
Medical Devices Vigilance Market Report 2026
Global Outlook – By Delivery Mode (On-Demand, On-Premise), By Application (Therapeutics, Diagnostics, Surgical, Research, Other Applications), By End-User (Original Equipment Manufacturers (OEMs), Clinical Research Organizations (CROs), Business Process Outsourcing (BPO) Firms) - Market Size, Trends, And Global Forecast 2026-2035
Medical Devices Vigilance Market Overview
• Medical Devices Vigilance market size has reached to $97.66 billion in 2025 • Expected to grow to $149.03 billion in 2030 at a compound annual growth rate (CAGR) of 8.8% • Growth Driver: Surging Medical Device Recalls Fuel Growth In The Medical Device Vigilance Market • Market Trend: AI-Driven Modules Transforming Post-Market Safety Surveillance • North America was the largest region in 2025 and Asia-Pacific is the fastest growing region.What Is Covered Under Medical Devices Vigilance Market?
Medical device vigilance refers to monitoring and reporting adverse events or incidents related to the use of medical devices. It is a critical component of post-market surveillance to ensure the safety and effectiveness of medical devices throughout their lifecycle. It identifies and addresses potential risks associated with medical devices, ensures patient safety, and maintains public trust in the healthcare system. The main delivery modes of medical devices vigilance are on-demand and on-premise. On-demand medical device vigilance delivery mode refers to a system or approach where medical device vigilance services, such as monitoring, reporting, risk assessment, and communication related to adverse events or incidents with medical devices, are provided as needed or requested by healthcare providers, manufacturers, or regulatory authorities. It is used for therapeutics, diagnostics, surgical, research, and other applications by end-users such as original equipment manufacturers (OEMs), clinical research organizations (CROs), and business process outsourcing (BPO) firms.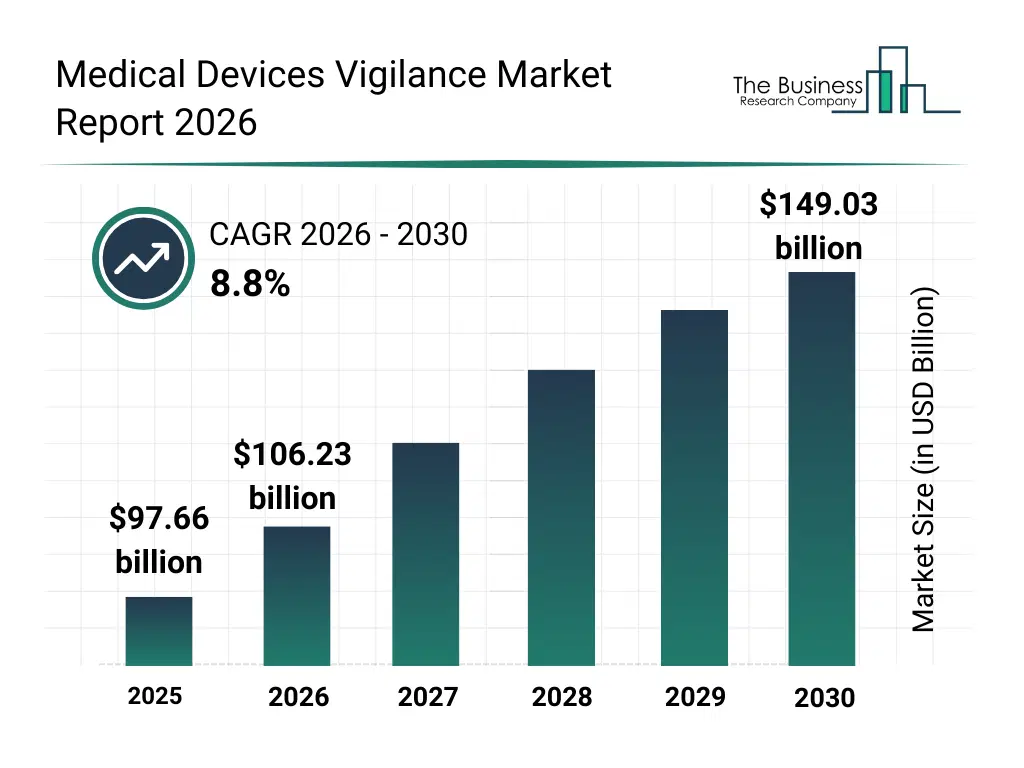
What Is The Medical Devices Vigilance Market Size and Share 2026?
The medical devices vigilance market size has grown strongly in recent years. It will grow from $97.66 billion in 2025 to $106.23 billion in 2026 at a compound annual growth rate (CAGR) of 8.8%. The growth in the historic period can be attributed to increase in reported device-related adverse events, implementation of post-market surveillance regulations, growth in medical device usage, rising patient safety awareness, expansion of global regulatory frameworks.What Is The Medical Devices Vigilance Market Growth Forecast?
The medical devices vigilance market size is expected to see strong growth in the next few years. It will grow to $149.03 billion in 2030 at a compound annual growth rate (CAGR) of 8.8%. The growth in the forecast period can be attributed to growth in connected and complex medical devices, stricter post-market compliance requirements, increasing cross-border device approvals, rising adoption of proactive risk management, demand for continuous safety monitoring. Major trends in the forecast period include strengthening post-market surveillance requirements, rising focus on real-world evidence collection, increasing regulatory scrutiny on adverse event reporting, expansion of global vigilance harmonization, growing demand for faster incident response mechanisms.Global Medical Devices Vigilance Market Segmentation
1) By Delivery Mode: On-Demand, On-Premise 2) By Application: Therapeutics, Diagnostics, Surgical, Research, Other Applications 3) By End-User: Original Equipment Manufacturers (OEMs), Clinical Research Organizations (CROs), Business Process Outsourcing (BPO) Firms Subsegments: 1) By On-Demand: Cloud-Based Solutions, Subscription Services, Remote Monitoring And Reporting 2) By On-Premise: In-House Software Solutions, Local Server Deployments, Customized System ImplementationsWhat Is The Driver Of The Medical Devices Vigilance Market?
The increasing number of medical recalls is expected to propel the growth of the medical device vigilance market going forward. Medical recalls refer to the action taken by regulatory authorities, manufacturers, or distributors to remove or correct medical products that pose a risk to public health or do not comply with regulatory standards. Medical device vigilance entails continuous monitoring of medical devices in the market to identify adverse events, malfunctions, or safety concerns that, upon identification, are reported to regulatory authorities, potentially leading to further investigation and, if warranted, a product recall. For instance, in March 2024, according to Sedgwick, a UK-based risk management and brand protection company, the company reported that in 2023, there were 3,306 medical device recalls in Europe (EU + UK), representing a 20% increase over 2022. Therefore, the increasing number of medical recalls is driving the growth of the medical device vigilance market.Key Players In The Global Medical Devices Vigilance Market
Major companies operating in the medical devices vigilance market are Johnson & Johnson, Intel Corporation, Oracle Corporation, Medtronic, Siemens Healthineers, RELX Group plc, MasterControl Inc., Laerdal Medical, Numerix, Smithers, Omnify Software Inc., Freyr, Sparta Systems, MDI Consultants Inc., Greenlight Guru, Jama Software, Sarjen Systems Pvt. Ltd, Arena Solutions Inc., Xybion Corporation, ZEINCRO Group, Extedo Gmbh, AssurX Inc., AB-Cube, Panacea Pharma Projects Limited, Qvigilance, General Electric (GE) Healthcare, Philips Healthcare, Baxter International, Stryker Corporation, Boston ScientificGlobal Medical Devices Vigilance Market Trends and Insights
Major companies operating in the medical device vigilance market are focusing on innovation in post-market safety surveillance, such as artificial intelligence (AI)-driven modules to automate adverse event reporting, enhance efficiency, and improve signal detection. Artificial intelligence (AI)-driven modules refer to intelligent software components that use artificial intelligence to process, analyze, and prioritize vigilance data for regulatory compliance and patient safety. For instance, in July 2024, AB Cube, a France-based medical software company, launched its enhanced customer vigilance platform to support post-market monitoring of medical devices. The platform consolidates adverse event reporting, trend analysis, and compliance documentation into a single interface; it functions by automating the collection, classification, and reporting of device-related incidents to regulatory authorities; unique features include real-time analytics dashboards, automated regulatory submissions, and customizable alerts for critical safety events; applications span medical device manufacturers, notified bodies, and hospitals; benefits include improved patient safety, reduced regulatory risk, faster incident response, and enhanced overall vigilance efficiency.What Are Latest Mergers And Acquisitions In The Medical Devices Vigilance Market?
In January 2024, GE Healthcare, a US-based medical technology company that offers a diverse portfolio of products, solutions, and services, acquired MIM Software Inc. for an undisclosed amount. The acquisition aims to reinforce GE HealthCare's digital strategy and enhance its imaging portfolio, particularly in medical device vigilance. MIM Software Inc. is a US-based company that provides services related to medical device surveillance through its specialization in medical imaging analysis and artificial intelligence (AI) solutions.Regional Outlook
North America was the largest region in the medical devices vigilance market in 2025. Asia-Pacific is expected to be the fastest-growing region in the forecast period. The regions covered in this market report are Asia-Pacific, South East Asia, Western Europe, Eastern Europe, North America, South America, Middle East, Africa. The countries covered in this market report are Australia, Brazil, China, France, Germany, India, Indonesia, Japan, Taiwan, Russia, South Korea, UK, USA, Canada, Italy, Spain.What Defines the Medical Devices Vigilance Market?
The medical device vigilance market consists of revenues earned by entities by providing services such as adverse event reporting, post-market surveillance, risk management, quality management systems, and regulatory compliance. The market value includes the value of related goods sold by the service provider or included within the service offering. The medical device vigilance market also includes sales of data collection devices, communication devices, data storage and processing devices, medical imaging equipment, and wearable devices. Values in this market are ‘factory gate’ values, that is the value of goods sold by the manufacturers or creators of the goods, whether to other entities (including downstream manufacturers, wholesalers, distributors and retailers) or directly to end customers. The value of goods in this market includes related services sold by the creators of the goods.How is Market Value Defined and Measured?
The market value is defined as the revenues that enterprises gain from the sale of goods and/or services within the specified market and geography through sales, grants, or donations in terms of the currency (in USD unless otherwise specified). The revenues for a specified geography are consumption values that are revenues generated by organizations in the specified geography within the market, irrespective of where they are produced. It does not include revenues from resales along the supply chain, either further along the supply chain or as part of other products.What Key Data and Analysis Are Included in the Medical Devices Vigilance Market Report 2026?
The medical devices vigilance market research report is one of a series of new reports from The Business Research Company that provides market statistics, including industry global market size, regional shares, competitors with the market share, detailed market segments, market trends and opportunities, and any further data you may need to thrive in the medical devices vigilance industry. The market research report delivers a complete perspective of everything you need, with an in-depth analysis of the current and future state of the industry.Medical Devices Vigilance Market Report Forecast Analysis
| Report Attribute | Details |
|---|---|
| Market Size Value In 2026 | $106.23 billion |
| Revenue Forecast In 2035 | $149.03 billion |
| Growth Rate | CAGR of 8.8% from 2026 to 2035 |
| Base Year For Estimation | 2025 |
| Actual Estimates/Historical Data | 2020-2025 |
| Forecast Period | 2026 - 2030 - 2035 |
| Market Representation | Revenue in USD Billion and CAGR from 2026 to 2035 |
| Segments Covered | Delivery Mode, Application, End-User |
| Regional Scope | Asia-Pacific, Western Europe, Eastern Europe, North America, South America, Middle East, Africa |
| Country Scope | The countries covered in the report are Australia, Brazil, China, France, Germany, India, ... |
| Key Companies Profiled | Johnson & Johnson, Intel Corporation, Oracle Corporation, Medtronic, Siemens Healthineers, RELX Group plc, MasterControl Inc., Laerdal Medical, Numerix, Smithers, Omnify Software Inc., Freyr, Sparta Systems, MDI Consultants Inc., Greenlight Guru, Jama Software, Sarjen Systems Pvt. Ltd, Arena Solutions Inc., Xybion Corporation, ZEINCRO Group, Extedo Gmbh, AssurX Inc., AB-Cube, Panacea Pharma Projects Limited, Qvigilance, General Electric (GE) Healthcare, Philips Healthcare, Baxter International, Stryker Corporation, Boston Scientific |
| Customization Scope | Request for Customization |
| Pricing And Purchase Options | Explore Purchase Options |
