they are produced. It does not include revenues from resales along the supply chain, either further along the supply chain or as part of other products.What Key Data and Analysis Are Included in the Medical Device Testing Market Report 2026?
The medical device testing market research report is one of a series of new reports from The Business Research Company that provides market statistics, including industry global market size, regional shares, competitors with the market share, detailed market segments, market trends and opportunities, and any further data you may need to thrive in the medical device testing industry. The market research report delivers a complete perspective of everything you need, with an in-depth analysis of the current and future state of the industry.Medical Device Testing Market Report Forecast Analysis
| Report Attribute | Details |
|---|---|
| Market Size Value In 2026 | $11.09 billion |
| Revenue Forecast In 2035 | $14.23 billion |
| Growth Rate | CAGR of 6.1% from 2026 to 2035 |
| Base Year For Estimation | 2025 |
| Actual Estimates/Historical Data | 2020-2025 |
| Forecast Period | 2026 - 2030 - 2035 |
| Market Representation | Revenue in USD Billion and CAGR from 2026 to 2035 |
| Segments Covered | Testing Type, Device Class, Sourcing Type, Service Type, End-User |
| Regional Scope | Asia-Pacific, Western Europe, Eastern Europe, North America, South America, Middle East, Africa |
| Country Scope | The countries covered in the report are Australia, Brazil, China, France, Germany, India, ... |
| Key Companies Profiled | Intertek Group plc., SGS SA, Bureau Veritas Group, TUV SUD AG, TUV Rheinland Group, Pace Analytical Services LLC., Charles River Laboratories Inc., UL Solutions, North American Science Associates LLC, Medistri SA, WuXi AppTec Inc., Cordant Health Solutions, GE Healthcare Technologies Inc., Thermo Fisher Scientific Inc., Biomedical Device Labs, NSF International, Analytical Lab Group LLC, Boston Analytical, DDL Inc., Element Materials Technology, Empirical Testing Corp., Eurofins Scientific, Gateway Analytical LLC, iuvo BioScience, Laboratory Corporation of America Holdings, Microtest Laboratories Inc., Nelson Laboratories LLC, National Technical Systems Inc., Pacific Biolabs Inc., Sterigenics International LLC, Toxikon Corporation, WCG Imaging, Westpak Inc. |
| Customization Scope | Request for Customization |
| Pricing And Purchase Options | Explore Purchase Options |
