
Medical Microbiology Testing Technology Market Report 2026
Global Outlook – By Product Type (Reagents, Instruments, Consumables, Software, Services), By Testing Method (Culture Test, Polymerase Chain Reaction, Immunoassays, Deoxyribonucleic Acid Sequence), By Technology (Cell Culture, Microscopy, Serology), By Application (Infectious Diseases, Non-infectious Diseases, Research And Development, Clinical Trials), By End Use (Hospitals, Diagnostic Laboratories, Pharmaceutical Companies, Research Institutes, Food And Beverage Industry) – Market Size, Trends, Strategies, and Forecast to 2035
Medical Microbiology Testing Technology Market Overview
• Medical Microbiology Testing Technology market size has reached to $6.00 billion in 2025 • Expected to grow to $8.82 billion in 2030 at a compound annual growth rate (CAGR) of 8% • Growth Driver: Rising Demand For Rapid Pathogen Detection And Identification Driving The Growth Of The Market Due To Increasing Infectious Disease Burden And The Need For Faster Clinical Decision-Making • Market Trend: Expansion Of Digital PCR Assays Enhances Pathogen Detection Precision • North America was the largest region in 2025 and Asia-Pacific is the fastest growing region.What Is Covered Under Medical Microbiology Testing Technology Market?
Medical microbiology testing technology refers to the use of specialized laboratory techniques and instruments to detect, identify, and study microorganisms such as bacteria, viruses, fungi, and parasites. It involves analyzing samples to understand microbial characteristics, behaviors, and interactions. This technology enables accurate diagnosis and monitoring of infectious diseases through precise and reliable testing methods. The main product types of medical microbiology testing technology include reagents, instruments, consumables, software, and services. Reagents refer to chemical or biological substances used in laboratory testing to detect, isolate, or quantify microorganisms such as bacteria, viruses, fungi, and parasites. testing using various methods such as culture tests, polymerase chain reaction (PCR), immunoassays, and deoxyribonucleic acid sequence. The technologies include cell culture, microscopy and serology. The applications such as infectious diseases, non-infectious diseases, research and development, and clinical trials and the key end users, including hospitals, diagnostic laboratories, pharmaceutical companies, research institutes, and the food and beverage industry.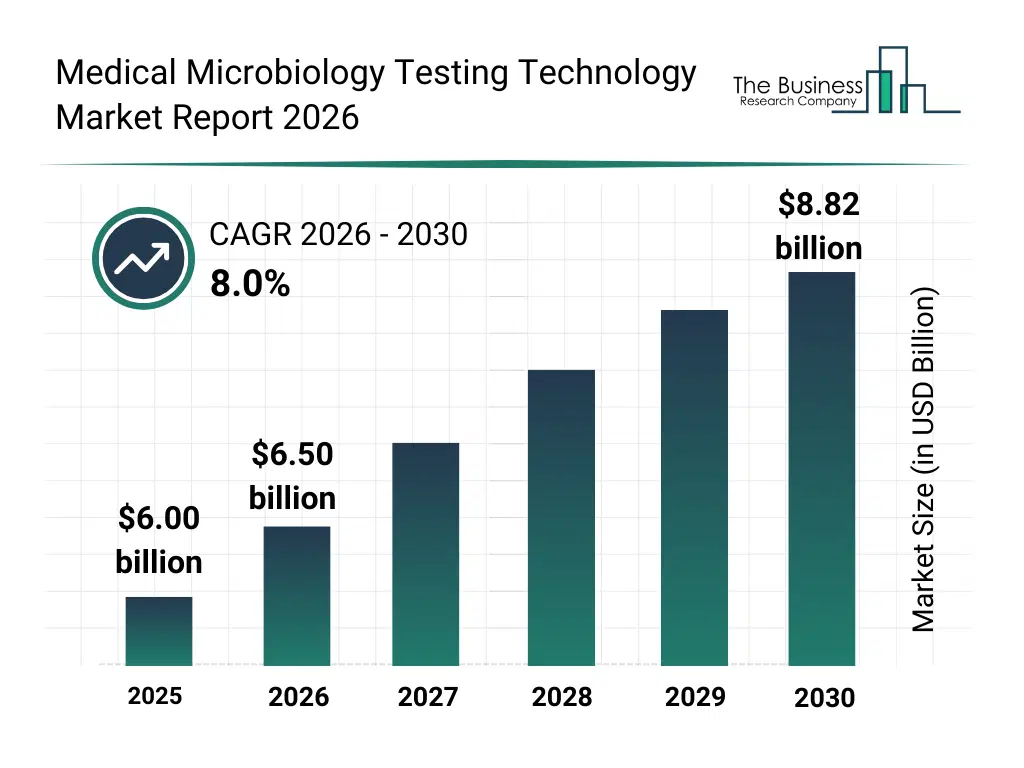
What Is The Medical Microbiology Testing Technology Market Size and Share 2026?
The medical microbiology testing technology market size has grown strongly in recent years. It will grow from $6.00 billion in 2025 to $6.50 billion in 2026 at a compound annual growth rate (CAGR) of 8.2%. The growth in the historic period can be attributed to increasing incidence of infectious diseases, growing antimicrobial resistance awareness, rising healthcare expenditure, expansion of clinical laboratories, increasing adoption of automated systems.What Is The Medical Microbiology Testing Technology Market Growth Forecast?
The medical microbiology testing technology market size is expected to see strong growth in the next few years. It will grow to $8.82 billion in 2030 at a compound annual growth rate (CAGR) of 8.0%. The growth in the forecast period can be attributed to rising demand for rapid and point-of-care testing, growing government initiatives and public health programs, increasing focus on food and pharmaceutical safety testing, expansion in emerging markets, rising integration of laboratory information systems. Major trends in the forecast period include advancements in molecular diagnostics, innovations in high-throughput testing platforms, developments in multiplex and rapid diagnostic tests, increasing research and development in microbiology, technological integration with serological and immunological testing.Global Medical Microbiology Testing Technology Market Segmentation
1) By Product Type: Reagents, Instruments, Consumables, Software, Services 2) By Testing Method: Culture Test, Polymerase Chain Reaction, Immunoassays, Deoxyribonucleic Acid Sequence 3) By Technology: Cell Culture, Microscopy, Serology 4) By Application: Infectious Diseases, Non-infectious Diseases, Research And Development, Clinical Trials 5) By End Use: Hospitals, Diagnostic Laboratories, Pharmaceutical Companies, Research Institutes, Food And Beverage Industry Subsegments: 1) By Reagents: Culture Media, Detection Reagents, Antigen And Antibody Reagents, Molecular Diagnostic Kits, Stains And Dyes, Antibiotic Or Susceptibility Testing Reagents 2) By Instruments: Microscopes, Incubators, Automated Culture Systems Or Microbiology Analyzers, Spectrophotometers, Centrifuges, Colony Counters Or Plate Readers, Autoclaves Or Sterilizers, Bioreactors Or Environmental Monitoring Systems 3) By Consumables: Petri Dishes, Test Tubes, Pipette Tips, Microplates, Sample Swabs Or Collection Containers, Slides And Cover Slips, Disposable Filters Or Membranes, Disposable Consumable Kits 4) By Software: Laboratory Information Management Software, Data Analysis Or Result Reporting Software, Quality Control Or Workflow Management Software, Automated Instrument Control Software, Database Or Reference Strain Or Pathogen Registry Software 5) By Services: Contract Testing Or Outsourced Microbiological Testing, Equipment Maintenance And Calibration, Training And Technical Support, Consulting And Compliance Services, Validation And Quality Assurance ServicesWhat Is The Driver Of The Medical Microbiology Testing Technology Market?
The rising demand for rapid pathogen detection and identification is expected to propel the growth of the medical microbiology testing technology market going forward. Rapid pathogen detection and identification refer to the use of diagnostic technologies that quickly and accurately detect microbes such as bacteria, viruses, and other pathogens directly from clinical samples without extended culture periods, thereby shortening time to diagnosis and enabling faster clinical decision-making. The demand for rapid pathogen detection is increasing as public health systems and clinical laboratories worldwide aim to improve turnaround times and respond effectively to infectious disease outbreaks and antimicrobial resistance pressures. Medical microbiology testing technology supports rapid pathogen detection by identifying infectious agents quickly and accurately through advanced diagnostic methods, supporting timely treatment and reducing infection spread. For instance, in November 2025, according to the Infectious Disease Surveillance Center, a Japan-based public health organization, data from April 2025 indicate that between 2,104 and 2,153 respiratory pathogen tests were conducted per pathogen category in a single reporting period, with positivity rates for severe acute respiratory syndrome coronavirus 2, influenza, respiratory syncytial virus, and parainfluenza ranging from 5.7% to 11.5%. Therefore, the rising demand for rapid pathogen detection and identification is driving the growth of the medical microbiology testing technology industry.Key Players In The Global Medical Microbiology Testing Technology Market
Major companies operating in the medical microbiology testing technology market are F. Hoffmann-La Roche Ltd., Thermo Fisher Scientific Inc., Siemens Healthineers AG, Danaher Corporation, Becton Dickinson and Company, bioMérieux SA, Hologic Inc., Sysmex Corporation, Bruker Corporation, Bio-Rad Laboratories Inc., QIAGEN N.V., Abbott Laboratories, Meridian Bioscience Inc., Fujirebio Diagnostics Inc., Seegene Inc., Oxford Nanopore Technologies plc, Molbio Diagnostics Pvt. Ltd., Hardy Diagnostics, HiMedia Laboratories Pvt. Ltd., Accelerate Diagnostics Inc., Liofilchem S.r.l.Global Medical Microbiology Testing Technology Market Trends and Insights
Major companies operating in the medical microbiology testing technology market are focusing on developing advanced molecular analysis products, such as digital PCR (dPCR) assays, to boost research efficiency, enhance detection accuracy, and improve public health surveillance. Digital PCR assays refer to highly sensitive technologies that can detect and precisely quantify specific DNA sequences, even at very low levels, enabling accurate pathogen detection and monitoring. For instance, in June 2024, QIAGEN N.V., a Netherlands‑based provider of molecular biology and life sciences solutions, launched 35 new wet-lab tested digital PCR Microbial DNA Detection Assays for its QIAcuity digital PCR platform. These assays are available on QIAGEN’s research platform GeneGlobe and are designed to target a wide range of pathogens responsible for tropical diseases (e.g., Dengue, Chikungunya, Malaria), sexually transmitted infections (STIs), and urinary tract infections (UTIs).What Are Latest Mergers And Acquisitions In The Medical Microbiology Testing Technology Market?
In June 2025, bioMérieux S.A., a France-based provider of in vitro diagnostics solutions, acquired Day Zero Diagnostics for approximately $25 million. With this acquisition, bioMérieux aims to strengthen its next-generation sequencing (NGS) capabilities and enhance its rapid diagnostics portfolio to better address antibiotic-resistant infections and improve the management of life-threatening infectious diseases. Day Zero Diagnostics is a US-based diagnostic company developing sequencing-based microbiology testing technology that provides rapid identification of pathogens and antimicrobial resistance profiles directly from clinical samples.Regional Insights
North America was the largest region in the medical microbiology testing technology market in 2025. Asia-Pacific is expected to be the fastest-growing region in the forecast period. The regions covered in this market report are Asia-Pacific, South East Asia, Western Europe, Eastern Europe, North America, South America, Middle East, Africa. The countries covered in this market report are Australia, Brazil, China, France, Germany, India, Indonesia, Japan, Taiwan, Russia, South Korea, UK, USA, Canada, Italy, Spain.What Defines the Medical Microbiology Testing Technology Market?
The medical microbiology testing technology market consists of revenues earned by entities by providing services such as clinical diagnostics, pathogen identification, antimicrobial susceptibility testing, molecular and genetic testing, infection monitoring, outbreak investigation, and laboratory support. The market value includes the value of related goods sold by the service provider or included within the service offering. The medical microbiology testing technology market also includes sales of microbiology analyzers, culture media, reagents and kits, automated identification systems, diagnostic instruments, laboratory consumables, and molecular testing equipment. Values in this market are ‘factory gate’ values, that is the value of goods sold by the manufacturers or creators of the goods, whether to other entities (including downstream manufacturers, wholesalers, distributors and retailers) or directly to end customers. The value of goods in this market includes related services sold by the creators of the goods.How is Market Value Defined and Measured?
The market value is defined as the revenues that enterprises gain from the sale of goods and/or services within the specified market and geography through sales, grants, or donations in terms of the currency (in USD unless otherwise specified). The revenues for a specified geography are consumption values that are revenues generated by organizations in the specified geography within the market, irrespective of where they are produced. It does not include revenues from resales along the supply chain, either further along the supply chain or as part of other products.What Key Data and Analysis Are Included in the Medical Microbiology Testing Technology Market Report 2026?
The medical microbiology testing technology market research report is one of a series of new reports from The Business Research Company that provides market statistics, including industry global market size, regional shares, competitors with the market share, detailed market segments, market trends and opportunities, and any further data you may need to thrive in the medical microbiology testing technology industry. The market research report delivers a complete perspective of everything you need, with an in-depth analysis of the current and future state of the industry.Medical Microbiology Testing Technology Market Report Forecast Analysis
| Report Attribute | Details |
|---|---|
| Market Size Value In 2026 | $6.50 billion |
| Revenue Forecast In 2035 | $8.82 billion |
| Growth Rate | CAGR of 8.2% from 2026 to 2035 |
| Base Year For Estimation | 2025 |
| Actual Estimates/Historical Data | 2020-2025 |
| Forecast Period | 2026 - 2030 - 2035 |
| Market Representation | Revenue in USD Billion and CAGR from 2026 to 2035 |
| Segments Covered | Product Type, Testing Method, Technology, Application, End Use |
| Regional Scope | Asia-Pacific, Western Europe, Eastern Europe, North America, South America, Middle East, Africa |
| Country Scope | The countries covered in the report are Australia, Brazil, China, France, Germany, India, ... |
| Key Companies Profiled | F. Hoffmann-La Roche Ltd., Thermo Fisher Scientific Inc., Siemens Healthineers AG, Danaher Corporation, Becton Dickinson and Company, bioMérieux SA, Hologic Inc., Sysmex Corporation, Bruker Corporation, Bio-Rad Laboratories Inc., QIAGEN N.V., Abbott Laboratories, Meridian Bioscience Inc., Fujirebio Diagnostics Inc., Seegene Inc., Oxford Nanopore Technologies plc, Molbio Diagnostics Pvt. Ltd., Hardy Diagnostics, HiMedia Laboratories Pvt. Ltd., Accelerate Diagnostics Inc., Liofilchem S.r.l. |
| Customization Scope | Request for Customization |
| Pricing And Purchase Options | Explore Purchase Options |
