
Medical Device Testing Services Market Report 2026
Global Outlook – By Product (Active Implant Medical Device, Active Medical Device, Non-Active Medical Device, In-Vitro Diagnostics Medical Device, Ophthalmic Medical Device, Orthopedic And Dental Medical Device, Vascular Medical Device, Other Products), By Service (Biocompatibility Tests, Chemistry Test, Microbiology And Sterility Test, Package Validation), By Phase (Preclinical, Clinical) - Market Size, Trends, And Global Forecast 2026-2035
Medical Device Testing Services Market Overview
• Medical Device Testing Services market size has reached to $10.18 billion in 2025 • Expected to grow to $15.31 billion in 2030 at a compound annual growth rate (CAGR) of 8.5% • Growth Driver: Role Of Chronic Diseases In Boosting The Medical Device Testing Services Market • Market Trend: Technological Advancements Drive Expansion of Medical Device Testing Services Market • Asia-Pacific was the largest region in 2025 and North America is the fastest growing region.What Is Covered Under Medical Device Testing Services Market?
Medical device testing services refer to the processes and procedures conducted to evaluate the safety, efficacy, reliability, and quality of medical devices. These services are crucial in ensuring that medical devices meet regulatory standards and requirements before they can be marketed and used for clinical purposes. The main types of products in medical device testing services are active implant medical devices, active medical devices, non-active medical devices, in-vitro diagnostic medical devices, ophthalmic medical devices, orthopedic and dental medical devices, vascular medical devices, and others. An active implantable medical device (AIMD) is a type of medical device that is designed to be implanted into the body to provide therapy or monitoring. It provides various services, such as biocompatibility tests, chemistry tests, microbiology and sterility tests, and package validation and it included both preclinical and clinical phases.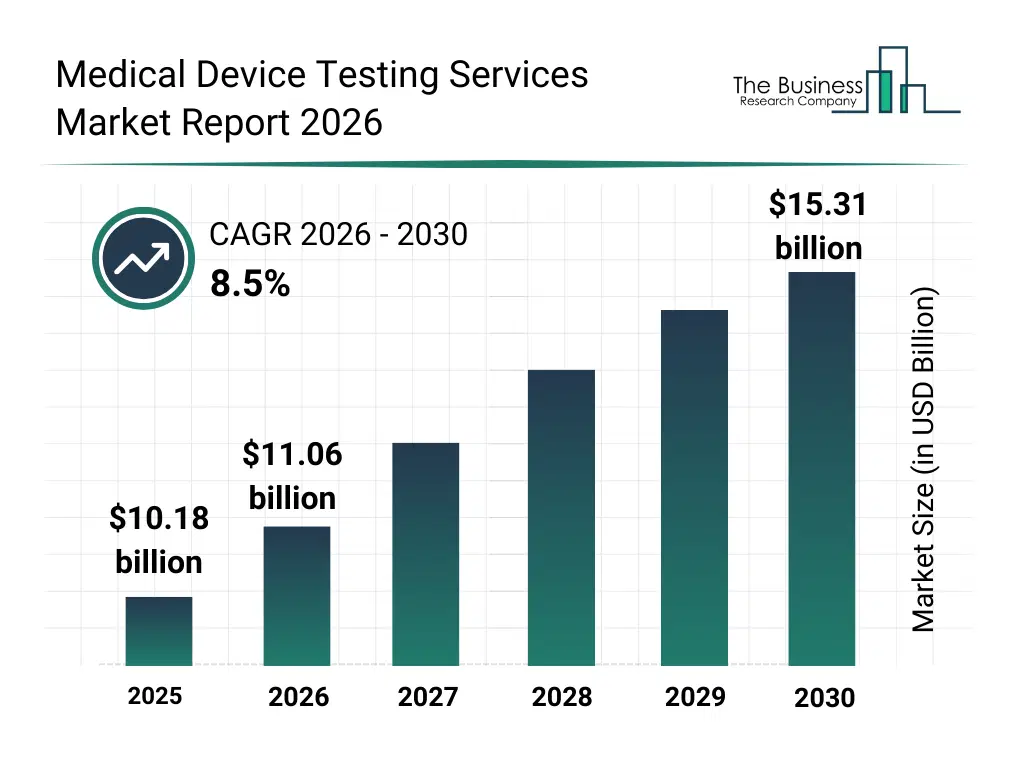
What Is The Medical Device Testing Services Market Size and Share 2026?
The medical device testing services market size has grown strongly in recent years. It will grow from $10.18 billion in 2025 to $11.06 billion in 2026 at a compound annual growth rate (CAGR) of 8.7%. The growth in the historic period can be attributed to increase in medical device approvals, tightening global regulatory standards, growth of implantable and active devices, expansion of clinical research activities, demand for standardized quality testing.What Is The Medical Device Testing Services Market Growth Forecast?
The medical device testing services market size is expected to see strong growth in the next few years. It will grow to $15.31 billion in 2030 at a compound annual growth rate (CAGR) of 8.5%. The growth in the forecast period can be attributed to rising complexity of combination devices, growth in in-vitro diagnostics demand, increasing focus on patient safety assurance, expansion of global clinical trials, evolution of harmonized regulatory frameworks. Major trends in the forecast period include rising demand for end-to-end regulatory testing services, increasing focus on biocompatibility and sterility validation, growth of outsourced preclinical and clinical testing services, higher testing requirements for active and implantable devices, expansion of comprehensive package and chemistry testing.Global Medical Device Testing Services Market Segmentation
1) By Product: Active Implant Medical Device, Active Medical Device, Non-Active Medical Device, In-Vitro Diagnostics Medical Device, Ophthalmic Medical Device, Orthopedic And Dental Medical Device, Vascular Medical Device, Other Products 2) By Service: Biocompatibility Tests, Chemistry Test, Microbiology And Sterility Test, Package Validation 3) By Phase: Preclinical, Clinical Subsegments: 1) By Active Implant Medical Device: Pacemakers, Implantable Defibrillators, Neurostimulators 2) By Active Medical Device: Infusion Pumps, Diagnostic Equipment, Therapeutic Equipment 3) By Non-Active Medical Device: Surgical Instruments, Patient Monitoring Equipment, Bandages And Dressings 4) By In-Vitro Diagnostics Medical Device: Blood Test Kits, Urine Test Strips, Molecular Diagnostic Devices 5) By Ophthalmic Medical Device: Contact Lenses, Surgical Instruments For Eye Surgery, Diagnostic Equipment For Eye Care 6) By Orthopedic And Dental Medical Device: Joint Replacement Implants, Orthotic Devices, Dental Crowns And Bridges 7) By Vascular Medical Device: Stents, Catheters, Vascular Grafts 8) By Other Products: Miscellaneous Medical Devices, Custom Medical Devices, Combination ProductsWhat Is The Driver Of The Medical Device Testing Services Market?
The rising prevalence of chronic diseases is expected to propel the growth of the medical device testing services market going forward. Chronic diseases refer to long-term health conditions that persist over an extended period, typically lasting for a year or more. This disease arises due to an aging population, unhealthy lifestyle choices, and environmental factors. Medical device testing services are used in the management of chronic diseases to support remote patient monitoring (RPM) and real-time detection of health changes, and these services ensures the safety, efficacy, reliability, and quality of devices, ultimately improving patient outcomes and quality of life. For instance, in February 2024, according to the Centers for Disease Control and Prevention (CDC), a US-based federal public health agency, the agency reported that an estimated 129 million people in the United States have at least one major chronic disease, including conditions such as heart disease, cancer, diabetes, obesity, and hypertension. Therefore, the rising prevalence of chronic diseases is driving the growth of the medical device testing services industry.Key Players In The Global Medical Device Testing Services Market
Major companies operating in the medical device testing services market are Laboratory Corporation of America Holdings, Eurofins Scientific SE, SGS SA, Bureau Veritas SA, WuXi AppTec Group, Steris plc, Sartorius AG, Charles River Laboratories International Inc., Altran Corporation, TUV SUD AG, Element Minnetonka, Underwriters Laboratories LLC, Intertek Group Plc., QTS Analytical Limited, Pace Analytical Services LLC, Sterigenics International LLC, Nelson Laboratories LLC, Exova Group Limited, Mérieux NutriSciences Corporation, American Preclinical Services LLC, Toxikon Corporation, North American Science Associates Inc., Avomeen Analytical Services LLC, Element Materials Technology Group Limited, Sionic Advisors LLC, Medical Engineering Technologies Ltd.Global Medical Device Testing Services Market Trends and Insights
Major companies operating in the medical device testing services market are concentrating on the adoption of technological advancements, such as new medical device testing services, to provide comprehensive testing solutions. These services refer to a range of tests conducted by specialized companies to ensure the effectiveness, safety, and compliance of medical devices with national and international regulations. For instance, in October 2023, UL Solutions, a US-based provider of medical testing, inspection, and certification services, launched new medical device testing services at its laboratory in Rochester Hills. These services include medical device makers' compliance training, usability testing, and third-party verification and validation. It contributes to the state's developing medical device sector and helps manufacturers improve the safety, security, usability, and interoperability of their devices. Additionally, it also provides third-party verification and validation, cybersecurity services, auditing, usability testing, and compliance training to medical device manufacturers, as well as addressing potential risks such as quality, safety, and cybersecurity issues, all while maintaining the pace of innovation and protecting users.What Are Latest Mergers And Acquisitions In The Medical Device Testing Services Market?
In January 2025, Applus Laboratories, a Spain-based testing, inspection, and certification company, acquired DE Testing Co., Ltd. for an undisclosed amount. With this acquisition, Applus Laboratories aims to expand its testing and inspection capabilities in the Asia-Pacific region, strengthen its local market presence, and enhance service offerings for industrial and consumer products. DE Testing Co., Ltd. is a China-based technology company that specializes in providing laboratory testing, quality assurance, and certification services across multiple industries.Regional Outlook
Asia-Pacific was the largest region in the medical device testing services market in 2025. North America is expected to be the fastest-growing region in the forecast period. The regions covered in this market report are Asia-Pacific, South East Asia, Western Europe, Eastern Europe, North America, South America, Middle East, Africa. The countries covered in this market report are Australia, Brazil, China, France, Germany, India, Indonesia, Japan, Taiwan, Russia, South Korea, UK, USA, Canada, Italy, Spain.What Defines the Medical Device Testing Services Market?
The medical device testing services market includes revenues earned by entities through services such as sterility testing, packaging testing, electrical safety testing, performance testing, usability testing, and post-market surveillance testing services. The market value includes the value of related goods sold by the service provider or included within the service offering. The medical device testing services market also includes sales of testing instruments and equipment, consumables and reagents, mechanical testing machines, electrical safety testers and sterilizers, and autoclaves. Values in this market are ‘factory gate’ values, that is, the value of goods sold by the manufacturers or creators of the goods, whether to other entities (including downstream manufacturers, wholesalers, distributors, and retailers) or directly to end customers. The value of goods in this market includes related services sold by the creators of the goods.How is Market Value Defined and Measured?
The market value is defined as the revenues that enterprises gain from the sale of goods and/or services within the specified market and geography through sales, grants, or donations in terms of the currency (in USD unless otherwise specified). The revenues for a specified geography are consumption values that are revenues generated by organizations in the specified geography within the market, irrespective of where they are produced. It does not include revenues from resales along the supply chain, either further along the supply chain or as part of other products.What Key Data and Analysis Are Included in the Medical Device Testing Services Market Report 2026?
The medical device testing services market research report is one of a series of new reports from The Business Research Company that provides market statistics, including industry global market size, regional shares, competitors with the market share, detailed market segments, market trends and opportunities, and any further data you may need to thrive in the medical device testing services industry. The market research report delivers a complete perspective of everything you need, with an in-depth analysis of the current and future state of the industry.Medical Device Testing Services Market Report Forecast Analysis
| Report Attribute | Details |
|---|---|
| Market Size Value In 2026 | $11.06 billion |
| Revenue Forecast In 2035 | $15.31 billion |
| Growth Rate | CAGR of 8.7% from 2026 to 2035 |
| Base Year For Estimation | 2025 |
| Actual Estimates/Historical Data | 2020-2025 |
| Forecast Period | 2026 - 2030 - 2035 |
| Market Representation | Revenue in USD Billion and CAGR from 2026 to 2035 |
| Segments Covered | Product, Service, Phase |
| Regional Scope | Asia-Pacific, Western Europe, Eastern Europe, North America, South America, Middle East, Africa |
| Country Scope | The countries covered in the report are Australia, Brazil, China, France, Germany, India, ... |
| Key Companies Profiled | Laboratory Corporation of America Holdings, Eurofins Scientific SE, SGS SA, Bureau Veritas SA, WuXi AppTec Group, Steris plc, Sartorius AG, Charles River Laboratories International Inc., Altran Corporation, TUV SUD AG, Element Minnetonka, Underwriters Laboratories LLC, Intertek Group Plc., QTS Analytical Limited, Pace Analytical Services LLC, Sterigenics International LLC, Nelson Laboratories LLC, Exova Group Limited, Mérieux NutriSciences Corporation, American Preclinical Services LLC, Toxikon Corporation, North American Science Associates Inc., Avomeen Analytical Services LLC, Element Materials Technology Group Limited, Sionic Advisors LLC, Medical Engineering Technologies Ltd. |
| Customization Scope | Request for Customization |
| Pricing And Purchase Options | Explore Purchase Options |
